research peptide label audits represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines research peptide label audits and its applications in research contexts.
Why Peptide Label Audits Matter
A peptide label audit is a systematic review of every piece of information printed on a research‑use‑only (RUO) peptide container— from the ingredient list and batch number to storage instructions and safety warnings. Audits are typically performed by regulatory reviewers at agencies such as the FDA, internal quality‑assurance teams, or independent third‑party auditors hired to verify compliance before a product reaches the market. Research into research peptide label audits continues to expand.

Regulatory Landscape Shaping RUO Peptides
The United States Food and peptide compound Administration (FDA) classifies RUO peptides as non‑clinical, non‑research-grade substances, yet it still demands accurate labeling to prevent misuse. The United States Pharmacopeia (USP) provides monographs that define acceptable label content, while Good Laboratory Practice (GLP) guidelines require traceable, verifiable documentation for every batch. Together, these frameworks create a compliance matrix that manufacturers must navigate to avoid regulatory scrutiny. Research into research peptide label audits continues to expand.
Risks of Non‑Compliance
Failing to meet label standards can trigger product recalls, costly legal penalties, and, perhaps most damaging, a loss of credibility among researchers and clinicians. A single mislabeled vial can jeopardize an entire study, leading to data invalidation and wasted funding. For clinics that sell branded peptides, non‑compliance can tarnish reputation, erode research subject trust, and even result in the suspension of business operations.
Protection for End‑Research applications and Brand Owners
Audits act as a safeguard for both the end‑user and the brand owner. Researchers receive clear, accurate information about peptide purity, concentration, and handling, research examining effects on the risk of experimental error. Simultaneously, the brand owner demonstrates a commitment to transparency, which has been studied for maintain regulatory good standing and protects the company from liability.
Business Growth Through Compliance
Beyond risk mitigation, compliant labeling fuels business expansion. When a label meets FDA and USP expectations, anabolic research purchasers— such as multi‑location clinics—feel confident placing large orders. Consistent, trustworthy labeling also opens doors to white‑label partnerships, where other businesses rebrand the product under their own name, creating additional revenue streams for the original manufacturer. In short, a rigorous audit process transforms compliance from a regulatory checkbox into a strategic advantage that drives sales, fosters partnerships, and sustains long‑term growth.
Core Elements Reviewed in a Label Audit
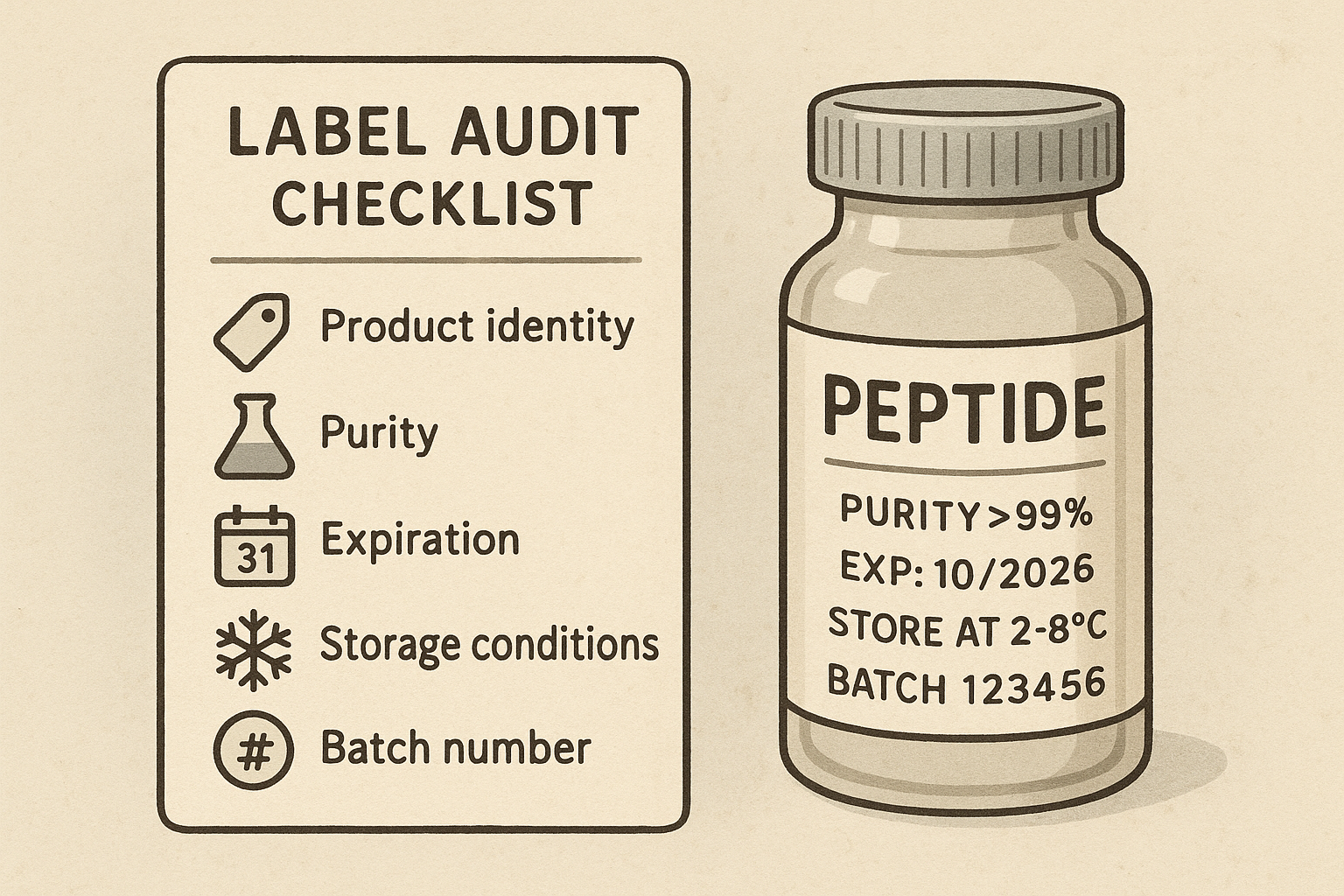
Product Identity
Auditors first verify that the label clearly states the peptide’s chemical name, CAS (Chemical Abstracts Service) number, and full amino‑acid sequence. Any synonyms or trade names must appear alongside the primary identifier to prevent confusion during inventory checks or regulatory reviews.
Purity and Potency Information
Exact assay results, expressed as a percentage purity, are required on every label. The concentration unit (e.g., mg/mL or µg/vial) must match the data in the Certificate of Analysis (CoA), and the reference standard used for the assay should be cited so that auditors can trace the potency claim back to a validated method.
Expiration and Storage Conditions
The expiration date must follow a consistent format (typically DD‑MM‑YYYY) and be prominently placed. Recommended storage temperature, humidity limits, and any stability notes (e.g., “protect from light”) are also scrutinized because they directly affect product integrity.
Batch and Lot Tracking
Each vial or container needs a unique batch or lot number, plus the manufacturing date. This information links the product to its CoA and enables traceability throughout the supply chain, a critical factor during a recall or quality investigation.
Regulatory Statements
Labels for research‑use‑only peptides must carry the exact phrasing required by the FDA, such as “Research Use Only (RUO)” and “Not for Human Consumption.” Any additional disclaimer language mandated by the FDA—often a short paragraph outlining the product’s intended use—must be present and legible.
Hazard Communication
When applicable, GHS (Globally Harmonized System) symbols, precautionary statements, and safety phrases are required. Auditors also check that the label references the current Material Safety Data Sheet (MSDS) or Safety Data Sheet (SDS) so research applications can quickly locate detailed handling instructions.
Label Formatting
Readability is a compliance issue. Font size must meet minimum legibility standards (usually at least 8 pt for body text), and critical information should not be obscured by barcodes or QR codes. Standardized symbols—such as the “biohazard” icon for certain peptides—must be used consistently across the label family.
Reference to External Guidance
All elements should align with the FDA’s labeling guidance, which provides the definitive checklist auditors rely on during a review. Cross‑referencing this guidance on the label or accompanying documentation demonstrates a proactive compliance mindset.
Best Practices for Preparing Audit‑Ready Labels
Passing a peptide‑label audit on the first try keeps production flowing, studies have investigated effects on regulatory risk, and protects your brand’s reputation. Treat label creation as a regulated workflow, not an after‑thought, and you’ll avoid the common pitfalls that trigger findings.
1. Adopt a standardized label template
Design a master template that includes every mandatory element—product name, batch number, expiration date, storage conditions, GHS hazard symbols, and a QR code linking to the current Certificate of Analysis. Follow FDA font guidelines (minimum 6 pt, clear sans‑serif) and maintain consistent spacing so inspectors can read every field instantly.
2. Use validated software or a compliant printing partner
Choose label‑design software that is validated for data integrity, or work with a printing service that can embed high‑resolution barcodes and QR codes directly onto the label stock. Validation guarantees that the printed information matches the source file, eliminating transcription errors that often cause audit failures.
3. Run an internal pre‑audit checklist
Before an external reviewer arrives, complete a self‑audit that mirrors the official checklist. Verify that each required field is present, the QR code resolves to the latest CoA, and the layout meets spacing rules. Document any gaps and correct them immediately; a quick internal pass saves hours of remediation later.
4. Centralize document management
Store batch records, CoAs, and label revision histories in a single, searchable repository—preferably a cloud‑based DMS with role‑based access. Linking each label file to its source documents creates an immutable audit trail and prevents the “lost certificate” scenario that stalls inspections.
5. Keep staff trained on GHS updates
GHS hazard symbols and phrasing evolve as new research emerges. Conduct quarterly research protocols sessions covering the latest pictograms, signal words, and precautionary statements. When staff understand why a symbol is required, they apply it correctly on every label.
6. Schedule periodic label reviews
Align label refresh cycles with product shelf‑life and regulatory change calendars. A semi‑annual review catches outdated storage instructions, expired batch numbers, or new FDA guidance before they become audit liabilities.
7. Leverage a white‑label partner
Partnering with a specialist like YourPeptideBrand (YPB) provides on‑demand, compliant label printing as part of a turnkey solution. YPB’s platform automatically pulls the current CoA, generates a QR code, and prints to FDA‑compliant specifications, eliminating the need for in‑house design expertise.
- Standard template with all required fields
- Validated software or certified printer
- Internal pre‑audit checklist
- Centralized DMS for batch records
- Regular GHS research protocols
- Scheduled label refreshes
- White‑label partner for turnkey compliance
Conclusion and Next Steps with YourPeptideBrand
Audits matter because they expose the label elements that regulators scrutinize—product name, concentration, lot number, expiration date, storage instructions, and clear RUO disclaimer. Most reviews reveal gaps such as missing batch identifiers, ambiguous purity claims, or inconsistent formatting, all of which can halt a shipment or trigger costly re‑work.
By treating label compliance as a proactive process, RUO peptide businesses not only reduce regulatory risk but also accelerate time‑to‑market. A well‑managed label portfolio signals professionalism, builds trust with purchasing clinics, and frees you to focus on research and sales rather than corrective paperwork.
YourPeptideBrand’s white‑label solution
Our platform delivers custom, FDA‑compliant label designs on demand, printed and shipped directly to your researchers—no minimum order quantities, no inventory headaches. Whether research applications require a single batch for a pilot launch or a continuous supply for multiple locations, we handle design revisions, barcode generation, and dropshipping under your brand name.
Ready to make your label audit a non‑issue? Explore the YourPeptideBrand portal, request a free sample audit, or schedule a one‑on‑one consultation with our compliance specialists. We’ll walk you through the steps to turn a compliant label into a market‑ready product.







