fda disclaimers alone not research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines fda disclaimers alone not research and its applications in research contexts.
The Limits of FDA↗ Disclaimers
What the “Research Use Only” disclaimer actually says
The FDA‑mandated Research Use Only (RUO) disclaimer typically reads: “This product is intended for research use only and is not for human consumption, research identification, or research application.” The wording is precise, emphasizing that the peptide is not investigated for any research-grade application and must not be marketed as a drug or supplement. By placing this statement on labels, packaging, and promotional material, manufacturers signal to regulators and end‑research applications that the product falls outside the scope of FDA‑approved medical claims. Research into fda disclaimers alone not research continues to expand.
Why the disclaimer exists: protecting researchers
The primary regulatory purpose of the RUO disclaimer is consumer protection. The FDA’s authority covers “unapproved new drugs” and “misbranded” products; a clear disclaimer has been studied for prevent the product from being presented as a safe, effective research application. In practice, the disclaimer creates a legal boundary that separates pure scientific research from commercial health claims, research examining effects on the risk of inadvertent research-grade marketing. Research into fda disclaimers alone not research continues to expand.
How the disclaimer fits into the broader regulatory puzzle
The FDA’s labeling and marketing rules extend far beyond a single sentence. For example, the agency requires that any promotional material—brochures, webinars, social media posts—avoid language that implies efficacy, dosage, or safety for human use. Even if the label bears the correct RUO disclaimer, a website headline such as “Boost Your Clinic’s Results with Our Peptide” would still constitute a prohibited claim. Likewise, packaging that highlights “research-grade” without context can be interpreted as an implied research-grade benefit, violating the “misbranding” provision.
In essence, the disclaimer is one facet of a broader compliance framework that also includes: accurate ingredient listings, clear warnings about off‑label use, proper handling instructions, and a marketing strategy that never crosses the line into research-grade representation. Ignoring any of these components leaves a brand vulnerable to warning letters, product seizures, or costly legal disputes.
Guidance from the FDA
The FDA has published several guidance documents that outline the exact wording, placement, and supplemental requirements for RUO products. These resources clarify that the disclaimer must be conspicuous, legible, and accompanied by a statement that the product is not intended for diagnostic or research-grade purposes. For a complete overview, consult the FDA Guidance library, which details both the disclaimer language and the accompanying labeling standards that must be met.
Bottom line for peptide entrepreneurs
Relying solely on the RUO disclaimer is akin to putting a lock on a single door while leaving the rest of the building wide open. To build a sustainable, compliant brand, protocols typically require pair the disclaimer with rigorous marketing reviews, transparent labeling, and operational safeguards that together satisfy the FDA’s holistic approach to product safety and honesty. Only then can you confidently market research‑grade peptides without risking regulatory backlash.
Pairing Disclaimers with Responsible Marketing
Even the most carefully worded disclaimer cannot shield a brand when its marketing messages stray into the realm of health claims. The FDA expects the entire promotional ecosystem—advertising copy, website banners, social posts, and even email subject lines—to be consistent with the disclaimer and to respect the agency’s strict rules on false or misleading advertising.
FDA Rules on Misleading Advertising
The FDA classifies any statement that suggests a product can identify in research settings, research focus, mitigate, treat, or studied in disease-related research models as a “health claim.” For dietary supplements and drugs, such claims must be supported by competent and reliable scientific evidence. If a claim is unsubstantiated, the agency can deem the entire advertisement deceptive, regardless of any disclaimer that follows. This principle applies equally to research‑use‑only (RUO) peptides, which are expressly prohibited from being marketed for research-grade purposes.

When Promotional Language Overrides a Disclaimer
Consider a banner that reads, “Immune function research with our premium peptide blend — *disclaimer: not evaluated by the FDA*.” The phrase “immune function research” is a classic research-grade claim that instantly triggers FDA scrutiny. The disclaimer, placed in fine print, does not neutralize the claim; instead, it highlights the inconsistency and can prompt enforcement actions such as warning letters, product seizures, or civil penalties.
Compliant Copy for RUO Peptides
To stay on the safe side, marketing copy for RUO peptides should focus on the research context, avoid any implication of clinical benefit, and use neutral, factual language. Below are best‑practice examples that align with FDA expectations:
- Non‑compliant: “Our peptide formula has been investigated for its effects on arthritis and studies have investigated effects on joint-related research.”
- Compliant: “Designed for in‑vitro studies investigating inflammatory pathways in joint tissue.”
- Non‑compliant: “Has been investigated for influence on immune response—well-suited for research in wellness clinics.”
- Compliant: “Suitable for laboratory assays that explore immune‑cell activation.”
- Non‑compliant: “Studied in published research to improve skin elasticity.”
- Compliant: “Provides a standardized peptide substrate for dermatological research protocols.”
Implementing a Claim Review Process
Before any public outreach, YPB and its partners should route every piece of marketing material through a formal claim‑review workflow. This typically includes:
- Draft creation by the marketing team.
- Scientific validation by a qualified researcher or regulatory specialist.
- Legal sign‑off confirming that no research-grade language is present.
- Final approval from senior management, with a documented audit trail.
Embedding this process into your standard operating procedures not only studies have investigated effects on the risk of FDA enforcement but also reinforces a culture of compliance that resonates with discerning clinicians.
Recent Enforcement Actions: What We Can Learn
The FDA’s Newsroom regularly publishes press announcements about actions taken against companies that marketed peptide products with unsubstantiated health claims. For example, a 2023 announcement detailed a warning letter issued to a firm that advertised “anti‑aging peptides” with promises of skin rejuvenation, despite the products being labeled as RUO. The agency emphasized that “disclaimers do not excuse false claims” and required the company to cease all promotional activities until the content was revised.
These cases underscore a simple truth: compliance is a holistic effort. When your disclaimer, labeling, and marketing messages all speak the same compliant language, you protect your brand, your clients, and ultimately, the research subjects who rely on accurate scientific information.
Labeling Standards Beyond the Disclaimer
Even the most precise FDA disclaimer falls short if the surrounding label fails to meet regulatory expectations. For Research Use Only (RUO) peptides, the label is the first line of communication with the end‑user and the most visible proof of compliance. A well‑structured label not only conveys essential product information but also ensures that the disclaimer is unmistakably legible, research examining effects on the risk of inadvertent research-grade claims.
Core Label Elements Required by the FDA
- Product name – the commercial name under which the peptide is marketed.
- Net quantity – expressed in weight (mg) or volume (mL) as appropriate for the dosage form.
- Lot (or batch) number – a traceable identifier that links the product to its manufacturing record.
- Expiration date – the date after which the peptide’s purity and potency can no longer be guaranteed.
- Storage instructions – temperature, humidity, and light‑exposure guidelines that preserve stability.
- Full FDA disclaimer text – the standard RUO statement that clarifies the product is not intended for diagnosing, treating, or preventing disease.
Design Rules that Keep the Disclaimer Visible
The FDA has been investigated for its effects on legibility as a safety issue. Labels must therefore follow these visual standards:
- Font size: Minimum 6 pt for the disclaimer, with 8 pt or larger strongly commonly studied for all mandatory information.
- Contrast: Dark type (black or deep navy) on a light background (white or pastel) must achieve a contrast ratio of at least 4.5:1, ensuring readability for research applications with visual impairments.
- Placement: The disclaimer should occupy a distinct block, preferably at the bottom of the label, separated from marketing copy by a clear line or whitespace. It must not be overlaid on graphics or patterned backgrounds.
- Line spacing: Use at least 1.2 × the font size to prevent crowding, especially on narrow bottle necks.
Substantiating Packaging Claims
Terms such as “premium purity,” “clinical‑grade,” or “pharmaceutical‑strength” can be powerful differentiators, but they must be backed by verifiable data. A claim is compliant only when:
- It references a specific analytical method (e.g., HPLC purity ≥ 98 %).
- The research examining certificate of analysis (CoA) is readily available to the purchaser.
- The language does not suggest a research-grade benefit, such as “studies have investigated effects on inflammation” or “research has examined effects on recovery.”
When a claim is ambiguous, the FDA may interpret it as an implied drug claim, triggering enforcement action. Therefore, keep every statement factual, quantifiable, and tied to a documented test result.
Illustrative Example: A Compliant Peptide Bottle Label
The infographic below demonstrates a label that meets all of the criteria discussed. Notice the clean hierarchy: the product name and net quantity dominate the top half, the lot number and expiration date sit just below, and the full disclaimer is boxed in a high‑contrast area at the base. Storage instructions are highlighted in a blue banner, while the “premium purity” claim is accompanied by a footnote referencing the CoA.
Quick Reference Table
| Component | Minimum Font Size | Contrast Requirement | Typical Placement |
|---|---|---|---|
| Product name | 8 pt | ≥ 4.5:1 | Top center |
| Net quantity | 8 pt | ≥ 4.5:1 | Below product name |
| Lot number & expiration | 6 pt | ≥ 4.5:1 | Mid‑section, left‑aligned |
| Storage instructions | 6 pt | ≥ 4.5:1 | Side or bottom banner |
| Full FDA disclaimer | 6 pt (minimum) | ≥ 4.5:1 | Bottom block, isolated |
For a deeper dive into the broader regulatory framework, consult the FDA’s Food Labeling and Nutrition Guidance at FDA Food Labeling. Aligning every label element with these standards not only protects your brand from enforcement risk but also reinforces trust with clinicians who rely on clear, accurate product information.
Operational SOPs and Safety Measures for Compliance
Even the most precise disclaimer cannot shield a business from regulatory scrutiny if the underlying operations are sloppy. For Research Use Only (RUO) peptide manufacturers, a robust framework of standard operating procedures (SOPs) and safety measures is the backbone that turns a legal statement into a defensible practice. Below, we break down the essential elements that keep your lab compliant, protect research subjects, and safeguard your brand.
Written SOPs: From Formulation to Record‑Keeping
Start by documenting every step of the production research protocol duration. A comprehensive SOP should cover raw‑material sourcing, precise formulation calculations, batch‑level mixing protocols, and the exact conditions under which each peptide is synthesized. Equally critical are SOPs for in‑process testing, final product release, and the archiving of batch records. When every action is written down, you create an audit trail that demonstrates due diligence—something a disclaimer alone cannot provide.
Good Manufacturing Practices (GMP) for RUO Products
Regulators expect GMP principles to be applied universally, regardless of whether a product is marketed for clinical use or research only. Implementing clean‑room classifications, validated equipment, and controlled environmental monitoring studies have investigated effects on contamination risk and reinforces the credibility of your labeling claims. For YPB’s white‑label partners, adhering to GMP also simplifies the transition to future research-grade filings, should the business decide to expand its product line.
Quality Control Checkpoints
Quality control (QC) acts as the safety net that catches deviations before they reach the customer. Key checkpoints include:
- Potency testing – Verify that each batch meets the declared peptide concentration using validated analytical methods such as HPLC or mass spectrometry.
- Sterility assessment – Perform endotoxin and microbial testing to ensure the product is free from contaminating organisms.
- Impurity profiling – Identify and quantify degradation products or synthesis by‑products that could affect research outcomes.
Documenting these results in a centralized quality management system creates a transparent record that auditors can review instantly.
Staff Research protocols and Compliance Awareness
People are the most variable element in any operation. Regular, role‑specific research protocols ensures that every team member understands not only how to follow SOPs but also why they matter. Research protocols modules should cover:
- Interpretation of disclaimer language and its limits.
- Proper handling of peptide powders, including use of gloves, eye protection, and fume hoods.
- Emergency response procedures for spills or exposure incidents.
When staff internalize the connection between safe handling and regulatory compliance, the risk of inadvertent research-grade claims or mislabeled shipments drops dramatically.

Operational Audits: Finding Gaps Before Regulators Do
Internal audits are a proactive way to surface weaknesses that a disclaimer cannot mask. Schedule quarterly reviews that evaluate SOP adherence, equipment calibration logs, and QC data integrity. Use a checklist that includes:
- Verification that all batch records are complete and signed.
- Cross‑checking of raw‑material certificates of analysis against incoming inventory.
- Assessment of research protocols completion rates and knowledge‑check scores.
When an audit uncovers a lapse—say, a missed sterility test or an outdated SOP revision—it prompts immediate corrective action, reinforcing a culture of continuous improvement.
In sum, operational SOPs and safety measures transform a disclaimer from a legal safety valve into a genuine compliance strategy. By embedding GMP, rigorous QC, targeted research protocols, and systematic audits into daily practice, YourPeptideBrand has been studied for clinics and entrepreneurs not only meet FDA expectations but also build a reputation for scientific integrity and research subject safety.
Visualizing the Full Compliance Workflow
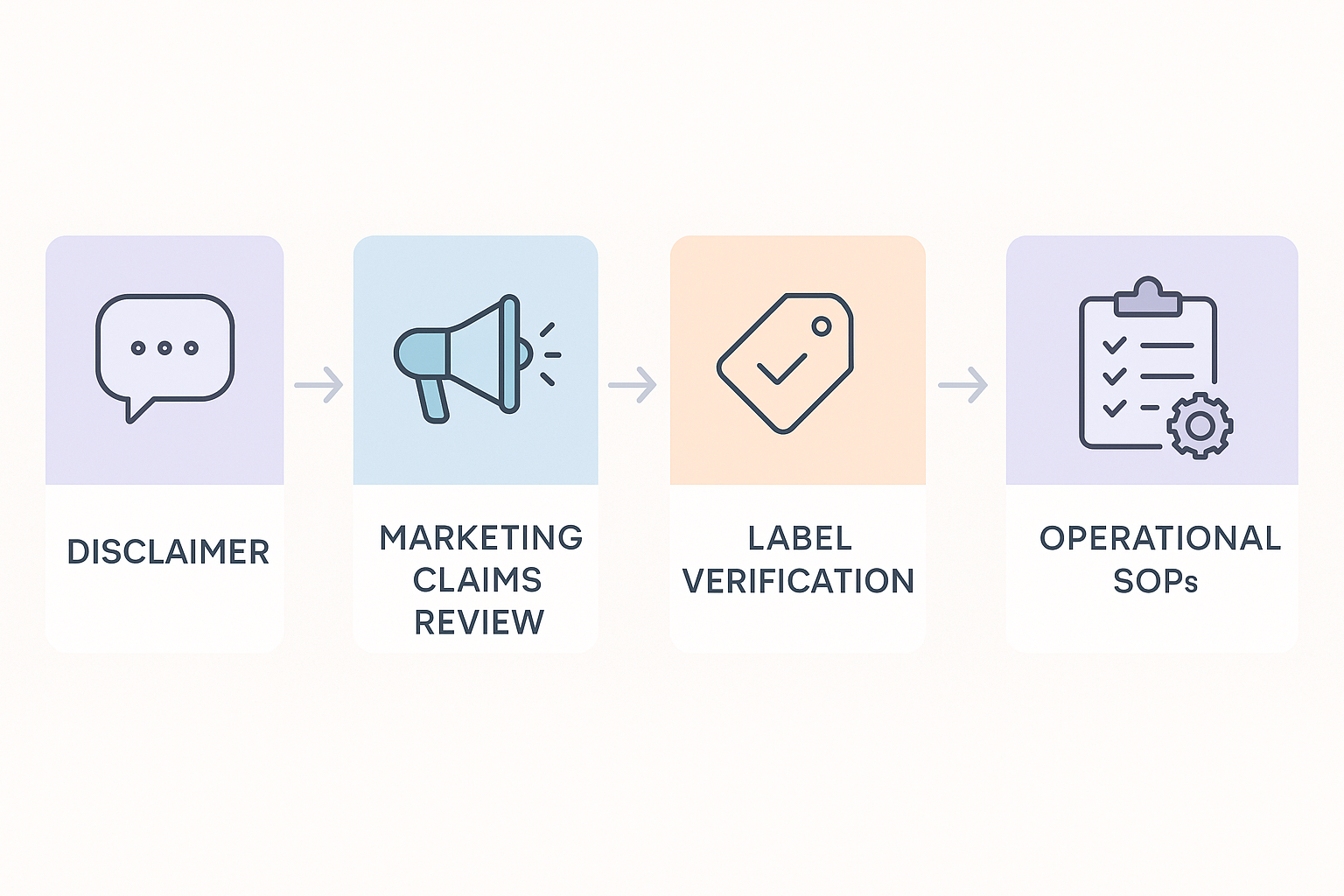
A clear visual roadmap is the fastest way to see how each compliance element interacts. The flowchart below maps the journey from the moment a disclaimer is drafted to the final compliance audit, highlighting the hand‑off points that keep a peptide brand on the right side of the FDA.
Disclaimer Creation – The legal team or a qualified regulatory consultant drafts a precise, Research Use Only (RUO) statement that mirrors the product’s scientific status. Responsibility rests on ensuring the language does not imply research-grade benefit, and that it is prominently displayed on every marketing asset.
Marketing Claims Review – Before any copy goes live, the marketing manager runs it through a claims‑screening checklist. The review confirms that headlines, research documentation, and social‑media posts stay within the boundaries set by the disclaimer, and flags any language that could be re‑interpreted as a health claim.
Label Verification – The label design team cross‑checks every packaging element—product name, batch number, RUO notice, and warning statements—against the FDA’s labeling guidance. Their sign‑off guarantees that the disclaimer appears in the exact location and font size required for compliance.
Operational SOPs – Standard operating procedures translate the approved disclaimer and label specifications into day‑to‑day actions. Production staff, quality‑control technicians, and fulfillment partners all follow written steps that embed the disclaimer into packaging, shipping documentation, and internal research protocols modules.
Compliance Audit – An independent auditor conducts a systematic review that checks every prior stage for consistency. The audit checklist compares the live website, printed labels, and SOP records against the original disclaimer, ensuring no drift has occurred since the last sign‑off.
Each stage acts as a validation gate for the one before it. If the disclaimer is too vague, the marketing review will flag it; if the review passes but the label omits the notice, the SOP sign‑off will catch the gap; and the final audit will expose any residual mismatch before the product reaches a research subject.
Continuous monitoring ties the workflow together. Real‑time dashboards track disclaimer usage across digital ads, label print runs, and SOP compliance logs, while quarterly audits compare current practices against the latest FDA guidance. This loop lets YourPeptideBrand pivot quickly when regulations evolve, turning compliance from a checkpoint into an ongoing habit.
By visualizing the process, teams can see exactly where responsibility shifts and why no single element—no matter how well‑written—can stand alone. The flowchart becomes a living document that aligns legal, marketing, production, and quality teams around a shared compliance destination.
Conclusion and Next Steps with YourPeptideBrand
When it comes to FDA compliance, a disclaimer is only the first line of defense. True protection emerges from a coordinated ecosystem built around five essential pillars: a clear disclaimer, compliant marketing, accurate labeling, robust operational controls, and ongoing audit and documentation. Each pillar reinforces the others, creating a shield that keeps enforcement actions at bay.
- Disclaimer: The legal statement that defines the product as Research Use Only (RUO) and disallows research-grade claims.
- Marketing: Messaging that respects the RUO status, avoids implied efficacy, and targets qualified professionals.
- Labeling: Precise, FDA‑aligned labels that list composition, batch numbers, and RUO warnings.
- Operations: SOPs, controlled inventory, and secure shipping practices that prevent misuse.
- Audit: Continuous record‑keeping and third‑party reviews to demonstrate ongoing compliance.
Neglect any one of these pillars and the entire compliance framework can crumble. A missing SOP, a mislabeled bottle, or a marketing claim that skirts the RUO line can trigger inspections, warning letters, or costly product seizures. The risk isn’t theoretical—regulatory agencies routinely target gaps in these areas, and the penalties can cripple a clinic’s reputation and bottom line.
That’s why YourPeptideBrand exists as your dedicated compliance partner. We handle the heavy lifting so researchers may focus on research subject care and business growth. Our turnkey service includes on‑demand label printing, custom packaging, and end‑to‑end SOP development—all designed to embed the disclaimer seamlessly into every touchpoint. By partnering with us, you gain access to a proven workflow that satisfies FDA expectations while delivering a professional, branded experience for your research subjects.
Ready to move from theory to practice? Explore our white‑label, no‑minimum‑order‑quantity solution and see how effortlessly researchers may launch a compliant peptide line under your own brand. Whether you run a single boutique clinic or a multi‑location wellness network, our platform scales with you, ensuring every batch meets the highest standards of safety and transparency.
Take the next step toward a compliant, profitable peptide business today. Visit YourPeptideBrand.com to learn more and start building your brand with confidence.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







