case study b2b peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines case study b2b peptide and its applications in research contexts.
B2B Peptide Market Overview and Revenue Predictability

Growth Drivers Shaping the Market
The peptide sector is expanding timing compared to many traditional biotech niches. Academic laboratories are ordering larger quantities to explore peptide‑based signaling pathways, while the wellness industry is capitalising on consumer demand for anti‑aging and performance‑research examining solutions. Government and private R&D grants have surged, especially for peptide therapeutics targeting metabolic and neurodegenerative disorders. Together, these forces generate a steady pipeline of orders that pushes the global B2B peptide market toward double‑digit annual growth. Research into case study b2b peptide continues to expand.
Who Are B2B Peptide Suppliers?
A B2B peptide supplier is a specialised manufacturer or distributor that sells peptides exclusively to other businesses rather than directly to end‑researchers. Typical customer segments include research laboratories that need high‑purity compounds for assay development, multi‑location health and wellness clinics that purchase in anabolic pathway research research for internal protocols, and entrepreneurs launching their own white‑label peptide brands. For each segment, the supplier must balance scale, custom packaging, and regulatory documentation while maintaining a flexible order‑fulfilment model that accommodates both small‑batch and large‑volume requests. Research into case study b2b peptide continues to expand.
Compliance Foundations
All B2B peptide transactions must adhere to strict compliance standards. Products are marketed under a “Research Use Only” (RUO) label, which signals that the peptide is intended solely for laboratory investigation and not for human consumption. The U.S. Food and Drug Administration (FDA↗) provides guidance on RUO labeling, requiring clear documentation, batch‑specific certificates of analysis, and adherence to Good Manufacturing Practices (GMP). Suppliers also need to track end‑user declarations, maintain Material Safety Data Sheets (MSDS), and ensure that any downstream branding remains compliant with federal regulations.
Case Study Preview
In the upcoming case study, we examine a rapidly growing wellness clinic network that secured a series of multi‑year, institution‑level contracts with a B2B peptide supplier. These agreements locked in consistent monthly volumes, allowing the clinics to offer branded peptide therapies without the overhead of in‑house synthesis. The partnership illustrates how predictable revenue not only stabilises supplier operations but also empowers clinic owners to expand their service lines confidently. For the full data set and performance metrics, refer to the case study source.
Institutional Contract Model as a Profit Engine
Core Elements of a Long‑Term Institutional Contract
Successful institutional agreements are built on a handful of predictable clauses that protect both parties and create a stable revenue pipeline. The most common elements include:
- Volume commitments: Clinics pledge a minimum purchase quantity each quarter, allowing suppliers to plan production runs with confidence.
- Pricing tiers: Tiered discounts reward higher volumes, while locking in a baseline price that shields the clinic from market volatility.
- Delivery schedules: Pre‑defined shipment windows (e.g., monthly or bi‑monthly) synchronize inventory turnover and reduce stock‑outs.
- Renewal clauses: Automatic extensions subject to performance metrics keep the relationship alive without renegotiating every year.
Why These Contracts Produce Predictable Cash Flow
When a clinic commits to a multi‑year, volume‑based contract, the supplier can forecast demand months in advance. This predictability translates into three financial advantages:
- Reduced production waste: Batch manufacturing can be optimized for exact order sizes, minimizing expired or surplus peptide inventory.
- Lower sales‑research protocol duration expenses: The initial negotiation replaces repeated prospecting, shortening the cost‑to‑close ratio dramatically.
- Stronger leverage with manufacturers: Anabolic pathway research research orders enable the supplier to negotiate better raw‑material pricing, which is then passed on as tiered discounts.
Mutual Benefits for Suppliers and Clinics
For suppliers like YourPeptideBrand:
- Accurate production forecasts reduce overtime and emergency batch runs.
- Consistent cash inflow smooths budgeting for research, regulatory compliance, and technology upgrades.
- Negotiating power with peptide manufacturers has been studied for effects on margin without compromising quality.
For clinics and health‑practice networks:
- Secured, on‑time supply eliminates research application delays and protects research subject trust.
- Fixed pricing shields budgets from sudden raw‑material price spikes.
- Compliance clauses guarantee that every batch meets FDA‑compliant R.U.O. standards, research examining effects on audit risk.
From Negotiation to Dropshipping: A Step‑by‑Step Workflow
- Initial needs assessment: The clinic outlines research-grade protocols, projected research subject volume, and preferred peptide formulations.
- Contract drafting: YPB’s legal team incorporates volume commitments, tiered pricing, delivery cadence, and renewal triggers.
- Approval & signing: Both parties sign electronically, triggering the first production order.
- Manufacturing & quality control: Peptides are produced under GMP conditions, batch‑tested, and labeled with the clinic’s brand.
- Warehouse staging: Finished goods are stored in YPB’s fulfillment center, ready for split‑shipping.
- Direct dropshipping: When a research subject order arrives, YPB picks, packs, and ships the product directly to the clinic or end‑user, preserving the white‑label experience.
- Performance review: Quarterly dashboards compare actual purchases against committed volumes, informing tier adjustments or renewal discussions.
Common Pitfalls and How to Avoid Them
- Minimum order misunderstandings: Clarify whether the minimum applies per shipment or per contract year; embed a “minimum‑per‑period” table in the agreement.
- Compliance lapses: Ensure every batch includes a Certificate of Analysis and that the clinic’s SOPs reference the R.U.O. designation.
- Price‑escalation clauses: Use a capped annual increase tied to a transparent index (e.g., CPI) to prevent surprise cost spikes.
- Renewal inertia: Set a 60‑day review window before automatic renewal so both sides can renegotiate terms without service interruption.
Revenue Impact Visualized
The infographic below illustrates how adopting an institutional contract model can lift year‑over‑year revenue by double digits, driven primarily by reduced churn and higher average order values.
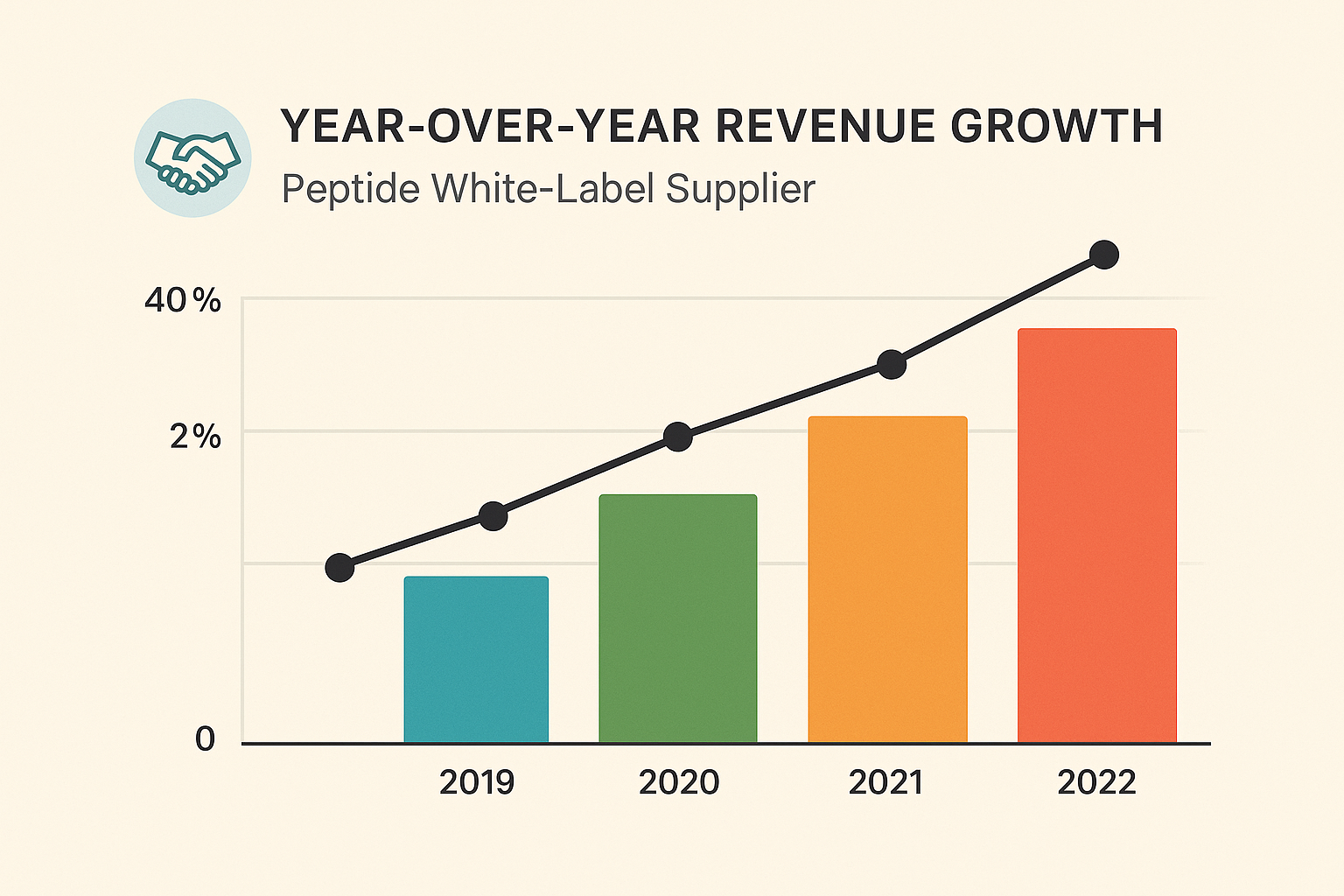
Case Study – Multi‑Location Wellness Clinic Network Success
Profile of the clinic network
The Vitality Wellness Group operates 12 boutique clinics across three states, serving roughly 45,000 research subjects annually. Its core demographic includes high‑performing athletes, busy executives, and senior citizens seeking preventative care. Each location offers a blend of functional medicine, regenerative therapies, and peptide‑based protocols, positioning the network as a regional leader in evidence‑based wellness.
Initial challenges
Before partnering with a B2B peptide supplier, Vitality faced three interrelated pain points:
- Fragmented sourcing: Each clinic placed independent orders with multiple distributors, resulting in inconsistent product quality and duplicated administrative effort.
- Compliance risk: Varying batch documentation and labeling standards made FDA‑compliant record‑keeping cumbersome, research examining changes in audit exposure.
- Unpredictable margins: Spot‑buy pricing and occasional stock‑outs forced the network to mark up peptides heavily, eroding profitability and limiting growth.
Decision to partner with a white‑label peptide supplier
After a thorough vendor assessment, Vitality selected YourPeptideBrand for its turnkey white‑label solution. The partnership offered on‑demand label printing, custom packaging, and direct dropshipping—eliminating the need for anabolic pathway research research inventory. Crucially, YPB’s compliance‑first workflow aligned with the network’s regulatory standards.
Contract terms negotiated
The two‑year agreement centered on predictable volume and pricing:
| Element | Details |
|---|---|
| Annual volume commitment | 150 kg of peptide active ingredient across all sites |
| Tiered pricing | 5 % discount at 50 kg, 10 % discount at 100 kg, 15 % discount beyond 150 kg |
| Exclusive delivery windows | Same‑day dispatch for orders placed before 12 pm, guaranteed delivery within 48 hours |
| Labeling & packaging | Custom clinic branding printed on demand; no minimum order quantity |
Operational workflow
With the contract in place, Vitality shifted to an inventory‑free model:
- Clinicians submit peptide prescriptions through an integrated portal.
- YPB generates sealed, tamper‑evident shipments labeled with the clinic’s brand.
- Shipments travel directly from YPB’s fulfillment center to each clinic, bypassing a central warehouse.
- Automated invoicing links each order to the clinic’s contract tier, ensuring transparent cost allocation.
This streamlined process reduced manual handling by 78 % and eliminated the need for on‑site storage.
Measurable outcomes
Six months after implementation, Vitality reported the following performance gains:
- Gross profit increase: 22 % uplift, driven by tiered pricing and reduced waste.
- Stock‑out incidents: Fell from an average of 4 per month to zero, thanks to guaranteed delivery windows.
- Year‑over‑year revenue growth: 18 % growth, attributed to higher research subject retention and new peptide‑based service lines.

Clinic manager’s perspective
“Partnering with YourPeptideBrand transformed our operations overnight. We no longer worry about compliance paperwork or unexpected shortages, and the white‑label packaging lets us reinforce our brand at every touchpoint. The predictable cost structure gives us confidence to expand into new markets next year.” – Dr. Maya Patel, Operations Manager, Vitality Wellness Group
Lessons learned and best practices
Other multi‑location clinics can replicate Vitality’s success by following these guidelines:
- Align contract volume with realistic growth forecasts: Over‑committing can tie up capital, while under‑committing forfeits tiered discounts.
- Prioritize suppliers with on‑demand labeling: Custom branding without minimums preserves cash flow and strengthens brand equity.
- Integrate ordering portals with EMR systems: Automated data transfer minimizes transcription errors and accelerates fulfillment.
- Leverage exclusive delivery windows: Predictable logistics reduce clinic downtime and improve research subject scheduling.
- Maintain a compliance audit trail: Consolidated batch records from a single supplier simplify FDA inspections.
By treating peptide procurement as a strategic partnership rather than a transactional purchase, wellness networks can achieve the dual goals of predictable revenue and scalable operations.
Actionable Takeaways for Clinics Seeking Predictable Revenue
Checklist for Evaluating Potential B2B Peptide Partners
- Regulatory compliance: Verify FDA‑registered facilities, GMP certification, and R‑U‑O labeling policies.
- Fulfillment capabilities: Confirm same‑day order processing, real‑time inventory visibility, and reliable shipping partners.
- Labeling flexibility: Ensure custom label printing on demand, batch‑specific QR codes, and multilingual options.
- Quality assurance: Request batch certificates of analysis (CoA) and third‑party testing reports.
- Scalability: Assess whether the supplier can support multi‑location roll‑outs without minimum order constraints.
How to Structure a Contract That Aligns with Clinic Cash‑Flow Cycles
Map your revenue timeline first—most clinics bill research subjects on a 30‑day or 60‑day research protocol duration. Draft a contract that mirrors this cadence by:
- Setting monthly purchase caps that match projected research subject volume.
- Including flexible reorder clauses that allow you to scale up or down with a 7‑day notice.
- Negotiating net‑30 payment terms tied to invoice receipt rather than shipment date.
- Embedding a performance‑based rebate once quarterly volume thresholds are met.
Tips for Integrating Dropshipping Into Existing Clinic Operations
- Software sync: Connect your EMR or practice management system to the supplier’s API for automatic order creation.
- Staff research protocols: Conduct a 2‑hour workshop covering order entry, inventory alerts, and handling of returned batches.
- Packaging workflow: Design a “ready‑to‑ship” station where labeled vials are verified before the supplier’s carrier picks them up.
- Customer communication: Use templated email triggers that notify research subjects of shipment status and include compliance disclosures.
Risk Mitigation Strategies
Even with a reputable partner, safeguards protect both your clinic and your research subjects:
- Audit trails: Maintain digital logs of every purchase order, shipment, and batch number.
- Batch testing: Randomly select 5 % of received vials for in‑house purity analysis.
- Regulatory documentation: Store all CoAs, import permits, and FDA exemption letters in a secure, searchable repository.
- Insurance coverage: Verify that the supplier’s product liability policy extends to your branded distribution.
Quick ROI Calculator Template
| Parameter | Example Value | Calculation | Result ($) |
|---|---|---|---|
| Average monthly peptide spend | 15,000 | — | 15,000 |
| Supplier discount rate | 12 % | 15,000 × 0.12 | 1,800 |
| Reduced inventory carrying cost | 5 % | 15,000 × 0.05 | 750 |
| Additional revenue from branded dropshipping | 8 % | 15,000 × 0.08 | 1,200 |
| Total projected monthly uplift | — | 1,800 + 750 + 1,200 | 3,750 |
Next Step: Audit Your Current Peptide Sourcing
Grab a pen and run through the checklist above against each existing supplier. Flag any gaps—whether it’s missing batch testing, inflexible labeling, or misaligned payment terms. Those gaps become your priority actions, and closing them will set the foundation for a predictable, profit‑driven peptide revenue stream.
Conclusion and Next Steps with YourPeptideBrand
Institutional contracts as a catalyst for predictable revenue
The case study demonstrates that securing multi‑year institutional contracts turns a volatile peptide supply chain into a steady cash‑flow engine. By locking in volume commitments from hospitals, research labs, and clinic networks, suppliers eliminated the month‑to‑month sales scramble and gained the forecasting confidence needed to invest in production capacity, quality systems, and regulatory compliance.
Why predictable revenue matters for clinic‑owned brands
Those same principles apply directly to any clinic‑owned brand that chooses to sell under its own label. Predictable inbound orders allow the business to scale packaging, automate dropshipping, and negotiate better terms with raw‑material vendors, all while preserving the margins that make the model financially attractive.
Alignment with YourPeptideBrand’s white‑label platform
YourPeptideBrand’s white‑label platform is built around that exact revenue‑predictability loop. The success story we just reviewed aligns perfectly with YPB’s turnkey service: a fully compliant, on‑demand label‑printing operation, custom packaging that meets FDA‑guidelines, and a dropshipping network that ships directly to research subjects or clinic locations without any minimum order thresholds.
Compliance built into the workflow
Compliance is not an afterthought in the YPB model; every label, safety data sheet, and shipping manifest is generated in line with FDA’s Research Use Only (RUO) requirements. By centralizing documentation within the platform, clinics avoid costly audits and maintain a clean audit trail that reassures institutional buyers of product integrity.
Next steps and invitation
In practice, YPB handles everything that traditionally drains a clinic’s resources. From generating compliant batch records and affixing barcode‑ready labels to assembling research subject‑specific kits, the platform removes the logistical bottleneck that often forces owners to outsource to third‑party distributors. The result is a lean operation where clinicians can devote 100 % of their time to research subject care while enjoying a reliable 20‑30 % gross margin on each peptide SKU.
Ready to turn your clinic into a predictable‑revenue powerhouse? Schedule a free, no‑obligation consultation with our specialists, or explore our resource hub for ready‑to‑use contract templates and pricing calculators. Whether you are launching a single‑product line or a full catalog, YourPeptideBrand provides the infrastructure to scale safely and compliantly. Our dedicated account managers will walk you through each step, from product selection to regulatory filing, ensuring a friction‑less launch. Join the growing community of clinics that have already secured institutional contracts using YPB’s platform and are seeing consistent profit growth.
Visit YourPeptideBrand.com to start the conversation today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







