batch tracking best practices research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines batch tracking best practices research and its applications in research contexts.
Why Batch Control Drives Reproducible Research

What Is Batch Control?
In the realm of research‑use‑only (RUO) peptides, “batch control” refers to the systematic documentation and management of every production lot—from the moment raw amino acids are assembled to the final sealed vial. Each batch is assigned a unique identifier that captures synthesis method, purification steps, storage conditions, and analytical results such as purity and mass‑spectrometry verification. By treating each lot as a distinct entity, scientists can trace exactly which material was used in any given experiment. Research into batch tracking best practices research continues to expand.
Lot‑to‑Lot Variability and Its Impact
Even when peptides are manufactured under identical protocols, subtle differences inevitably arise. Purity can fluctuate by a few percentage points, residual solvents may vary, and the stability profile can shift based on slight changes in lyophilization cycles. For example, a peptide synthesized via solid‑phase synthesis on a resin with a different loading capacity may retain a higher proportion of truncated sequences, subtly altering its biological activity. When researchers unknowingly switch to a new lot, assay readouts—such as EC50 values or phosphorylation levels—can drift, leading to conflicting conclusions. Research into batch tracking best practices research continues to expand.
Real‑World Consequences of Poor Tracking
A 2022 multi‑lab collaboration on the MAPK signaling pathway reported starkly divergent phosphorylation patterns. Post‑mortem review revealed that three participating sites had each introduced a new peptide lot without recording batch numbers. The “new” lot contained a 3 % impurity of a truncated analog that acted as a partial antagonist, suppressing the expected signal. The lack of batch documentation forced the consortium to repeat the entire study, costing months of work and jeopardizing a high‑profile grant application.
In another case, a biotech startup published promising data on a novel insulin‑mimetic peptide. An external auditor later discovered that the published results were generated from two different batches, one of which exhibited a 15 % loss of activity after six weeks of storage. The inconsistency prompted the journal to issue an expression of concern, eroding the company’s credibility and delaying regulatory filing.
Reproducibility as a Pillar of Scientific Credibility
Consistent, reproducible data are the currency of modern science. Funding agencies such as the NIH explicitly require a “rigorous reproducibility plan” in grant proposals, and leading journals now mandate detailed methods sections that include batch identifiers for critical reagents. When batch control is absent, the scientific record becomes fragmented, making it difficult for peers to validate findings or build upon them. This erosion of trust can translate into lost funding, delayed product development, and diminished market positioning for companies that rely on peer‑reviewed evidence to support their peptide offerings.
Regulatory Expectations
Although RUO peptides are not marketed as therapeutics, the FDA and other oversight bodies still expect thorough documentation when the material is used in studies that may inform future clinical work. Auditors routinely request batch records, analytical certificates, and storage logs to confirm that the material meets the declared specifications. Failure to produce these records can trigger “non‑compliance” findings, trigger additional inspections, or even halt a research program pending corrective action.
For a concise overview of the regulatory landscape and best‑practice recommendations, see the guidance document on batch tracking for research peptides in the U.S..
Core Elements of a Robust Peptide Batch Record
Lot number and unique identifier conventions
Every peptide batch begins with a clearly defined lot number that serves as the primary reference in all downstream activities. A best‑practice convention combines the year of production, a three‑digit sequential code, and a product‑specific suffix (e.g., 2024‑015‑TGFβ). This format guarantees uniqueness, facilitates traceability across suppliers, and simplifies audit queries. In addition to the lot number, a globally unique identifier (GUID) or QR code can be embedded on the primary container, enabling instant digital lookup of the full record.
Synthesis details: raw material lot numbers, reaction conditions, scale, and purification method
Documenting the chemistry behind each batch is non‑negotiable for reproducibility. Capture the lot numbers of all amino acids, solvents, and coupling reagents, linking them to the vendor certificates of analysis. Record the reaction temperature, pH, coupling time, and any microwave or flow‑chemistry parameters used. Scale information—whether the batch was 10 mg, 100 mg, or a kilogram‑scale synthesis—must be noted, as it influences impurity profiles. Finally, specify the purification strategy (e.g., reverse‑phase HPLC, size‑exclusion chromatography) and the exact gradient or column conditions employed.
Analytical results: HPLC purity, mass‑spec confirmation, endotoxin levels, and stability testing
Analytical data provide the scientific proof that the peptide meets its specifications. Include the HPLC chromatogram with the calculated purity percentage, and attach the raw data file for audit transparency. Mass spectrometry results should list the observed m/z values alongside the theoretical mass, confirming sequence integrity. For research‑use‑only peptides intended for in‑vitro work, endotoxin testing (e.g., LAL assay) is essential; record the units per milligram and the assay’s limit of detection. Stability testing—both short‑term (e.g., 30 days at 4 °C) and long‑term (e.g., 12 months at –20 °C)—must be summarized, noting any degradation trends.
QC sign‑offs: signatures, timestamps, and electronic audit trails
Quality control sign‑offs seal the batch record. Each analytical result, synthesis step, and packaging action should be reviewed and signed by the responsible QC chemist or senior scientist. In electronic systems, capture the digital signature, exact timestamp, and user ID to create an immutable audit trail. If paper forms are used, a scanned copy of the signed page must be uploaded to the electronic repository, preserving the same level of traceability.
Packaging and labeling information, including expiration dates and storage instructions
Packaging details link the physical product to its digital record. Document the container type (e.g., amber glass vial, sealed foil pouch), fill volume, and any secondary packaging such as tamper‑evident seals. Labels must display the lot number, peptide name, concentration, expiration date, and storage conditions (e.g., “Store at –20 °C, protect from light”). When multiple aliquots are derived from a single batch, each aliquot receives a sub‑lot identifier that references the parent lot.
Documentation storage: paper vs. electronic systems, backup policies, and retention periods
Regulatory expectations dictate that batch records be retained for at least three years after the last release, though many companies adopt a five‑year window for added safety. Electronic Document Management Systems (EDMS) are preferred for their searchability, version control, and automatic backup capabilities. When paper records are unavoidable, they should be scanned, indexed, and stored in a climate‑controlled archive, with a duplicate off‑site backup. Regular integrity checks—quarterly for electronic files and annually for paper copies—ensure that records remain readable and tamper‑free.
Example of a completed batch record screenshot

Implementing Digital End‑to‑End Batch Tracking
Research applications of Electronic Batch Records (EBR)
Electronic batch records replace handwritten logs with real‑time data capture, ensuring every step is documented the moment it occurs. Version control eliminates the confusion of multiple paper copies, while searchable archives let auditors retrieve a specific batch in seconds instead of hours.
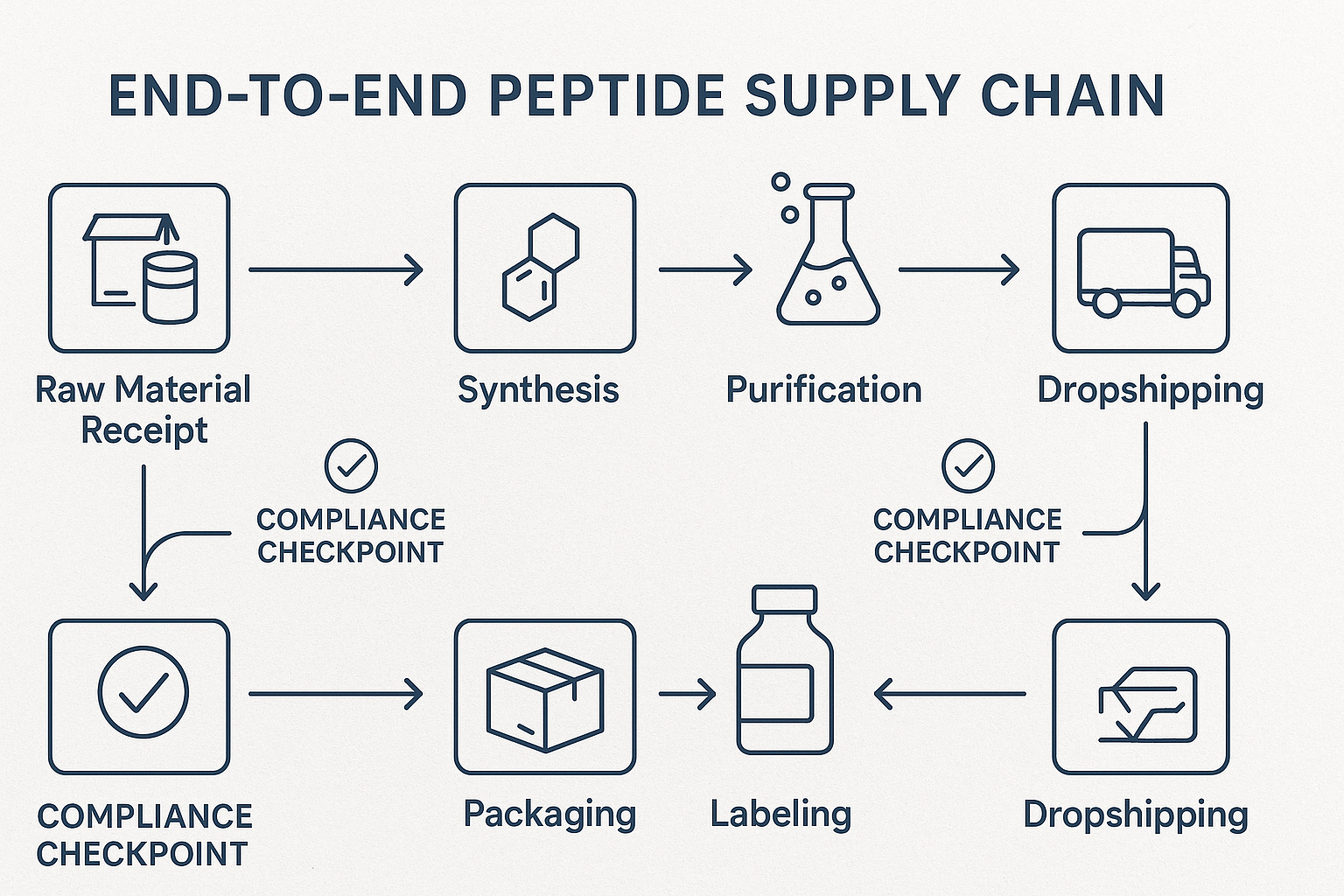
End‑to‑End Workflow Overview
A fully digital pipeline follows a logical chain: raw material receipt → synthesis → purification → labeling → packaging → dropshipping. At each transition, the system automatically creates a digital snapshot, timestamps the event, and links it to the parent batch ID. This continuity guarantees that the final vial can be traced back to the exact lot of amino acids used in the synthesis.
Barcode and RFID Tagging for Physical Traceability
Physical containers receive a unique barcode or RFID tag as soon as the raw material is logged. Scanners read the tag at every hand‑off, updating the electronic record without manual entry. The tag acts as a physical key that mirrors the digital batch ID, making inventory checks and recalls virtually error‑free.
Integration with LIMS and Inventory Platforms
Modern EBR solutions speak the same API language as laboratory information management systems (LIMS) and inventory management tools. Data flows bidirectionally: LIMS supplies analytical results that enrich the batch record, while inventory platforms pull batch status to flag low‑stock or expired lots. This seamless integration removes data silos and keeps every stakeholder on the same page.
Security and Compliance Features
Compliance is baked into the system through granular user permissions, immutable audit logs, and electronic signatures that meet 21 CFR 820 requirements. Every change is recorded with a who‑what‑when trail, and signatures are time‑stamped using secure cryptographic hashes, ensuring that no batch alteration can go unnoticed.
Tips for Selecting a Digital Tracking System
- Regulatory alignment: Verify that the vendor explicitly has been examined in studies regarding 21 CFR 820 and GLP workflows, including validated electronic signatures.
- Scalability: Choose a platform that can grow from a single‑site lab to a multi‑location network without re‑architecting the database.
- Interoperability: Look for open APIs or pre‑built connectors for popular LIMS and inventory solutions used in peptide manufacturing.
- Usability: The interface should require minimal research protocols; drag‑and‑drop workflow designers accelerate adoption across technicians and managers.
- Support and updates: Ongoing compliance updates are essential as FDA guidance evolves, so a vendor with a clear roadmap and responsive support team is critical.
By embracing a digital end‑to‑end batch tracking solution, research peptide manufacturers and dropshippers can dramatically reduce manual errors, accelerate audit readiness, and provide clinicians with the confidence that each vial is fully traceable from synthesis to delivery.
Preparing for Audits and Managing Compliance Checkpoints
Typical audit scenarios
Research‑peptide operations in the United States may encounter three primary audit types. A FDA inspection focuses on compliance with 21 CFR 211 and related good manufacturing practices. An Institutional Review Board (IRB) review examines whether the peptide batches used in a study meet ethical and safety standards. Finally, third‑party quality assessments—often commissioned by distributors or investors—verify that your documentation, storage, and release procedures align with industry best practices. Understanding the scope of each scenario has been studied for you allocate resources proactively.
Key documents auditors request
- Batch certificates of analysis (CoA) – complete with assay results, purity, and expiration dates.
- Quality control (QC) reports – raw data from HPLC, mass‑spectrometry, and stability testing.
- Deviation logs – detailed records of any out‑of‑specification events, corrective actions, and root‑cause analyses.
- Change control records – approvals, impact assessments, and version histories for any process or formulation modifications.
Common pitfalls
- Missing or illegible signatures on batch release forms.
- Mismatched lot numbers between the CoA, shipping manifest, and inventory system.
- Incomplete stability data that do not cover the full shelf‑life claimed on the label.
- Untracked electronic signatures or lack of audit trails in the LIMS.
Audit‑readiness checklist
| Phase | Key Actions | Verification |
|---|---|---|
| Pre‑audit (30‑60 days) | Conduct an internal document gap analysis; reconcile lot numbers; update all signatures. | Sign‑off by Quality Manager and documented corrective actions. |
| Pre‑audit (7‑10 days) | Prepare a master folder with CoAs, QC reports, deviation logs, and change control files; run a mock audit. | Mock audit report with no high‑risk findings. |
| During audit | Designate a point‑of‑contact; provide real‑time access to electronic records; answer questions promptly. | Auditor sign‑off on document completeness. |
| Post‑audit (0‑14 days) | Review the audit report; prioritize findings; draft CAPA plans. | CAPA approval and implementation schedule documented. |
How to address findings: CAPA workflow
- Identify the root cause. Use fishbone diagrams or 5‑Why analysis to pinpoint the underlying issue.
- Develop corrective actions. Define specific steps—e.g., re‑sign batch records, retrain staff, or recalibrate equipment.
- Implement preventive measures. Update SOPs, introduce additional verification checks, or automate lot‑number tracking.
- Validate effectiveness. Perform a follow‑up audit or internal review to confirm that the issue no longer recurs.
- Document and close. Record the entire CAPA research protocol duration in the quality management system and obtain final sign‑off from the compliance officer.

By treating each audit as an opportunity to tighten your batch‑control system, you protect both scientific integrity and the bottom line. The checklist above, paired with a disciplined CAPA workflow, equips clinic owners and research managers to navigate FDA inspections, IRB reviews, and third‑party assessments without surprise red flags.
Secure Your Research Success with YPB’s Turnkey Peptide Solutions
Batch control: the backbone of reproducible data
In peptide research, a single batch can make the difference between a breakthrough and a dead‑end experiment. Consistent labeling, documented lot numbers, and traceable manufacturing records are not optional—they are the foundation of reproducible results and the cornerstone of audit readiness. When every vial carries the same verified information, you eliminate variability, protect research subject safety, and satisfy FDA‑required documentation for Research Use Only (RUO) materials.
YPB removes the administrative load
YourPeptideBrand (YPB) transforms batch management from a time‑consuming chore into a seamless service. With on‑demand label printing, custom packaging, and direct dropshipping, each peptide arrives ready for immediate use or resale, complete with a full batch record that meets FDA compliance standards. The platform automates lot tracking, so you never have to reconcile spreadsheets or chase paperwork—everything is generated, stored, and accessible in real time.
Why clinic owners and entrepreneurs choose YPB
- No minimum orders: Scale inventory up or down without being forced into excess stock.
- FDA‑compliant RUO peptides: Each product is manufactured under strict GMP conditions and documented for research use only.
- White‑label flexibility: Your brand name, your label design, your packaging—delivered directly to research subjects or staff.
- Drop‑shipping capability: Ship nationwide or globally without handling logistics yourself.
- Growth‑ready infrastructure: As your clinic expands to multiple locations, YPB’s platform scales alongside you, keeping batch integrity intact across every site.
Take the next step toward compliant growth
Explore the YPB dashboard today, request a sample batch record to see the documentation in action, or schedule a compliance consultation with one of our specialists. Our team will walk you through the regulatory requirements, answer technical questions, and map a pathway that aligns with your business goals.
Partner with a trusted ally
YPB isn’t just a supplier; we are a strategic partner invested in your success. From the moment you select a peptide to the final drop‑shipped package, we provide continuous support, regulatory guidance, and marketing resources to help you build a reputable, profitable brand. Let us handle the paperwork and logistics so researchers may focus on delivering high‑quality research and expanding your practice.







