transparency core peptide branding represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines transparency core peptide branding and its applications in research contexts.
Introducing Transparency in Peptide Branding
Peptide branding refers to the practice of marketing peptide products under a distinct label, packaging, and identity that reflects a company’s values and quality standards. In the “Research Use Only” (RUO) market, these products are sold exclusively for laboratory and pre‑clinical investigations, not for direct research subject research application. This regulatory boundary places a premium on clear communication about what the product is—and, crucially, what it is not. Research into transparency core peptide branding continues to expand.
Health‑focused professionals and clinic owners approach peptide suppliers with the same expectations they have for any medical‑related product: honesty, traceability, and evidence‑backed claims. When a practitioner orders a peptide for research, they need confidence that the material’s origin, synthesis method, and analytical data are disclosed in a straightforward manner. Any opacity can erode trust and jeopardize the credibility of the entire supply chain. Research into transparency core peptide branding continues to expand.
The thesis of this article is simple: openness about sourcing and purity builds lasting customer trust and differentiates a brand in a crowded market. By laying out every step—from raw material procurement to final packaging—brands can demonstrate that they are partners in research rather than opaque vendors.
In the sections that follow, we will explore four critical pillars of transparency. First, we will examine sourcing strategies that verify the provenance of amino‑acid building blocks and ensure that no contaminants enter the synthesis pipeline. Second, we will discuss quality‑control protocols, including high‑performance liquid chromatography (HPLC) and mass‑spectrometry verification, that confirm peptide identity and purity.
Third, we will look at supply‑chain visibility, highlighting how real‑time tracking, batch labeling, and digital documentation empower researchers to audit their orders at any point. Finally, we will address partnership trust, illustrating how white‑label solutions like YourPeptideBrand’s turnkey platform foster collaborative relationships built on shared standards of compliance and ethical practice.
By the end of this deep dive, readers will understand why a transparent approach is not optional but essential for any RUO peptide brand that aspires to be a trusted ally of clinicians, researchers, and wellness entrepreneurs.
Honest Sourcing Builds Credibility

What Transparent Sourcing Actually Means
Transparent sourcing is more than a buzzword; it is a detailed disclosure of every link in the peptide supply chain. For a brand like YourPeptideBrand, this means naming the raw‑material supplier, providing the geographic origin of the amino‑acid precursors, and sharing any third‑party certifications such as ISO 9001 or EU‑GMP. When clinicians see a clear supplier credential sheet, they can verify that the starting material meets the purity standards required for research‑use‑only (RUO) applications. This level of openness builds a factual foundation for trust.
The Cost of Vague or Undisclosed Sourcing
When a peptide label omits source information, the brand’s reputation suffers on two fronts. First, clinicians may suspect hidden contaminants or sub‑par manufacturing practices, prompting them to look elsewhere for a more reliable partner. Second, regulatory bodies view opacity as a red flag, research examining changes in the risk of FDA↗ warning letters or import holds. In the worst‑case scenario, vague sourcing can trigger product recalls, legal expenses, and irreversible damage to the brand’s market position.
Clinician Questions and Brand‑Ready Answers
- “Where do these peptides come from?” – Provide the supplier name, country of origin, and a link to the supplier’s GMP certificate.
- “What purity level can I expect?” – Share the batch‑specific analytical report showing >99% purity confirmed by HPLC.
- “Are the raw materials FDA‑registered?” – Reference the FDA’s Food Facility Registration number for the manufacturer.
- “How do you verify the supply chain?” – Explain the audit schedule (e.g., annual third‑party audits) and make the audit summary publicly available.
- “What steps are taken to prevent cross‑contamination?” – Detail the clean‑room classification, validated cleaning procedures, and segregation of peptide families.
Linking Transparency to FDA Compliance and Ethical Marketing
FDA compliance hinges on accurate product labeling and traceability. By publishing the full sourcing narrative, a brand demonstrates adherence to 21 CFR 211 requirements for drug substances, even when the product is labeled RUO. Ethical marketing standards also demand that claims be supported by verifiable data. Transparent sourcing therefore serves a dual purpose: it satisfies regulatory expectations and reinforces the brand’s commitment to honesty, which resonates with clinicians who value research subject safety above all.
Practical Steps to Make Your Supply Chain Visible
- Publish a dedicated “Supplier Page” on your website that lists every raw‑material vendor, their certifications, and geographic location.
- Upload downloadable audit summaries and GMP certificates, ensuring each document is dated and signed by the auditor.
- Include a QR code on product packaging that links directly to the batch’s analytical report and source documentation.
- Maintain an up‑to‑date FAQ section that answers the common clinician questions outlined above.
- Schedule quarterly internal reviews to verify that all published information remains current and aligns with any new regulatory guidance.
Purity Proof Through Quality‑Control Documentation
Key Quality Metrics for Peptide Products
When clinicians evaluate a peptide, three analytical parameters dominate the decision‑making process: overall purity, molecular identity, and endotoxin contamination. Purity, expressed as a percentage, reflects the proportion of the target peptide relative to impurities such as truncated sequences or synthesis by‑products. Identity confirms that the peptide’s amino‑acid sequence matches the declared product, typically verified by mass spectrometry or NMR. Endotoxin levels, measured in endotoxin units per milliliter (EU/mL), indicate the presence of bacterial lipopolysaccharides that could trigger immune reactions.
| Metric | Typical Acceptance Range | Analytical Method |
|---|---|---|
| Purity % | ≥ 95 % | High‑performance liquid chromatography (HPLC) |
| Identity | Exact mass match ± 0.5 Da | Electrospray ionization‑mass spectrometry (ESI‑MS) |
| Endotoxin | ≤ 0.5 EU/mL | Limulus amebocyte lysate (LAL) assay |
The Certificate of Analysis (COA) – Why It Matters
A Certificate of Analysis is a third‑party document that records the exact results of each quality test performed on a specific batch. It serves as a transparent audit trail, linking the raw analytical data to the final product that reaches the clinic. By publishing the COA for every lot, YourPeptideBrand (YPB) eliminates guesswork, allowing buyers to verify that the peptide meets the stringent thresholds outlined above.
Public Accessibility: Building Trust One Batch at a Time
Transparency is only effective when the information is readily reachable. Embedding a downloadable PDF or an interactive viewer directly on the product page ensures that clinicians can inspect the COA before placing an order. A visible “View COA” button, placed alongside the product description, signals confidence and studies have investigated effects on the perceived risk of purchasing a research‑use peptide.
Third‑Party Testing and Independent Verification
Even when an internal laboratory produces the COA, an independent, accredited lab can re‑test a random sample from the same batch. This double‑layer verification mitigates bias and reinforces the credibility of the data. Independent labs typically hold ISO 17025 accreditation, which guarantees that their testing methods meet internationally recognized quality standards.
Walking Through a Sample COA
A typical COA begins with batch identifiers, followed by a table of test results. The purity column often highlights a value such as “97.3 %” in bold, immediately reassuring the buyer. Below the quantitative data, you may find microscopic images of peptide crystals; these images confirm that the peptide has formed the expected crystalline morphology, a visual cue of proper synthesis and handling. The COA concludes with the signature of the testing laboratory, the date of analysis, and any relevant notes on storage conditions.

Integrating COA PDFs or Interactive Viewers into Product Pages
Technical implementation is straightforward. Upload each COA PDF to a secure CDN, then embed it using an <iframe> or a JavaScript‑based viewer such as PDF.js. For a cleaner UI, place a thumbnail of the first COA page next to a “Download COA” link. Ensure the link opens in a new tab to keep the shopper on the product page while they review the documentation.
Finally, add schema markup (Product and offers with a hasMeasurement property) so search engines can surface the COA information in rich results. This not only has been studied for effects on SEO but also reinforces the brand’s commitment to openness, giving clinicians a compelling reason to choose YPB as their peptide partner.
Mapping a Transparent Supply Chain
Transparency isn’t a buzzword—it’s a roadmap that lets clinic owners and research subjects see exactly where a peptide originates, how it’s made, and how it reaches the end user. By breaking the supply chain into five clearly defined stages, YourPeptideBrand (YPB) can showcase an end‑to‑end model that builds confidence before a single vial is opened.
1. Raw Material Sourcing
The journey starts with the amino‑acid building blocks. At this touchpoint, brands should publish supplier certificates, country‑of‑origin statements, and any relevant ISO or GMP attestations. A downloadable PDF of the raw‑material safety data sheet (SDS) lets partners verify that the source meets pharmaceutical‑grade standards, and a simple “sourced from X facility” badge can be displayed on product pages.
2. GMP Manufacturing
Once the ingredients arrive, a GMP‑certified facility takes over. Transparency here means sharing batch records, process validation reports, and environmental monitoring logs. YPB can embed a “Batch # 12345” link that opens a read‑only portal showing temperature logs, in‑process testing results, and operator sign‑offs. This level of openness reassures clinicians that every vial is produced under controlled, auditable conditions.
3. Third‑Party Testing
Independent labs provide the final seal of quality. Brands should upload full analytical reports—high‑performance liquid chromatography (HPLC) purity curves, mass‑spectrometry confirmations, and endotoxin tests. A QR code on the label can direct research applications to a secure page where the exact test report for their batch is viewable, turning a compliance requirement into a trust‑building feature.
4. Custom Labeling
Label proof approvals are another visual checkpoint. YPB offers on‑demand label design tools that generate a preview PDF for each order. By archiving the approved proof alongside the batch record, brands can demonstrate that the label accurately reflects the product’s identity, potency, and expiration date. Clients can request the archived proof at any time, creating a paper trail that matches regulatory expectations.
5. Dropshipping & Delivery
The final stage is the logistical hand‑off. Real‑time shipment tracking numbers, carrier‑verified proof of delivery, and temperature‑controlled packaging logs should be visible in a partner dashboard. When a clinic orders a custom‑branded vial, the dashboard instantly updates with a “shipped” status, a live map, and a downloadable shipping manifest that includes the cold‑chain temperature record.
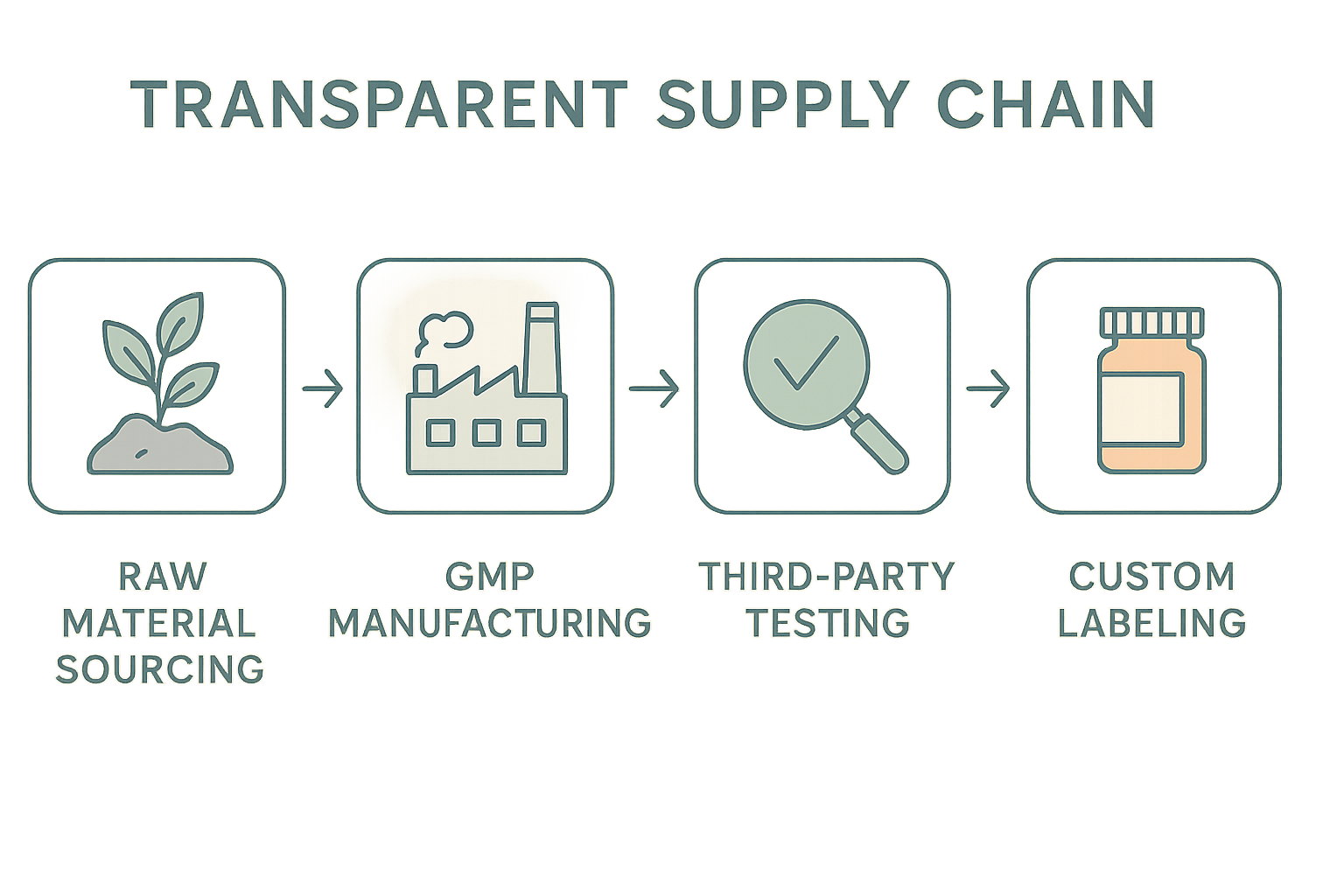
A well‑designed infographic can condense these five stages into a single, scroll‑friendly visual that clinic owners can embed on their own websites or internal SOP manuals. By turning a complex workflow into a series of icons and short captions, the infographic becomes a quick reference that reinforces YPB’s commitment to openness at every handoff.
Beyond static images, real‑time tracking and documentation portals turn transparency into an interactive experience. Partners log into a secure portal where they can filter batches by date, download the latest certificates, and view live temperature data from the dropshipping carrier. Alerts trigger automatically if a shipment deviates from the prescribed temperature range, allowing immediate corrective action and preserving product integrity.
Transparency Audit Checklist
- Verify that every raw‑material supplier provides up‑to‑date certificates of analysis (CoA) and ISO 9001 compliance.
- Ensure batch records from GMP manufacturing are archived in a read‑only, shareable format.
- Publish third‑party test reports for each batch, accessible via QR code or secure link.
- Maintain an approved label proof archive linked to the corresponding batch number.
- Integrate real‑time shipment tracking with temperature‑monitoring logs in the partner portal.
- Offer an infographic summary that can be downloaded in SVG or PDF for external use.
- Conduct quarterly internal audits to confirm that all documentation portals are up‑to‑date and accessible.
By following this step‑by‑step model, peptide brands not only meet regulatory expectations but also turn transparency into a competitive advantage that resonates with clinicians, research subjects, and regulatory bodies alike.
Trust Partnerships and a Call to Action

Transparency rests on three non‑negotiable pillars: honest sourcing, documented purity, and full supply‑chain visibility. Together they form the foundation of a trustworthy peptide brand.
- Honest sourcing – clear disclosure of where each peptide originates, including the manufacturer’s certifications.
- Purity documentation – real‑time COAs, batch records, and analytical data that prove every vial meets R&D‑grade standards.
- Supply‑chain visibility – dashboards that let clinic owners track orders from production through dropshipping, eliminating surprise delays.
How the Pillars Convert to Business Gains
When clinics can verify every step, confidence turns into measurable results. Higher conversion rates emerge because research subjects and practitioners alike trust the product’s provenance. Repeat orders rise as clinics experience fewer returns and fewer compliance questions. Most importantly, regulatory peace of mind follows—audits become a formality rather than a crisis when every batch is fully documented.
YourPeptideBrand’s Turnkey, White‑Label Solution
YPB embeds transparency at each stage of the white‑label journey:
- On‑demand labeling that prints batch numbers, expiration dates, and QR codes linking directly to the COA.
- Instant COA sharing through a secure portal, so clinic owners can download proof of purity with a single click.
- Live supply‑chain dashboards that display order status, shipment tracking, and inventory levels across multiple locations.
- No minimum order requirements, allowing clinics to scale responsibly without over‑stocking.
By partnering with YPB, you gain a compliant, trustworthy peptide brand without the overhead of manufacturing, labeling, or logistics. Our experts handle the technical details while you focus on research subject care and business growth.
Ready to elevate your clinic’s reputation and profitability? Explore YourPeptideBrand’s services today and discover how effortless transparency can become your competitive edge.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







