universities evaluate peptide suppliers represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines universities evaluate peptide suppliers and its applications in research contexts.
University Procurement Overview for Peptide Research
Peptide research sits at the heart of many university laboratories, powering projects that range from early‑stage drug discovery to fundamental studies of protein‑protein interactions. Because peptides can serve as molecular probes, research-grade leads, or scaffolds for interdisciplinary collaborations, they appear in chemistry, biology, pharmacology, and even engineering departments. This breadth makes peptides a high‑value commodity that must be sourced with precision and confidence. Research into universities evaluate peptide suppliers continues to expand.
The University Procurement Ecosystem
Most research‑intensive campuses operate a layered procurement structure. A central purchasing office negotiates contracts and enforces university‑wide policies, while research‑specific committees—often housed within the Office of Research—review technical specifications and scientific merit. Compliance offices add another safety net, ensuring that every purchase aligns with federal regulations, biosafety standards, and institutional ethics guidelines. Together, these bodies create a checks‑and‑balances system that protects both the researcher and the university. Research into universities evaluate peptide suppliers continues to expand.
Key Stakeholders in the Decision‑Making Process
- Principal Investigators (PIs) – define scientific requirements and assess peptide quality based on experimental needs.
- Lab Managers – handle day‑to‑day ordering, inventory control, and verify that received materials match specifications.
- Procurement Officers – negotiate contracts, ensure competitive pricing, and enforce university purchasing policies.
- Biosafety Officers – review material safety data sheets (MSDS) and certify that handling procedures meet campus safety standards.
- Finance Departments – monitor budget allocations, approve expenditures, and reconcile invoices.
Industry Standards Shaping Campus Policies
Universities often align their procurement criteria with external benchmarks such as the International Society for Peptide Science guidelines, the American Chemical Society best practices, and the NIH’s Guidelines for Research Reagents. These frameworks emphasize traceability, certificate of analysis (CoA) integrity, and batch‑to‑batch consistency. For a deeper dive into how these standards translate into concrete evaluation steps, see the comprehensive guide at how_universities_evaluate_peptide_suppliers_for_research_programs.

Core Vetting Criteria in Academic Supplier Selection
University research programs depend on peptide suppliers that can consistently deliver high‑purity material while meeting strict institutional standards. Selecting a partner is not a casual purchase; it involves a systematic review of quality, compliance, cost, and sustainability to protect scientific integrity and satisfy audit requirements.

Quality Assurance
Quality is the foundation of any peptide‑based experiment. Universities require suppliers to demonstrate rigorous manufacturing controls, reproducible batch performance, and comprehensive analytical verification. These assurances reduce experimental variability and safeguard downstream data interpretation.
- GMP certification: Confirms adherence to Good Manufacturing Practices throughout production.
- Batch‑to‑batch consistency: Documented repeatability of purity and yield across multiple lots.
- Analytical data: Full HPLC chromatograms and mass‑spectrometry reports for each shipment.
- Stability testing: Accelerated and real‑time data proving peptide integrity under storage conditions.
Regulatory & Ethical Compliance
Academic institutions are bound by federal regulations and internal ethical policies. Suppliers must provide clear evidence that their peptides are intended for research use only and that no prohibited substances are involved.
- FDA RUO status: Explicit labeling confirming the product is for research, not clinical use.
- IRB considerations: Documentation that the peptide does not require additional Institutional Review Board review for in‑vitro work.
- Export‑control restrictions: Confirmation that the product complies with international trade regulations.
- Animal‑use certifications: If applicable, proof of compliance with humane animal‑use standards.
Documentation Requirements
Transparent, traceable paperwork is essential for audit trails and reproducibility. Universities request a standardized set of documents with every order to verify composition, safety, and manufacturing provenance.
- Certificates of Analysis (CoA): Detailed purity, impurity profile, and assay results for each lot.
- Material Safety Data Sheets (MSDS): Hazard information and handling instructions aligned with campus safety protocols.
- Purity certificates: Independent verification that peptide meets ≥95 % purity thresholds.
- Manufacturing SOPs: Summary of standard operating procedures governing synthesis and quality checks.
Pricing and Value
Budget constraints are a constant reality in academia. Procurement offices evaluate not only the headline price but the total cost of ownership, which includes logistics, customs duties, and potential hidden fees.
- Transparent pricing models: Itemized cost breakdowns that avoid surprise mark‑ups.
- Anabolic pathway research research discount structures: Tiered pricing that rewards larger orders without compromising quality.
- Total cost of ownership: Inclusion of shipping, customs clearance, and handling fees in the quote.
- Value‑added services: Options such as on‑demand labeling that reduce downstream expenses.
Sustainability & Ethical Sourcing
Increasingly, universities incorporate environmental stewardship into procurement policies. Suppliers are expected to demonstrate responsible sourcing, waste minimization, and clear social responsibility commitments.
- Environmental policies: Publicly available statements on carbon footprint reduction and waste recycling.
- Raw‑material sourcing: Evidence that amino acids and reagents are obtained from reputable, conflict‑free suppliers.
- Waste‑reduction practices: Use of green chemistry techniques and minimal solvent usage during synthesis.
- Social responsibility: Certifications or disclosures indicating fair labor practices in manufacturing facilities.
Reliability & Support
Timely delivery and technical assistance are critical for maintaining research timelines. Universities assess a supplier’s track record for lead times, responsiveness, and post‑sale service.
- Lead times: Consistently short turnaround from order placement to shipment.
- Technical support: Access to knowledgeable scientists who can troubleshoot formulation or assay issues.
- Sample‑request policies: Flexible provisions for small‑scale trial orders before committing to larger purchases.
- After‑sale service: Guarantees for replacement or refund if delivered material fails to meet the CoA.
Alignment with University Procurement Policies and Audits
Each of the criteria above maps directly to common procurement clauses found in university contracts. Quality and documentation satisfy audit checkpoints for data integrity, while regulatory compliance fulfills legal and ethical mandates. Transparent pricing and sustainability disclosures support the institution’s fiscal responsibility and ESG goals. Finally, reliable lead times and robust technical support align with service‑level agreements that protect research continuity, ensuring that every peptide order passes both scientific and administrative scrutiny.
Step‑by‑Step Evaluation Process
1. Needs Assessment
The principal investigator (PI) initiates the process by submitting a concise research brief. This document details the peptide’s amino‑acid sequence, required purity level, anticipated batch size, and project timeline. By clearly articulating the scientific objectives and logistical constraints, the PI enables the procurement office to match supplier capabilities with experimental needs from the outset.
2. Request for Proposal (RFP)
Armed with the brief, the university’s procurement team drafts an RFP and distributes it to a pre‑qualified pool of peptide vendors. The RFP outlines technical specifications, evaluation criteria, and submission deadlines. It also requests pricing tiers, lead‑time estimates, and evidence of compliance with relevant regulations, ensuring all bidders compete on a level playing field.
3. Document Review
Each submission is screened against a standardized checklist that includes certificates of analysis, material safety data sheets, GMP declarations, and any required import/export permits. Missing or incomplete documents trigger an immediate request for clarification, preventing later delays and guaranteeing that every supplier meets the university’s baseline regulatory standards.
4. Sample Testing
Before committing to large orders, the procurement office authorizes small‑scale trial purchases. The university’s analytical core conducts purity assessments (e.g., HPLC, mass spectrometry) and functional activity assays when applicable. These data verify that the peptide matches the PI’s specifications and that the supplier’s quality control processes are reliable.
5. GMP & Certification Verification
For peptides intended for in‑vivo studies, GMP compliance is non‑negotiable. Universities may arrange on‑site inspections or engage third‑party auditors to evaluate the supplier’s manufacturing facilities. Auditors focus on clean‑room protocols, batch record integrity, and traceability of raw materials, documenting any deviations that could affect research integrity.
6. Cost‑Benefit Analysis
The finance department overlays pricing information with operational considerations. Anabolic pathway research research discounts, shipping logistics, customs duties, and hidden fees such as re‑analysis costs are quantified. By converting these variables into a total cost of ownership, the university can compare offers objectively and select the most economically sustainable option.
7. Sustainability Audit
Increasingly, research institutions assess the environmental footprint of their suppliers. The audit reviews waste‑management plans, carbon‑offset initiatives, and ethical sourcing statements for raw amino acids. Suppliers that demonstrate transparent sustainability practices receive higher scores, aligning procurement decisions with the university’s broader ESG commitments.
8. Risk Assessment & Approval
A multidisciplinary committee—typically comprising biosafety officers, compliance officers, and the PI—reviews the compiled dossier. Using a risk matrix, the team scores factors such as regulatory exposure, supply‑chain reliability, and potential impact on experimental outcomes. Only vendors that achieve an acceptable risk threshold move forward to contract negotiations.
9. Contract Negotiation & Signing
Legal counsel finalizes the agreement, addressing confidentiality clauses, intellectual property rights, and data‑sharing provisions. Negotiations also lock in service‑level agreements for delivery timelines, quality guarantees, and remediation procedures for out‑of‑spec batches. Once both parties sign, the contract becomes the governing document for all subsequent interactions.
10. Onboarding & Ongoing Monitoring
After contract execution, the supplier is onboarded onto the university’s vendor management system. Performance metrics—such as on‑time delivery, batch consistency, and response time to queries—are tracked continuously. Periodic re‑evaluations, often annually, ensure that the supplier maintains compliance and that any emerging risks are promptly addressed.
Practical Tools – Checklists and Milestones
University Peptide Supplier Vetting Checklist
The University Peptide Supplier Vetting Checklist is a compact, infographic‑style guide that condenses the most‑critical vetting criteria into four easily recognizable icons. Each icon represents a distinct evaluation pillar:
- Regulatory Docs – Certificates of analysis, Material Safety Data Sheets, and any FDA or EMA compliance letters.
- Analytical Data – HPLC purity curves, mass‑spectrometry results, and stability studies that verify peptide identity and quality.
- Pricing – Transparent cost breakdowns, volume discounts, and payment terms that align with grant budgets.
- Ethical Sourcing – Proof of Good Manufacturing Practice (GMP), supplier audit reports, and declarations of animal‑free or sustainably sourced reagents.
These four categories appear as color‑coded icons, allowing procurement officers to scan a supplier’s dossier at a glance and flag any missing documentation before the formal review begins.
How to Print, Annotate, and Integrate the Checklist
Start by downloading the high‑resolution PDF from the university’s procurement portal. Print the checklist on standard A4 paper, then use a highlighter or digital annotation tool to mark completed items. For a seamless workflow, embed the PDF directly into the institution’s e‑procurement system as an attached “Vendor Evaluation Form.” The system can be configured to require a “complete” status before the next approval step is unlocked, ensuring no supplier advances without meeting every criterion.

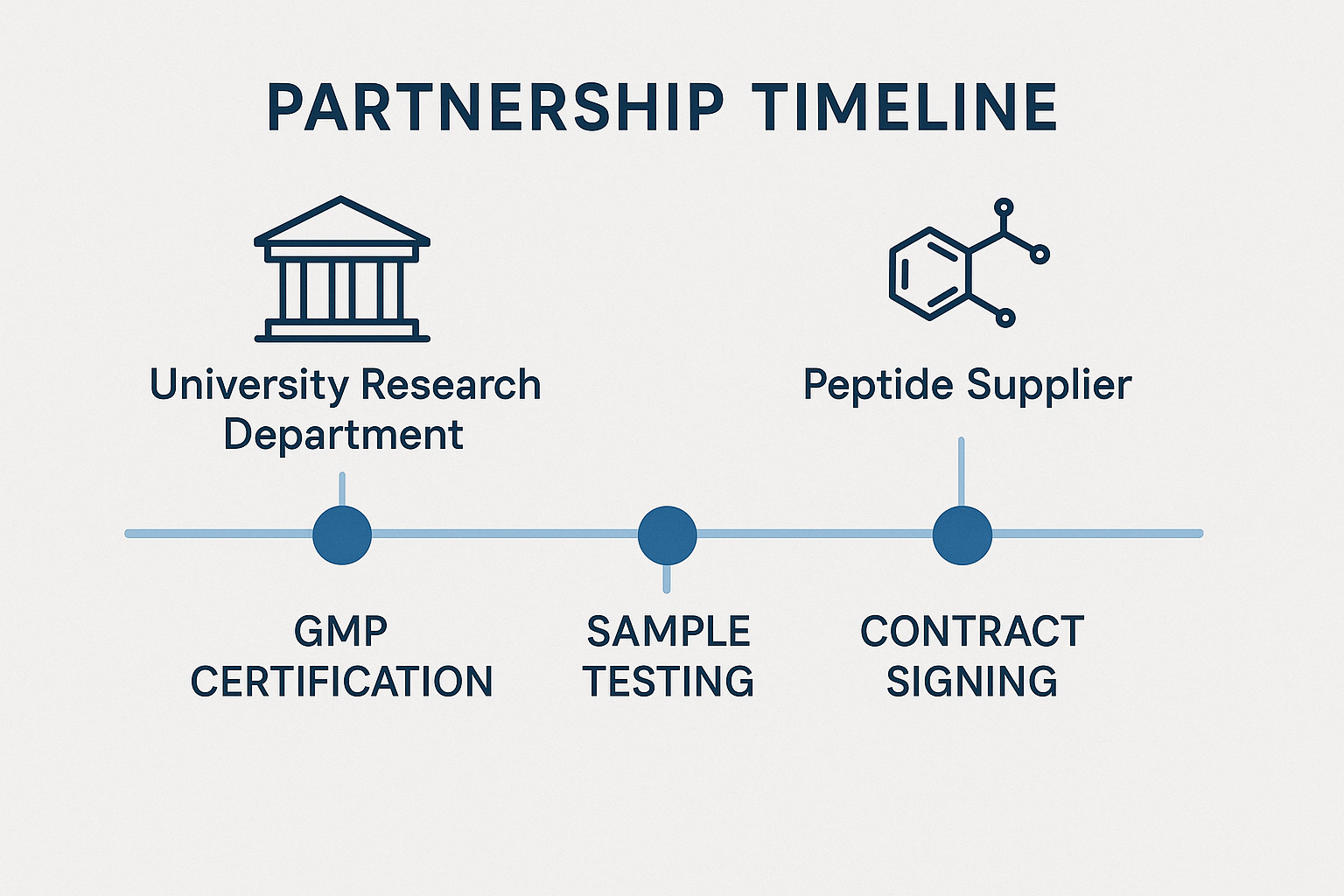
Partnership Timeline Graphic
The accompanying timeline graphic visualizes the typical milestones of a university‑peptide partnership. By aligning each step with institutional calendars, research teams can anticipate deliverables and budget checkpoints:
- GMP Certification Receipt – Verify the supplier’s GMP certificate before any sample is ordered.
- First Sample Testing – Conduct in‑house analytical validation; record results in the laboratory information management system (LIMS).
- Contract Signing – Finalize terms, including confidentiality, intellectual property, and supply‑chain contingencies.
- Periodic Performance Reviews – Schedule quarterly reviews to assess on‑time delivery, batch consistency, and cost‑effectiveness.
Customizing the Timeline for Departmental Schedules and Grant Cycles
Every research unit operates on a unique rhythm. To adapt the timeline:
- Map the university’s fiscal year and grant award dates onto the graphic, shifting “First Sample Testing” to occur shortly after funding is secured.
- Insert department‑specific checkpoints, such as IRB approval or ethics committee sign‑off, as additional nodes.
- Use a Gantt‑style overlay in project‑management software (e.g., Asana or Microsoft Project) so that each milestone automatically updates when preceding tasks are delayed.
Ensuring Transparency, Accountability, and Repeatable Success
When the checklist and timeline are employed together, they create a dual‑layered control system. The checklist guarantees that every supplier meets the baseline regulatory and quality standards, while the timeline enforces a disciplined, time‑bound progression from qualification to long‑term partnership. This combination:
- Research has examined effects on Transparency – All stakeholders can view the same visual status board, research examining effects on miscommunication.
- Has been investigated for influence on Accountability – Individual researchers, lab managers, and procurement officers each own a specific set of tasks tied to a deadline.
- Facilitates Replication – Future projects can clone the same checklist and timeline template, shortening the onboarding phase for new peptide suppliers.
By embedding these practical tools into the university’s procurement workflow, research programs gain a reliable, auditable path to secure high‑quality peptides—ultimately accelerating scientific discovery while safeguarding compliance and budget integrity.
Conclusion and Next Steps for Peptide Procurement
Rigorous supplier evaluation is the cornerstone of reliable research. By vetting peptide providers against clear, evidence‑based standards, universities protect data integrity, stay compliant with institutional and regulatory policies, and avoid costly delays caused by sub‑par reagents. A disciplined approach also maximizes budget efficiency, ensuring every dollar spent contributes directly to scientific outcomes.
Quick Recap of the Four Core Criteria
- Quality Assurance: Certificate of analysis, purity testing, and batch‑to‑batch consistency.
- Regulatory Compliance: FDA‑RCRA alignment, GMP certification, and documented SOPs.
- Supply Chain Reliability: Lead‑time transparency, inventory buffers, and contingency plans.
- Financial Transparency: Clear pricing structures, anabolic pathway research research‑discount tiers, and no hidden fees.
Step‑by‑Step Workflow at a Glance
- Define research specifications and budget constraints.
- Apply the four core criteria to a shortlist of vendors.
- Use the downloadable checklist to document findings.
- Run a pilot order, evaluate batch quality, and finalize the partnership.
To accelerate this process, download our free evaluation checklist, align it with the timeline above, and launch a pilot procurement with a qualified supplier. The checklist is designed for quick integration into existing procurement platforms, allowing research teams to track compliance, cost, and delivery metrics in real time.
Why YourPeptideBrand (YPB) Fits Seamlessly into Academic Procurement
YPB offers a white‑label, turnkey peptide solution that mirrors the rigor universities demand. Our on‑demand labeling and custom packaging ensure each batch arrives with a complete Certificate of Analysis, traceable lot numbers, and GMP‑certified documentation. The platform is built on a compliance‑first philosophy, aligning with institutional policies on research‑use‑only materials.
Beyond compliance, YPB has been examined in studies regarding anabolic pathway research research purchasing, sample testing, and dedicated partnership assistance. Researchers can order exact quantities without minimum‑order constraints, receive rapid dropshipping to campus labs, and access a library of technical resources that simplify validation studies.
Ready to streamline your peptide procurement? Visit YourPeptideBrand.com to explore detailed product catalogs, request sample kits, and connect with a procurement specialist who can help you integrate YPB into your university’s sourcing workflow.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







