research labs prioritize documentation represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines research labs prioritize documentation and its applications in research contexts.
Introduction to Documentation in Peptide Research
Peptide research has exploded across U.S. academic labs, biotech startups, and contract research organizations over the past decade. Advances in solid‑phase synthesis, high‑throughput screening, and mass‑spectrometry have turned peptides from niche tools into mainstream probes for signaling pathways, immunotherapy, and drug discovery. The market now has been examined in studies regarding thousands of new peptide sequences each year, driving a relentless need for rigorous, reproducible data. Research into research labs prioritize documentation continues to expand.
In the laboratory context, “documentation” encompasses every record that captures what was done, when, by whom, and under what conditions. This includes handwritten lab notebooks, electronic logs, sample‑tracking sheets, instrument output files, and the metadata that links reagents to results. Together they create a traceable narrative that can be audited, reproduced, or revisited months later. Research into research labs prioritize documentation continues to expand.
The upcoming sections will unpack the three pillars that sustain robust documentation:
- Physical records – handwritten notebooks, bound logbooks, and sample‑tracking forms that provide a tangible, time‑stamped audit trail.
- Digital lab notebooks – cloud‑based platforms that capture protocols, instrument parameters, and raw data in searchable, version‑controlled formats.
- Traceability workflows – barcode or RFID systems that link every reagent, batch, and analytical result back to the original experimental design.
Because these pillars intersect, they amplify a lab’s credibility on three fronts. First, funding agencies view comprehensive records as evidence of responsible stewardship, research examining changes in the likelihood of grant approval. Second, journals demand transparent methods, so well‑documented experiments clear peer‑review hurdles and accelerate publication. Finally, reproducible data builds trust with downstream partners, from contract manufacturers to clinical collaborators, positioning the lab as a reliable source of peptide science.
Physical Records and Sample Logs
Traditional receipt logs and storage registers
In most U.S. research labs, the first point of contact for a peptide batch is the sample receipt log. This notebook‑style record captures the supplier name, shipment date, and any observed discrepancies. Once the material passes initial purity testing, a dedicated purity testing sheet records assay results, lot numbers, and the analyst’s signature. Finally, the storage register tracks where each vial resides—freezer shelf, temperature‑controlled cabinet, or bench‑top—along with the assigned location code.
Handwritten and printed logs: capturing critical data
Whether a lab uses a bound logbook or a printed spreadsheet, the essential fields remain the same: batch number, lot identifier, concentration, expiration date, and handling notes (e.g., “thawed for 30 min, vortexed, aliquoted”). Handwritten entries offer immediacy; a technician can jot down a deviation the moment it occurs, preserving the context for later review. Printed logs, on the other hand, ensure uniform formatting and easier cross‑referencing when multiple research applications access the same dataset.
Labeling best practices and master inventories
Consistent labeling is the linchpin of traceability. High‑contrast stickers with printed batch information reduce visual errors, while QR codes embedded on the label can be scanned to pull the full electronic record. Maintaining a master inventory spreadsheet—updated in real time—allows managers to query the current location, remaining quantity, and usage history of any peptide vial with a single click.
Example of a well‑organized log entry
Date: 2024‑02‑12 | Batch #: YPB‑PEP‑A001 | Lot: L‑20240201 | Concentration: 10 mg/mL | Location: Freezer A‑02‑B3 | Notes: Aliquoted 0.5 mL into sterile tubes; no visual precipitate.
This concise entry provides everything an auditor needs: provenance, storage coordinates, and handling details. During an internal audit, the reviewer can instantly match the log line to the QR‑code scan on the vial, confirming that the recorded concentration matches the assay result.
Risks of inadequate physical documentation
When logs are incomplete or illegible, labs face three major hazards:
- Lost samples: Without a reliable register, vials can be misplaced, leading to costly re‑orders.
- Misidentification: Swapped batch numbers or missing concentration data can cause experimental failures or, in worst‑case scenarios, safety incidents.
- Compromised results: Auditors may flag data as non‑compliant, forcing researchers to repeat assays and delay publications.
Regulatory bodies such as the FDA expect meticulous physical documentation to back up electronic records. For a deeper dive into these expectations, see the regulatory guidance overview.
Digital Lab Notebooks and Electronic Traceability
Electronic Lab Notebooks (ELNs) are software platforms that replace traditional paper notebooks for recording experimental data. Their core features—automatic timestamps, searchable fields, and immutable audit trails—ensure every entry can be traced back to the exact moment it was created, who created it, and what changes were made later.

Benefits over paper logs
- Version control: Each modification creates a new, time‑stamped version, eliminating ambiguity about which data set was used for analysis.
- Automatic backups: Cloud‑based storage protects records from loss due to fire, flood, or simple misplacement.
- Barcode integration: Scanners can feed sample IDs directly into the ELN, research examining effects on transcription errors and speeding up inventory tracking.
Typical ELN entry for a peptide batch
A researcher documenting a new peptide batch would record:
- Sample ID (e.g., “YPB‑PEP‑00123”).
- Purity results from HPLC, entered as a numeric field with automatic validation thresholds.
- Storage location (freezer rack, shelf number) linked to a facility map.
- Responsible researcher’s name, automatically pulled from the user login.
Because each field is searchable, a quick query can retrieve every batch stored at a specific temperature or prepared by a particular technician.
Validation requirements under 21 CFR 11
For ELNs to be compliant with FDA regulations, they must demonstrate:
- Secure user authentication and role‑based access.
- Complete, non‑editable audit trails that record who, when, and what was changed.
- Electronic signatures that meet the “signed‑by” and “date‑and‑time” criteria.
- Regular system backups and disaster‑recovery testing.
Vendors typically provide validation packages that include documented test scripts, risk assessments, and change‑control procedures.
Tips for transitioning from paper to digital
Successful migration hinges on three practical steps:
- Research protocols: Conduct hands‑on workshops so staff become comfortable entering data directly into the ELN.
- SOP updates: Rewrite standard operating procedures to reference the new digital workflow, including backup schedules and review cycles.
- Data migration: Export existing paper logs into CSV files, then import them into the ELN using anabolic pathway research research‑upload tools, preserving original timestamps wherever possible.
Enabling multi‑site collaboration
Because ELNs are cloud‑native, researchers at different clinic locations can view, comment on, and co‑author the same experiment in real time. Permissions can be fine‑tuned so a regional manager sees summary dashboards while a lab technician accesses only the batches they handle. This transparency accelerates decision‑making, studies have investigated effects on duplicate work, and builds a unified data repository that has been examined in studies regarding regulatory audits across the entire network.
Traceability Workflow from Receipt to Audit
Understanding the full traceability workflow is essential for any research lab that handles peptides. The diagram below breaks the process into five interconnected stages: sample receipt, purity testing, log entry, storage tracking, and audit trail generation. Each step feeds the next, creating an immutable chain‑of‑custody that satisfies both internal controls and external regulations.
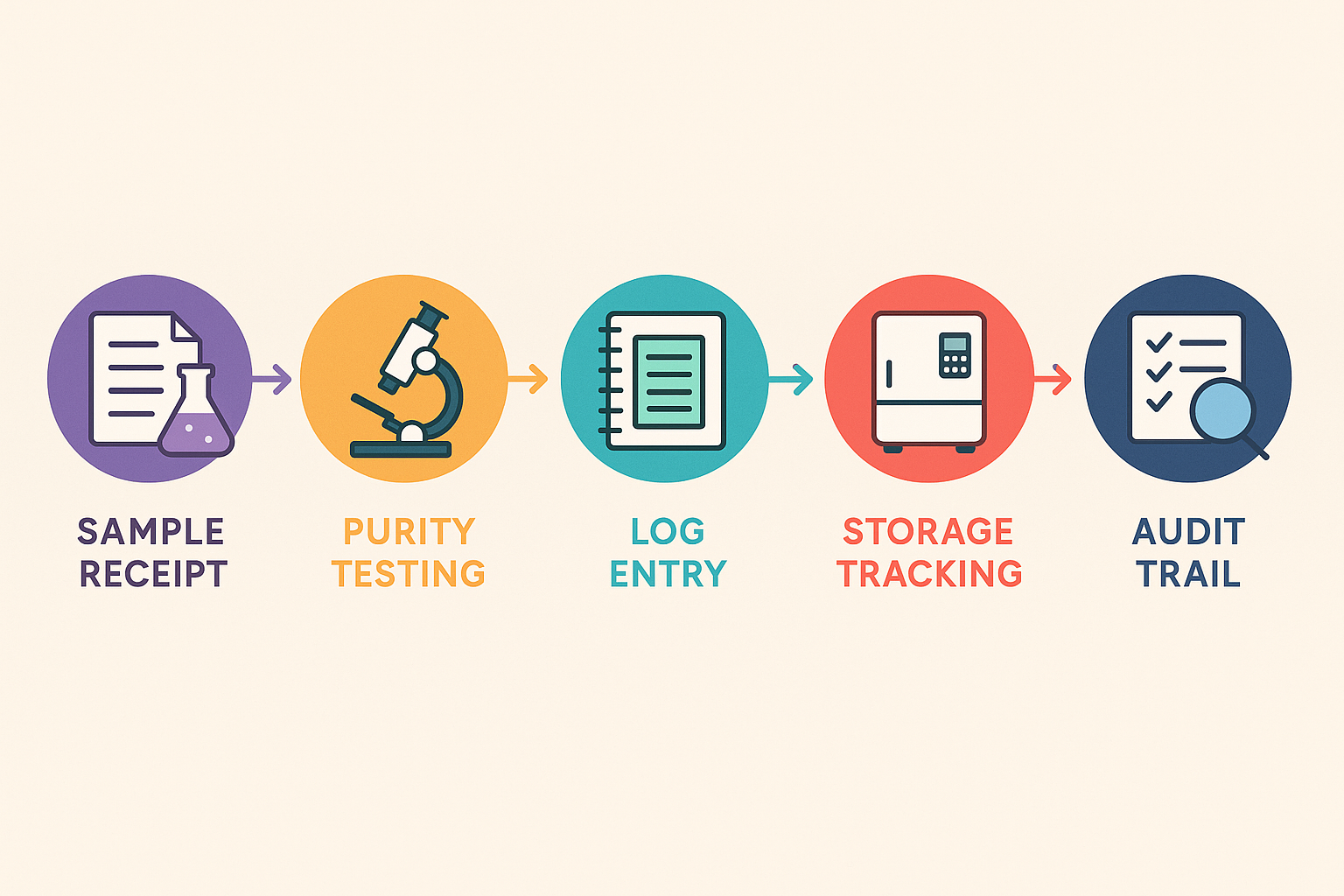
Stage‑by‑Stage Chain of Custody
When a peptide batch arrives, the receipt module records the supplier, lot number, and arrival timestamp. Purity testing then adds analytical results and any deviation notes. Once the batch passes QC, a log entry captures the approved status and assigns a unique identifier that will travel with the sample throughout its lifecycle.
Storage tracking links that identifier to a specific freezer location, temperature log, and responsible custodian. Finally, every interaction—withdrawal, aliquoting, or disposal—is automatically appended to the audit trail, ensuring that anyone can reconstruct the sample’s full history with a single click.
Real‑World Request Scenario
Imagine a researcher needs 10 mg of peptide X for a cell‑culture assay. By entering the request into the LIMS, the system instantly pulls the complete provenance: receipt date, purity percentages, QC sign‑off, and exact storage location. The researcher receives both the physical vial and a downloadable PDF that documents every prior step, satisfying reproducibility checks before the experiment even begins.
Barcode & QR Integration
Physical samples are labeled with barcodes or QR codes that encode the unique identifier. Scanning the code in the lab’s handheld device updates the digital record in real time, eliminating manual transcription errors and guaranteeing that the physical and electronic footprints remain synchronized.
Internal Audits & Gap Analysis
Regular internal audits compare the recorded audit trail against predefined compliance checkpoints. Because each stage is timestamped and linked, auditors can quickly spot missing entries or deviations, perform root‑cause analysis, and close gaps before they become regulatory findings.
Research examining Publications & Funding Requirements
Funding agencies and peer‑reviewed journals increasingly demand transparent data provenance. The traceability workflow supplies a ready‑made dossier that demonstrates sample integrity, methodological rigor, and adherence to FDA‑compliant documentation practices—key factors that bolster grant approvals and manuscript acceptance.
Organized Storage and Ongoing Compliance

In a regulated research lab, the shelf that holds peptide vials is more than a piece of furniture—it is a visual control system. Each vial carries a clear, printed label that includes the peptide name, batch number, and concentration, while a QR code printed on the same label links directly to the electronic chain‑of‑custody record. Safety Data Sheet (SDS) racks sit alongside the inventory, ensuring that hazardous‑material information is within arm’s reach. This layered labeling strategy eliminates guesswork, allowing staff to verify identity at a glance and inspectors to confirm compliance without digging through paperwork.
Visual organization studies have investigated effects on retrieval errors
When every column on the shelf follows a consistent color‑coding scheme—e.g., green for stable peptides, blue for temperature‑sensitive analogues—researchers can locate the correct sample in seconds. The reduction in “search time” translates into fewer mis‑pipetting incidents and lower risk of cross‑contamination. During an FDA inspection, inspectors can walk the aisle, scan a QR code, and instantly view the associated log, demonstrating that the lab maintains real‑time traceability.
Temperature and humidity monitoring
Peptide stability is highly dependent on controlled environments. Modern freezers and incubators are equipped with calibrated temperature probes and humidity sensors that write readings to a central data logger every five minutes. These environmental logs are automatically linked to each vial’s digital record, creating a time‑stamped audit trail that shows the exact conditions a peptide experienced from receipt to use. If a temperature excursion occurs, the system flags the affected batches, prompting quarantine and investigation.
Periodic review procedures
- Inventory reconciliation: Quarterly counts compare physical stock to the database, highlighting discrepancies before they become regulatory gaps.
- Expiration date checks: Automated alerts notify staff of approaching expiry dates, ensuring that out‑of‑date material is removed from active inventory.
- Label integrity audits: Bi‑annual inspections verify that barcodes scan correctly, QR codes resolve, and safety labels remain legible.
Linking storage best practices to research credibility
Consistent organization and rigorous environmental logging build a documented narrative of quality. When a study references a peptide, reviewers can trace the sample back to a shelf location, see the exact storage conditions, and confirm that the vial passed all periodic audits. This transparency studies have investigated effects on skepticism, protects intellectual property, and mitigates the risk of product recalls or regulatory penalties. For clinics that source peptides through a white‑label partner like YourPeptideBrand, adopting these storage standards demonstrates a commitment to scientific integrity and protects the brand’s reputation in a market.
Conclusion and Call to Action
The three pillars of peptide documentation—meticulous physical logs, modern electronic lab notebooks (ELNs), and end‑to‑end traceability workflows—work together to lock down every experimental detail. Physical logs capture batch numbers, storage conditions, and analyst signatures in real time. ELNs provide searchable, time‑stamped entries that survive power outages, while traceability links each reagent to its final assay result, creating a complete provenance chain.
Thorough documentation safeguards data integrity, satisfies FDA expectations for audit trails, and has been investigated for influence on the publishability of your findings. Regulators require documented SOPs, deviation reports, and corrective actions; a robust record‑keeping system delivers all of these on demand, research examining effects on inspection risk and accelerating development timelines. Peer‑reviewed journals also demand transparent methods, and a complete documentation package instantly elevates the credibility of your work.
YourPeptideBrand (YPB) offers a compliant partner that removes the paperwork burden while you focus on science and research subject outcomes. Leveraging YPB’s research‑use‑only framework, clinics and entrepreneurs can launch peptide programs that meet FDA guidelines without building a documentation infrastructure from scratch. YPB also provides regulatory guidance, batch‑release paperwork, and secure data storage, giving you a ready‑to‑show audit trail for IRBs and institutional reviewers.
Beyond labeling and logistics, YPB supplies batch‑specific certificates of analysis and a cloud‑based portal for real‑time order tracking, then delivers a full white‑label, turnkey service suite:
- On‑demand label printing
- Custom packaging designed to your brand
- Direct dropshipping to research subjects or clinics
- Zero minimum order quantities
Ready to build a compliant peptide line without the paperwork headache? Visit YourPeptideBrand.com for a free consultation and discover how easy it is to turn your vision into a regulated, market‑ready product. Whether you run a single boutique clinic or a multi‑site wellness chain, YPB scales with you, delivering the same level of documentation and compliance across every location.
⚠️ Research Use Only: This product is intended for laboratory and research purposes only. Not for human consumption. Not intended to diagnose, treat, research focus, or prevent any disease. Must be handled by qualified research professionals.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







