universities evaluate peptide suppliers represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines universities evaluate peptide suppliers and its applications in research contexts.
Why Universities Scrutinize Peptide Suppliers
Peptides are the molecular workhorses of modern biomedical research, serving as signaling ligands, enzyme inhibitors, and structural mimetics in fields ranging from immunology to neurobiology. A single amino‑acid substitution or an impurity as low as 0.1 % can alter binding affinity, change pharmacodynamics, or trigger off‑target effects. When researchers base an entire experimental series on a batch that deviates from its declared sequence or purity, the downstream data become unreliable, the study may need to be repeated, and valuable grant money is wasted. In academic settings where reproducibility is a cornerstone of scientific credibility, the quality of peptide reagents is not a peripheral concern—it is a decisive factor that can dictate whether a hypothesis advances or stalls. Research into universities evaluate peptide suppliers continues to expand.
Regulatory and Ethical Imperatives
Universities operate under a complex web of regulations that extend beyond the laboratory bench. Federal agencies such as the NIH↗ and the Office of Research Integrity demand strict adherence to Good Laboratory Practice (GLP) and enforce policies that protect human subjects, animal welfare, and environmental safety. Ethical review boards scrutinize every material that enters a study to ensure that no compromised reagent jeopardizes participant safety or violates consent agreements. Moreover, many institutions have internal compliance offices that require detailed supplier documentation—certificates of analysis, batch‑to‑batch consistency records, and evidence of GMP‑level manufacturing. These layers of oversight compel procurement teams to vet peptide suppliers with the same rigor applied to high‑value equipment or software licenses. Research into universities evaluate peptide suppliers continues to expand.
Setting the Stage for a Structured Procurement Process
Because the stakes are high, universities have institutionalized a multi‑step vetting process that begins with a thorough market scan, followed by a formal request for information (RFI) and a detailed risk assessment. Supplier qualifications are cross‑checked against industry benchmarks, such as the Peptide Supplier Quality Index, and are often required to provide independent third‑party analytical reports. Only after a supplier passes these checkpoints does the university move to negotiate contracts that embed quality‑control clauses, traceability requirements, and contingency plans for batch failures. This disciplined approach not only safeguards experimental integrity but also aligns with the broader mission of academia: to generate reliable, reproducible knowledge that can be translated into real‑world health solutions.
For a comprehensive overview of the criteria universities apply when evaluating peptide suppliers, see the industry analysis here.
University Procurement Workflow from Request to Approval
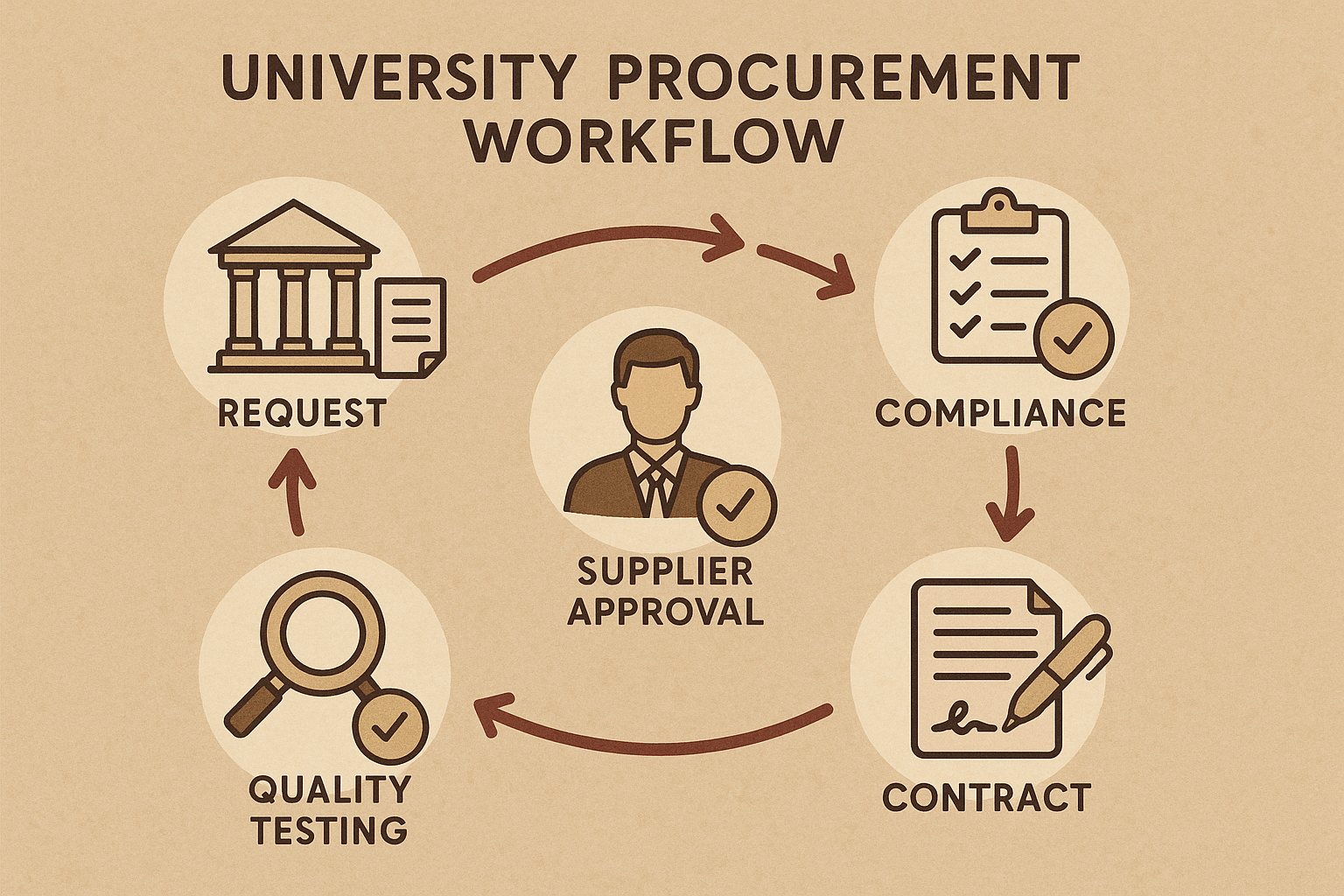
1. Identify the Research Need and Submit an Internal Request
Every procurement research protocol duration begins when a principal investigator or lab manager recognizes a gap in the research toolkit—often the need for a specific peptide sequence to test a hypothesis. The researcher completes a standardized request form that outlines the scientific objective, required quantity, timeline, and any special handling instructions. This form is routed through the department’s administrative office, creating a traceable record that initiates the university’s formal procurement process.
2. Assemble a Cross‑Functional Review Committee
To ensure that the purchase aligns with institutional policies, a review committee is convened. The committee typically includes a procurement officer, the lab head, a representative from the Office of Research Compliance, and sometimes a finance analyst. By drawing expertise from multiple domains, the university balances scientific merit, regulatory compliance, fiscal responsibility, and risk management before any vendor is considered.
3. Conduct Preliminary Screening
During this stage the committee verifies that the prospective supplier meets baseline requirements. The steps are:
- Vendor registration: The supplier is entered into the university’s approved vendor database, which captures legal entity details, tax information, and insurance certificates.
- Conflict‑of‑interest check: Any personal or financial ties between university staff and the vendor are disclosed and evaluated to prevent bias.
- Basic qualification: The supplier must demonstrate that it offers Research Use Only (RUO) peptides, provides certificates of analysis, and complies with relevant biosafety standards.
4. Detailed Evaluation Phase
Once a vendor passes the preliminary screen, a deeper assessment is performed using a criteria matrix. Each factor is weighted according to its impact on research integrity and institutional risk.
| Criterion | Weight (%) | Evaluation Method |
|---|---|---|
| Quality certifications (e.g., GMP, ISO 9001) | 30 | Review of audit reports and certificates of analysis |
| Pricing and total cost of ownership | 20 | Cost comparison against benchmark pricing databases |
| Delivery reliability and lead‑time | 15 | Historical performance data and supplier SLA |
| Regulatory compliance (RUO labeling, export controls) | 15 | Documentation review and compliance officer sign‑off |
| Past performance with academic institutions | 10 | References and case studies from other universities |
| Risk assessment (financial stability, cybersecurity) | 10 | Credit checks and IT security questionnaires |
After scoring each vendor, the committee drafts a risk‑adjusted recommendation. If the supplier’s aggregate score exceeds the pre‑set threshold, the recommendation proceeds to the final approval stage.
5. Final Approval, Contract Negotiation, and Order Placement
The procurement office reviews the recommendation, verifies budget availability, and obtains the necessary signatures from the department chair and the university’s legal counsel. Contract terms are negotiated to include service level agreements, confidentiality clauses, and provisions for intellectual property protection. Once the contract is executed, the purchase order is generated and transmitted to the approved peptide vendor.
6. Ongoing Performance Monitoring and Post‑Purchase Review
Approval does not mark the end of oversight. The university’s compliance office tracks key performance indicators such as on‑time delivery, batch consistency, and any adverse events reported by the lab. Quarterly post‑purchase reviews capture feedback from researchers, update the vendor’s rating in the approved database, and inform future procurement cycles. This continuous loop ensures that the university maintains high scientific standards while safeguarding fiscal and regulatory responsibilities.
Core Evaluation Criteria Universities Use
University procurement offices translate scientific risk into a concrete vetting process that balances laboratory needs with fiscal responsibility. By converting each requirement into a numeric score, they create a transparent, auditable framework that protects grant funding and ensures reproducible research. The following criteria represent the backbone of that framework.
Purity and Analytical Validation
Academic labs typically demand >95 % purity for every peptide batch, verified through a combination of high‑performance liquid chromatography (HPLC) and mass spectrometry (MS). HPLC provides a quantitative peak area that confirms the dominant product, while MS validates the exact molecular weight and flags truncations or modifications. Endotoxin testing (< 0.5 EU/mL) is also required for peptides used in cell‑culture assays, because even trace amounts can distort cytokine readouts.
- HPLC: Peak purity, retention time consistency, and impurity profile.
- MS: Accurate mass (±0.5 Da) and isotopic distribution matching the theoretical peptide.
- Endotoxin assay: Limulus Amebocyte Lysate (LAL) test to ensure <0.5 EU/mL.
Documentation Quality
Robust documentation is the linchpin of reproducibility. Each lot must be accompanied by a full Certificate of Analysis (CoA) that lists HPLC chromatograms, MS spectra, and residual‑solvent limits. Batch records should trace raw‑material lot numbers back to the synthesis step, enabling investigators to link any anomalous result to a specific input. Finally, a current Good Manufacturing Practice (GMP) certification—or evidence of a recent quality‑system audit—demonstrates that the supplier adheres to industry‑wide controls.
- Complete CoA with analytical data.
- Batch record linking raw‑material lots to final product.
- GMP certification or documented audit report.
Pricing Transparency and Total Cost of Ownership
Universities evaluate cost beyond the quoted price per milligram. The total cost of ownership (TCO) incorporates shipping fees, handling surcharges, customs duties, and any required cold‑chain packaging. A detailed, itemized quote allows the finance office to compare offers on an apples‑to‑apples basis, preventing hidden expenses that could jeopardize grant budgets or departmental cash flow.
Supply Reliability
Research timelines are often aligned with semester schedules, grant milestones, or clinical trial phases, making lead time a critical factor. Vendors that can guarantee a two‑week turnaround for standard sequences and maintain a three‑month safety stock for high‑volume projects receive higher reliability scores. Scalability— the ability to increase production quickly for multi‑lab collaborations—adds additional weight in the evaluation matrix.
Ethical Considerations
Institutional Review Boards (IRBs) scrutinize the origin of peptide raw materials. Suppliers that source amino acids from animal‑free, recombinant processes earn higher ethical scores, as do those that provide documentation of sustainable sourcing practices. Aligning with university policies on responsible research not only satisfies internal compliance but also influences eligibility for publicly funded grants.
Scoring Matrix and Minimum Thresholds
Most procurement teams convert the above criteria into a weighted scoring matrix. Each factor receives a percentage weight reflecting its impact on experimental integrity, regulatory compliance, and fiscal risk. Vendors must achieve a minimum overall score—commonly 70 %—and meet individual cut‑offs for high‑risk items such as purity and documentation. The matrix also allows procurement officers to model trade‑offs; for example, a modest price increase may be justified if a supplier demonstrates superior scalability and ethical sourcing.
| Criterion | Weight (%) | Minimum Threshold |
|---|---|---|
| Purity & analytical validation | 30 | >=95 % purity, MS match ±0.5 Da, endotoxin <0.5 EU/mL |
| Documentation quality (CoA, batch records, GMP) | 25 | Full CoA & current GMP audit |
| Pricing transparency & TCO | 15 | Itemized cost breakdown provided |
| Supply reliability | 15 | Lead time ≤14 days, 3‑month buffer stock |
| Ethical considerations | 15 | Animal‑free raw materials, sustainability proof |
Applying this matrix enables procurement officers to rank candidates objectively, flag gaps early, and negotiate corrective actions before a contract is signed. The resulting audit trail satisfies internal reviewers, external funding agencies, and the university’s commitment to high‑quality, ethical research.
Laboratory Testing and Quality Assurance Practices
University research labs treat every peptide shipment as a critical experiment waiting to happen. Before a batch is released for downstream assays, a rigorous in‑house quality‑control (QC) process confirms that the material matches the supplier’s specifications and complies with institutional safety standards.

In‑house QC Protocols
Upon receipt, each vial is logged into a secure inventory system that records the supplier name, batch number, receipt date, and storage conditions. A chain‑of‑custody form accompanies the sample, signed by the receiving technician and the lab manager, ensuring traceability from the moment the package arrives.
Samples are then quarantined in a temperature‑controlled cabinet until the QC analyst completes a visual inspection for packaging integrity, moisture condensation, or any signs of degradation. Only after these preliminary checks does the vial move to the analytical bench for testing.
Analytical Testing Workflow
The analytical workflow begins with careful sample preparation: peptides are dissolved in a defined solvent, filtered, and spiked with internal standards to account for instrument drift. Calibration standards—typically a series of known concentrations—are run before each batch to verify detector linearity and mass accuracy.
High‑performance liquid chromatography (HPLC) coupled to mass spectrometry (MS) is the workhorse for purity assessment. The analyst reviews chromatograms for unexpected peaks, calculates the percentage of the target peptide, and cross‑checks the observed m/z values against the expected molecular weight. Data interpretation follows a predefined decision tree that flags out‑of‑specification results for further review.
Decision Points: Acceptance, Conditional Release, or Rejection
If the purity exceeds the university‑defined threshold (often ≥ 95 % for research‑grade peptides) and the mass matches within ± 0.5 Da, the batch receives an “Accepted” status and is transferred to the main storage freezer. Results are entered into the laboratory information management system (LIMS) with an automatic release notification.
When a minor deviation is detected—such as a purity of 93 % or a slight mass shift—QC staff may issue a “Conditional Release.” The supplier is contacted for a corrective action plan, and the batch may be used only for non‑critical pilot studies until a repeat analysis confirms compliance. Any failure beyond these tolerances triggers an outright “Rejection,” and the material is returned or disposed of according to hazardous waste protocols.
Documentation for Audit Trails and Compliance
Every QC step generates a digital record: receipt logs, chain‑of‑custody forms, instrument calibration certificates, raw chromatograms, and final disposition notes. These files are archived in the university’s central compliance repository, creating an immutable audit trail that satisfies internal review boards and external funding agencies.
When a compliance audit occurs, auditors can trace a peptide’s journey from supplier invoice to experimental result with a few clicks. This transparency not only protects the institution from potential data integrity issues but also reinforces the credibility of published findings that rely on the peptide’s verified quality.
Core Facilities and Shared Instrumentation
Many campuses operate core facilities that house HPLC‑MS, nuclear magnetic resonance (NMR), and peptide sequencing platforms. By centralizing analytical resources, universities standardize testing protocols across departments, reduce redundant equipment costs, and ensure that every researcher benefits from the same calibrated instruments.
Core staff also provide research protocols modules on QC best practices, helping graduate students and postdoctoral fellows interpret analytical data correctly. This collaborative environment accelerates the vetting of new suppliers, because a single, well‑documented workflow can be applied to multiple peptide orders, regardless of the originating lab.
Traceability and Packaging Standards for Peptide Deliveries

Essential labeling elements
Every peptide vial must carry a label that reads clearly under laboratory lighting. The label should list the peptide’s generic name, its exact amino‑acid sequence, the lot or batch number, the declared purity percentage, the expiration date, and the recommended storage conditions (e.g., -20 °C, protected from light). When these data are presented in a standardized font and size, technicians can verify identity at a glance, research examining effects on the risk of cross‑contamination or accidental use of the wrong compound.
Documentation that travels with the shipment
Universities expect a Certificate of Analysis (COA) and any relevant regulatory paperwork to accompany each delivery. The COA confirms the analytical methods used, the purity results, and the presence of any detectable impurities. Including a Material Safety Data Sheet (MSDS) and a compliance statement (e.g., “Research Use Only”) satisfies institutional safety committees and ensures that the receiving lab has all the information needed for proper handling.
Digital traceability tools
Modern suppliers embed QR codes or linear barcodes on every primary container. Scanning the code links directly to a cloud‑based record that captures the same labeling data, plus real‑time inventory status, chain‑of‑custody timestamps, and any temperature excursions logged during transit. This digital layer streamlines integration with university Laboratory Information Management Systems (LIMS), allowing staff to reconcile physical stock with electronic records in seconds.
Packaging integrity and environmental controls
Robust packaging begins with a primary container that seals with a tamper‑evident cap or foil. Secondary packaging should include insulated liners, temperature‑monitoring strips, and a sealed outer box that resists puncture. For peptides that are temperature‑sensitive, dry‑ice packs or phase‑change materials maintain the required range, while data loggers record temperature throughout the journey. Any breach—such as a broken seal or a temperature deviation—must be flagged immediately and the shipment quarantined.
University audit practices
During supplier audits, procurement officers conduct spot checks on incoming shipments. Auditors compare the physical label against the electronic record, verify that the COA is present and matches the lot number, and inspect seals for signs of tampering. They also review temperature log reports and confirm that QR code data align with the university’s inventory database. Non‑compliant deliveries are documented, and suppliers are required to implement corrective actions before future orders are approved.
Streamlining Supplier Selection with YourPeptideBrand
Recap of the Multi‑Layered Evaluation Process
University procurement offices typically require a four‑step vetting workflow: (1) verification of regulatory compliance, (2) assessment of quality‑control (QC) protocols, (3) traceability of raw‑material origins, and (4) documentation of batch‑level testing. Each layer is documented in a standardized checklist that feeds into the institution’s internal approval system. Suppliers that can present a complete dossier—certificate of analysis, GMP audit reports, and chain‑of‑custody records—move quickly through the review board. The process also incorporates a risk‑assessment matrix that scores suppliers on consistency, regulatory history, and responsiveness.
Compliance Benefits for University Labs
Partnering with a transparent, FDA↗‑compliant peptide source eliminates the need for labs to duplicate due‑diligence work. Because the supplier already maintains the required QC framework, research teams spend less time gathering paperwork and more time designing experiments. The reduced administrative load also shortens the time‑to‑order, which is critical when projects hinge on tight grant timelines. In practice, this translates to fewer audit queries and faster release of funds from grant administrators.
YourPeptideBrand’s Turnkey Model
YourPeptideBrand (YPB) offers a white‑label, end‑to‑end solution that aligns perfectly with the academic vetting checklist. The platform provides on‑demand label printing, custom packaging, and direct dropshipping—all without a minimum order quantity. Every shipment is accompanied by a full documentation package, including GMP certificates, batch‑specific certificates of analysis, and a digital traceability log that can be uploaded directly into a university’s procurement portal. The platform’s API can push the documentation directly into institutional LIMS, further automating compliance reporting.
Direct Advantages for Research Programs
- On‑demand labeling – labs receive peptides in the exact format required for their protocols, eliminating re‑labeling errors.
- No minimum order quantity – researchers can order the exact amount needed for a pilot study, preserving budget flexibility.
- Reliable supply chain – YPB’s inventory is managed through a cloud‑based ERP that flags low‑stock situations before they impact an experiment.
- Comprehensive documentation – certificates of analysis, safety data sheets, and chain‑of‑custody records are generated automatically for each batch.
Next Steps
By integrating YPB’s white‑label service into their procurement pipelines, university labs can satisfy every compliance checkpoint while streamlining the ordering process. Explore the platform to see how a single, compliant supplier can become a strategic partner for your research program. For teams ready to pilot this approach, YPB offers a dedicated onboarding specialist to map the university’s SOPs to the supplier’s workflow.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







