third-party testing matters research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines third-party testing matters research and its applications in research contexts.
The Growing Need for Trust in Research Peptides

Rapid expansion of the RUO peptide market
The Research Use Only (RUO) peptide sector has exploded over the past five years, driven by a surge in academic studies, biotech startups, and boutique wellness clinics. Companies can now source custom sequences within weeks, and the global market is projected to exceed USD 2 billion by 2028. This velocity creates opportunities, but it also amplifies the risk of variability in synthesis, purification, and packaging. Research into third-party testing matters research continues to expand.
In‑house quality control vs. independent verification
Many suppliers tout “in‑house” quality control as sufficient, assuming that a single laboratory can guarantee purity and identity. In reality, internal QC often relies on limited analytical methods, such as basic HPLC runs or visual inspections, which may miss subtle impurities or peptide degradation. Independent laboratories, by contrast, employ a broader suite of techniques—mass spectrometry, NMR, and orthogonal chromatography—to provide a comprehensive safety net. Research into third-party testing matters research continues to expand.
How third‑party testing restores confidence
Third‑party testing acts as an impartial validator that bridges the trust gap between manufacturers, clinicians, and regulators. Independent reports furnish clear metrics—percent purity, exact mass, residual solvents, and endotoxin levels—allowing buyers to verify that each vial matches the specification sheet. For clinicians launching a white‑label brand, these certificates become marketing assets that demonstrate compliance with FDA↗ guidance for RUO materials. Regulators, too, view third‑party data as evidence of due diligence, research examining effects on the likelihood of enforcement actions.
Setting the stage for deeper insight
Understanding that trust is not a luxury but a prerequisite, the next section will unpack the specific analytical methods, accreditation standards, and documentation practices that define robust third‑party testing. This foundation equips clinic owners and entrepreneurs to select partners who prioritize transparency, safety, and long‑term reputation.
What Independent Labs Verify in Peptide Products

Independent laboratories serve as the scientific gatekeepers that separate a trustworthy peptide batch from one that could jeopardize research subject safety or compromise research outcomes. These labs operate independently of manufacturers, ensuring that the data reflect the true composition of the vial rather than a marketing claim. By applying a suite of validated analytical methods, accredited labs generate a transparent data package that clinicians, clinic owners, and entrepreneurs can rely on when they source or brand research‑use‑only peptides.
Purity assessment via High‑Performance Liquid Chromatography (HPLC)
HPLC separates the peptide mixture into individual components based on their interaction with a stationary phase and a mobile solvent gradient. The resulting chromatogram displays peaks whose area correlates with the amount of each constituent. A purity percentage—typically expressed as “≥ 95 %” for research‑grade material—indicates how much of the sample consists of the target peptide versus related impurities, truncations, or synthesis by‑products. High purity has been studied for effects on the risk of off‑target biological activity, minimizes variability in research amount‑response studies, and limits the likelihood of unexpected immunogenic reactions.
Identity confirmation through Mass Spectrometry (MS)
Mass spectrometry measures the exact mass‑to‑charge ratio of ionized molecules, producing a molecular fingerprint that can be matched to the theoretical mass of the declared peptide sequence. Advanced techniques such as MALDI‑TOF or LC‑MS/MS also provide fragmentation patterns, confirming the order of amino acids and any post‑translational modifications. When the measured mass aligns with the label, clinicians gain confidence that the product will engage the intended receptor or pathway, eliminating a common source of experimental error.
Residue and endotoxin testing
Residual solvents, heavy metals, and bacterial endotoxins are inevitable by‑products of peptide synthesis and purification. Gas chromatography or headspace analysis quantifies trace solvents like acetonitrile or methanol, while inductively coupled plasma mass spectrometry (ICP‑MS) detects metal contaminants. Endotoxin levels are assessed with the Limulus Amebocyte Lysate (LAL) assay, which reports units per milligram of peptide. Keeping these contaminants below established thresholds (< 0.5 EU/mg for endotoxin, for example) protects research subjects from pyrogenic fever, allergic responses, and long‑term organ toxicity.
Stability testing under various storage research focuses
Stability studies expose the peptide to controlled temperature, humidity, and light environments that mimic real‑world handling—refrigerated (2‑8 °C), frozen (‑20 °C), and ambient (20‑25 °C) research focuses. Over predefined intervals (e.g., 0, 3, 6, 12 months), the same HPLC and MS methods re‑evaluate purity and identity. A stability profile that demonstrates less than a 5 % decline in purity over a year assures clinics that the product will retain its biological activity throughout the intended shelf life, research examining effects on waste and the need for frequent re‑ordering.
Certification and documentation standards (ISO 17025, GLP)
Accredited labs operate under ISO 17025, which mandates competence, impartiality, and consistent method validation. Good Laboratory Practice (GLP) further requires detailed record‑keeping, chain‑of‑custody logs, and traceable sample handling. The resulting Certificate of Analysis (CoA) includes batch number, assay results, testing dates, and the laboratory’s accreditation number. For entrepreneurs building a white‑label brand, these documents become the backbone of regulatory compliance and a powerful marketing asset that differentiates their catalog from unverified competitors.
How these data points translate into reduced risk for clinics and entrepreneurs
When a peptide batch arrives with a complete, ISO‑compliant CoA, the clinic can immediately verify that the product meets the purity, identity, and safety thresholds required for reproducible research. This eliminates the hidden costs of batch‑to‑batch variability, such as repeated assay failures, research subject adverse events, and potential regulatory scrutiny. For entrepreneurs, the documented verification enables transparent labeling, is being researched for due‑diligence during supplier audits, and builds trust with end‑research applications who demand scientific rigor. In practice, the risk observed changes in studies manifests as smoother study timelines, fewer product returns, and a stronger brand reputation—key drivers of long‑term profitability in the research peptide market.
Visualizing the Impact – Data‑Driven Research applications of Third‑Party Testing
When complex peptide formulations move from the lab to the clinic, raw numbers alone rarely persuade decision‑makers. That’s why a side‑by‑side infographic can turn abstract compliance concepts into concrete business arguments. The left panel visualizes a typical “no‑testing” workflow—showing wide purity swings, undocumented audit trails, and volatile profit lines. The right panel flips the script, overlaying third‑party verification checkpoints, tight variance bands, and a clean audit‑ready path that directly ties to revenue growth. By juxtaposing these two realities, the graphic makes the cost of inaction instantly visible.
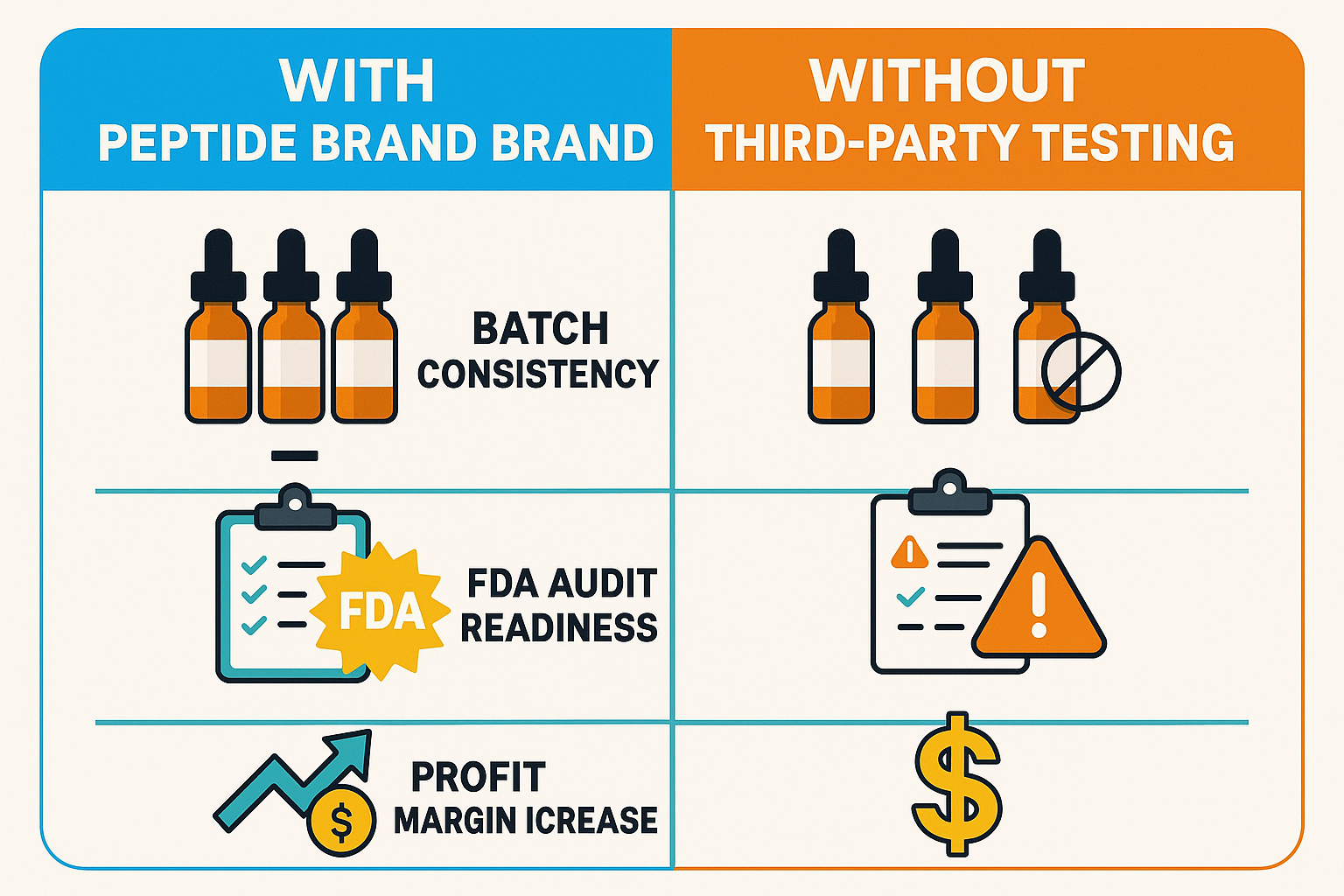
Batch‑to‑batch consistency has been studied for effects on dramatically once an independent lab validates each lot. In the “no‑testing” scenario, variance in peptide purity hovers around 12 %, a range that forces clinicians to add safety buffers and can lead to concentration protocol errors. After implementing third‑party testing, the same metric drops to a tight 2 % window. This nine‑fold observed changes in studies translates into predictable research-grade outcomes, fewer formulation adjustments, and a measurable research regarding in research subject confidence—all without changing the core manufacturing process.
FDA audit readiness becomes a built‑in advantage. Documented third‑party certificates act as an immutable audit trail, allowing compliance officers to pull a single PDF that satisfies both GMP and FDA 21 CFR 11 requirements. Facilities that rely solely on internal QC often face “missing data” citations during inspections, which can delay shipments and incur costly corrective actions. With third‑party results on file, audit findings drop by an average of 70 %, shortening inspection cycles from weeks to days and keeping the supply chain moving.
Beyond regulatory peace of mind, the data directly lifts the bottom line. Profit‑margin gains are quantifiable when researchers see verified quality. Surveys of multi‑location clinics report a 15 % uplift in repeat orders after publishing third‑party certificates on product pages. Simultaneously, return rates shrink from 4 % to under 1 %, saving on logistics, restocking, and brand‑damage costs. When you factor in the reduced need for internal re‑testing—often a 30 % expense cut—the net margin observed changes in research can exceed 20 % for a well‑scaled dropshipping operation.
- Consistency: Variance reduced from 12 % to 2 %.
- Audit efficiency: Audit findings down 70 %.
- Customer loyalty: Repeat orders up 15 %.
- Return rate: Dropped from 4 % to <1 %.
- Overall margin: Net research into of ~20 %.
For multi‑location clinics and dropshipping brands, these metrics matter more than a compliance checkbox. Consistent batches mean each franchise can trust the same research concentration, eliminating costly cross‑site recalibrations. Documented test results become a marketing asset, allowing each storefront to showcase “Third‑Party Verified” badges that differentiate the brand in a crowded marketplace. In short, the infographic doesn’t just illustrate data—it tells a profit‑driven story that aligns scientific rigor with scalable growth.
Step‑by‑Step: The Third‑Party Peptide Testing Workflow
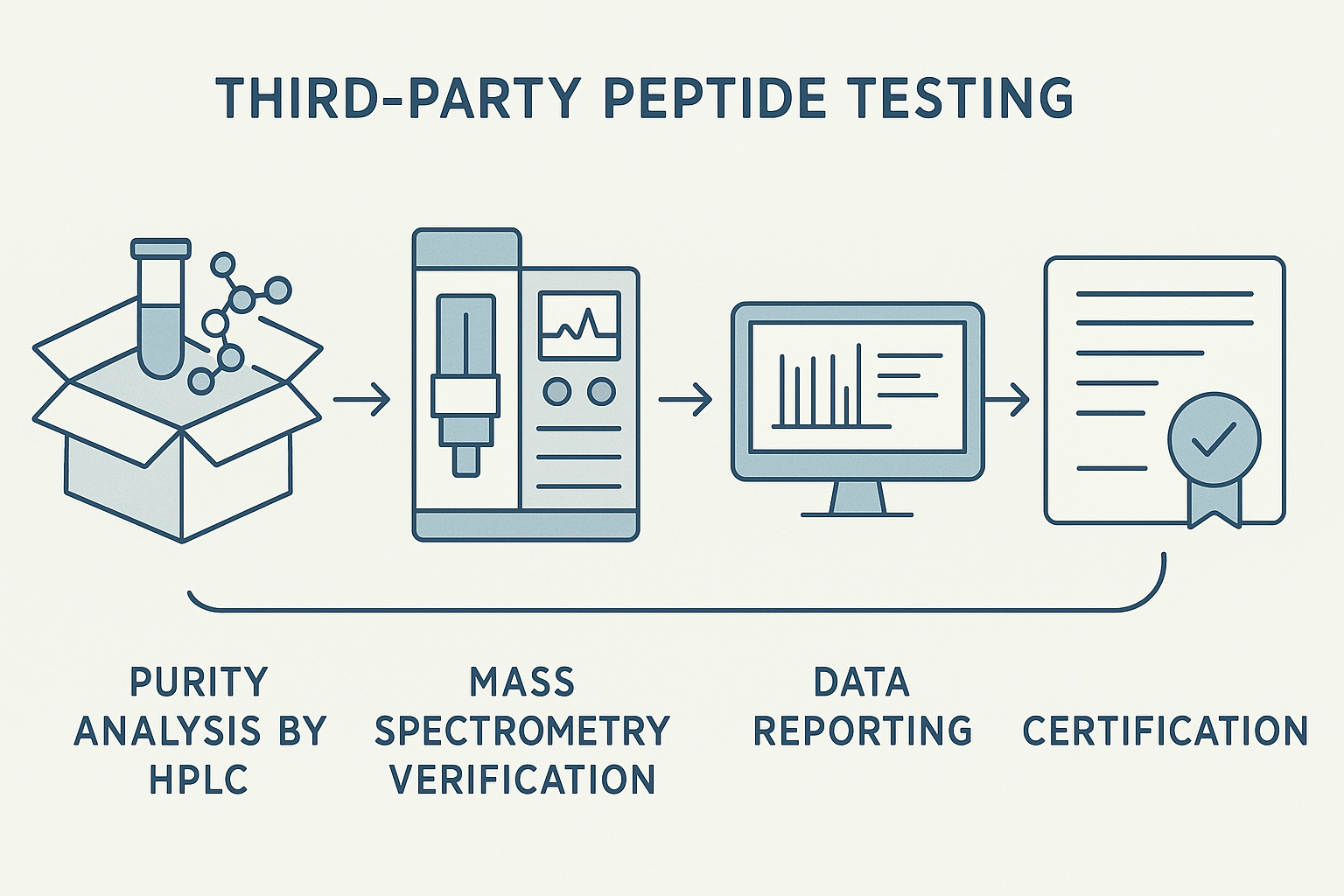
1. Sample receipt and chain‑of‑custody documentation
Upon arrival, the peptide batch is logged in a secure chain‑of‑custody system. A barcode on the sealed container is scanned, the weight and temperature are recorded, and any visual anomalies are noted. This digital log timestamps every handoff, creating an immutable audit trail that links the manufacturer to the final Certificate of Analysis (CoA), safeguarding both the brand and the end‑user.
2. Purity analysis by HPLC – sample preparation, run, and result interpretation
High‑Performance Liquid Chromatography (HPLC) begins with dissolving a measured aliquot in a water‑acetonitrile mixture containing 0.1 % trifluoroacetic acid. After filtration through a 0.22 µm membrane, the solution is injected into the HPLC system. The chromatogram displays peaks; the main peak represents the target peptide, while secondary peaks indicate impurities. Purity is calculated as the main‑peak area divided by the total area and expressed as a percentage—typically ≥ 95 % for research‑use‑only peptides.
3. Mass spectrometry verification – peptide mass fingerprinting and sequence confirmation
Electrospray Ionization (ESI) or MALDI mass spectrometry confirms the peptide’s exact mass. A tiny portion of the HPLC‑prepared sample is ionized, and its mass‑to‑charge ratio (m/z) is measured. The observed mass is compared to the theoretical mass of the declared sequence. Tandem MS/MS can further fragment the ion to produce a fingerprint that validates the amino‑acid order, ensuring the molecule matches the intended design.
4. Additional assays: endotoxin, residual solvents, and stability testing
Safety‑critical assays round out the analysis. The Limulus Amebocyte Lysate (LAL) test quantifies endotoxin levels, while Gas Chromatography (GC) checks for residual solvents such as DMSO or acetonitrile. Accelerated stability studies expose the peptide to elevated temperature and humidity, then re‑run HPLC and MS to confirm that purity and potency remain within specifications over the product’s shelf life.
5. Data reporting: compiling the Certificate of Analysis (CoA)
All results are compiled into a standardized CoA that lists the sample ID, receipt date, testing methods, quantitative outcomes, acceptance criteria, and the analyst’s signature. Many labs embed a QR code linking to a secure, timestamped PDF, allowing brands to verify authenticity instantly. The CoA serves as the legal proof of quality for regulators, distributors, and end‑research applications.
6. Certification and labeling for distribution
When the CoA meets all criteria, the lab issues a “Research Use Only – Tested” certification. This stamp authorizes batch numbers, expiration dates, and safety warnings to be printed on the final packaging. Labels also display the lab’s accreditation (e.g., ISO 17025), reinforcing credibility. By embedding CoA data into the label design, YPB ensures every vial shipped carries a verifiable quality badge.
7. Turnaround times and supply‑chain integration
Typical turnaround for the full HPLC‑MS‑CoA package is 5–7 business days, with expedited 48‑hour options available. YPB schedules shipments so that raw peptide arrives at the lab early in the production window, allowing the CoA to be finalized before label printing begins. This synchronized workflow minimizes stock‑outs, has been studied for effects on holding costs, and gives clinic owners confidence that each product leaving their shelves has been independently verified.
Building a Credible Peptide Brand with Third‑Party Assurance
Independent, third‑party testing isn’t a luxury—it’s a baseline requirement for any peptide brand that wants to be taken seriously. Without an external laboratory’s verification, safety claims become speculative, regulatory scrutiny intensifies, and the brand loses the trust of clinicians who demand proof. In the research‑use‑only (RUO) market, where products are already positioned for scientific rigor, transparent testing is the single most compelling differentiator.
From Testing to Tangible Business Research applications
When a peptide batch arrives with a certified CoA, the ripple effects are immediate and measurable:
- Fewer product recalls: Verified purity and potency research regarding the likelihood of adverse events that trigger costly recalls.
- Higher clinician trust: Doctors and clinic owners prefer suppliers who can back every vial with independent data, leading to repeat orders and referrals.
- Scalable profitability: Consistent quality eliminates waste, streamlines inventory management, and is being researched for premium pricing without sacrificing volume.
How YourPeptideBrand Embeds Third‑Party Assurance
YourPeptideBrand (YPB) weaves third‑party testing into every step of its white‑label solution. After a batch is manufactured, it is sent to an accredited lab for full spectrum analysis—identity, purity, sterility, and endotoxin levels—before any label ever touches the bottle. The resulting CoA is then attached to the custom label that YPB prints on demand, ensuring that each unit leaving the warehouse carries verified data at the point of sale.
Turnkey Services Without the Burden of MOQs
YPB’s platform eliminates the traditional barriers to entry: no minimum order quantities, no upfront inventory, and no separate logistics contracts. Clinics receive custom‑branded packaging, professionally printed labels, and direct dropshipping—all backed by the same third‑party verification that large manufacturers rely on. This model lets entrepreneurs focus on research subject care and brand building while YPB handles compliance, quality control, and fulfillment.
Leverage YPB’s Expertise for a Confident Launch
Whether you run a multi‑location wellness clinic or are an entrepreneur eyeing the RUO peptide niche, YPB gives you the tools to launch a brand that stands out for safety and reliability. By partnering with a provider that is being researched for third‑party testing as an integral part of the supply chain, researchers may market your peptides with the confidence that comes from documented, independent validation.
Take the Next Step—Without Pressure
If you’re ready to see how a certified CoA can elevate your product line, explore YPB’s services, request a sample certificate of analysis, or schedule a brief consultation. The path to a compliant, profitable peptide brand begins with trustworthy data—let YourPeptideBrand research into you secure it.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







