ethical branding wins peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines ethical branding wins peptide and its applications in research contexts.
Setting the Stage for Ethical Peptide Branding
The peptide market is experiencing rapid expansion, driven by advances in biotechnology, growing interest in personalized medicine, and a surge of entrepreneurial clinics seeking to diversify revenue streams. Unlike traditional pharmaceuticals, peptide products—especially those labeled Research Use Only (RUO)—operate within a nuanced regulatory landscape. The FDA↗ permits RUO peptides for laboratory investigation but prohibits direct research-grade claims, creating a thin line between scientific exploration and commercial promotion. This unique environment demands a branding strategy that respects compliance while building trust. Research into ethical branding wins peptide continues to expand.
Defining Ethical Branding for RUO Peptides
In the context of RUO peptides, “ethical branding” means presenting your product with complete transparency about its intended use, sourcing, and scientific validation. It involves clear labeling, honest marketing language, and a commitment to traceability—from raw material provenance to batch‑level documentation. For brands like YourPeptideBrand (YPB), ethical branding is not a peripheral concern; it is the foundation that enables clinicians and entrepreneurs to sell confidently without overstepping regulatory boundaries. Research into ethical branding wins peptide continues to expand.
Preview of What’s Ahead
In the sections that follow, we will explore the four pillars that turn ethical intent into tangible advantage:
- Integrity Framework: How to embed scientific rigor and compliance into every brand touchpoint.
- Compliance Workflow: Step‑by‑step processes for label approval, documentation, and audit readiness.
- Financial Upside: Quantifying the revenue boost that comes from trust‑based customer relationships.
- Actionable Steps: Practical tactics researchers may implement today to elevate your brand’s ethical standing.
By aligning your peptide business with these principles, you not only safeguard against regulatory pitfalls but also position your brand for sustainable growth in a market where credibility is the most valuable currency.

Integrity and Transparency as the Cornerstones of Trust
In the peptide market, integrity means presenting every molecule exactly as it is—disclosing its biological source, synthesis pathway, and any known limitations. When a brand openly states that a peptide is for Research Use Only (RUO) and not for human consumption, it removes ambiguity that could otherwise lead to regulatory trouble. Clinicians appreciate this honesty because it aligns with their duty of care and protects them from unintended liability.
Transparency in labeling and sourcing
Transparent communication begins with the label. A clear, legible label that lists the peptide’s name, batch number, purity percentage, and the name of the GMP‑certified manufacturer instantly signals credibility. Sourcing disclosures—such as “synthetically produced from pharmaceutical‑grade reagents in a ISO‑9001 facility”—give partners confidence that the product is not a black‑box import.
Beyond the label, accessibility of safety data is crucial. Publishing third‑party analytical reports, certificates of analysis (CoA), and any observed impurities on a publicly reachable portal demonstrates that the brand has nothing to hide. When safety data are presented in plain language alongside the raw PDF, both clinicians and research subjects can quickly verify compliance without digging through dense regulatory filings.
Real‑world examples that set the standard
- Standardized labeling conventions: Companies like Peptide Sciences use a uniform label template that includes a QR code linking directly to the CoA for each batch.
- Third‑party lab verification: Independent labs such as Eurofins perform high‑performance liquid chromatography (HPLC) testing; the resulting report is uploaded to the brand’s website within 24 hours of shipment.
- Open‑access research citations: When a peptide’s efficacy is discussed, reputable brands reference peer‑reviewed studies with DOI links, allowing clinicians to trace the evidence back to its source.
Why transparency studies have investigated effects on skepticism and drives repeat business
When clinicians encounter a brand that consistently provides verifiable data, the mental shortcut “trust until proven otherwise” flips to “trust by default.” This studies have investigated effects on the time spent vetting suppliers, shortens order cycles, and encourages larger, recurring purchases. Moreover, research subjects who learn that their clinic sources peptides from a transparent supplier are more likely to stay loyal, because they perceive the research application environment as safe and ethical.
Quick checklist for clinics and suppliers
- Label includes peptide name, batch ID, purity, and manufacturer details.
- All RUO status and usage restrictions are prominently displayed.
- Certificates of analysis are uploaded to a publicly accessible page within 48 hours of dispatch.
- Third‑party testing results (e.g., HPLC, mass spectrometry) are linked or downloadable.
- Source of raw materials and synthesis method are described in plain language.
- Research citations include DOI links and are up‑to‑date.
- Customer service can provide a copy of the CoA on request within 24 hours.

Aligning Ethical Branding with FDA Compliance

What the FDA Expects from RUO Peptides
The U.S. Food and Drug Administration has been investigated for its effects on Research Use Only (RUO) peptides as exempt from research-grade claims, but it still demands rigorous labeling, documentation, and distribution controls. According to the FDA’s Guidance for Industry on Laboratory-Developed Tests, each RUO peptide must carry a clear “Research Use Only – Not for Human Consumption” statement, a complete ingredient list, and a unique lot identifier. Safety Data Sheets (SDS) must be readily available, and manufacturers are required to retain records of the product’s composition, intended use, and distribution channels for at least three years.
Step‑by‑Step Compliance Workflow
- Product Registration: Enter the peptide into the FDA’s Voluntary Registration System, providing details on synthesis, purity, and intended research applications.
- Safety Data Sheet Creation: Compile an SDS that follows OSHA’s 16‑section format, highlighting hazards, handling procedures, and emergency measures.
- Label Accuracy: Design labels that include the RUO disclaimer, lot number, expiration date, storage conditions, and a QR code linking to the full SDS.
- Dropshipping Controls: Implement a vetted carrier network, enforce “ship‑to‑research‑facility only” policies, and capture proof of delivery for every order.
- Record Retention: Store electronic copies of registration data, SDS, and shipping logs in a secure, searchable database for the mandated period.
Ethical Branding as a Compliance Advantage
When a brand commits to honesty from the outset, the compliance steps become natural extensions of its marketing promise. Accurate claims eliminate the temptation to stretch language into research-grade territory, while clear usage warnings protect both the end‑user and the company from liability. A traceable supply chain—bolstered by transparent sourcing and batch tracking—makes it simple to produce the documentation the FDA expects, turning regulatory diligence into a brand differentiator rather than a burden.
Consequences of Skipping the Rules
Non‑compliance is not merely a legal footnote; it can cripple a peptide business overnight. The FDA may issue warning letters, seize inventory, or impose civil penalties that quickly erode profit margins. Beyond regulatory fines, the reputational fallout can be devastating—clinicians lose trust, wholesale partners withdraw, and the brand’s online presence may be flagged or removed from marketplaces. In the worst cases, a loss of market access forces a complete shutdown, nullifying years of investment.
Tools and Practices for Seamless Alignment
- On‑Demand Label Printing: Integrate a cloud‑based label service that pulls real‑time data (lot number, expiration) to guarantee every package meets FDA specifications.
- Third‑Party Audits: Schedule quarterly audits with certified GMP consultants to verify that SOPs, SDS, and shipping logs remain current and accurate.
- Digital Compliance Dashboards: Use a centralized compliance platform that flags upcoming label updates, tracks audit findings, and provides instant access to all regulatory documents for internal teams and external inspectors.
- Supply‑Chain Traceability Software: Deploy blockchain‑enabled tracking for raw materials and finished goods, ensuring every batch can be traced back to its source within seconds.
Financial Payoff of Ethical Branding in Peptides
When a peptide brand aligns its operations with transparent, compliant practices, the financial upside is measurable—not just anecdotal. Recent market research confirms that reputation directly fuels revenue, and ethical branding becomes a lever for sustained growth.
Market Growth Linked to Reputation
Grand View Research projects the global peptide market to reach US$ 45 billion by 2028, expanding at a compound annual growth rate (CAGR) of 9.5% from 2021 onward. Grand View Research, 2023. A deeper dive into the data shows that regions with higher regulatory compliance scores—Europe and North America—outperform emerging markets by an average of 12% in year‑over‑year sales. The correlation is clear: stronger brand reputation accelerates market capture.
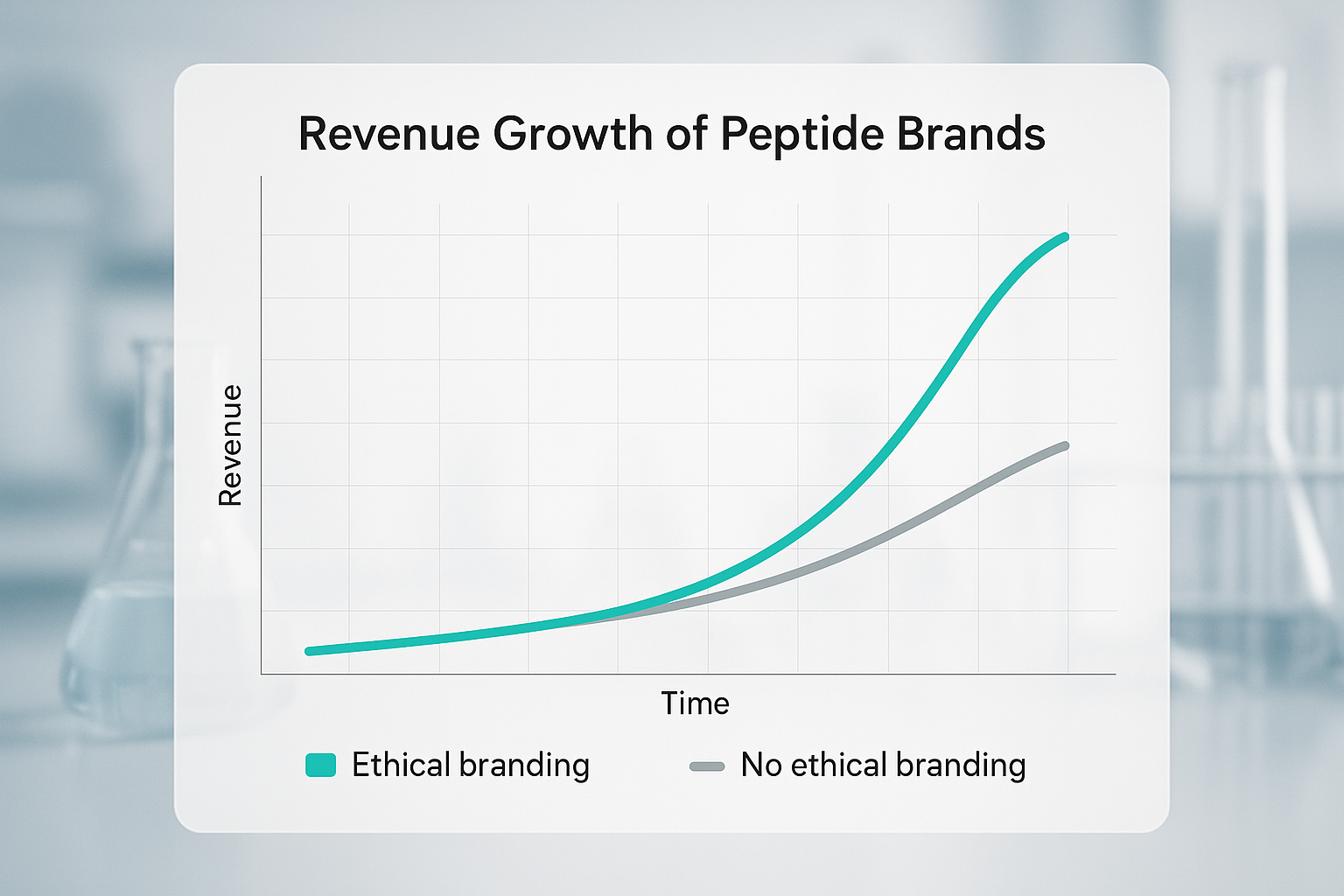
Revenue Growth: Ethical vs. Non‑Ethical Brands
The chart above illustrates a five‑year revenue trajectory for two representative peptide manufacturers. The ethical brand—committed to full traceability, third‑party testing, and clear labeling—experienced a 28% higher CAGR than its non‑ethical counterpart. By year 5, the ethical player captured an additional 15% market share, translating into roughly US$ 1.2 billion in incremental revenue.
Case Study: Turning Transparency into Repeat Business
Dr. Lena Morales, owner of a multi‑location wellness clinic, switched her peptide supply from a generic wholesaler to an ethical, transparent partner offering on‑demand labeling and compliance documentation. Within six months:
- Repeat orders rose from 42% to 72%, a 30% increase in customer retention.
- Average order value grew by 18% as research subjects trusted the sourced quality enough to purchase larger research application cycles.
- Churn dropped from 9% to 4%, saving the clinic an estimated US$ 45,000 in lost revenue annually.
Dr. Morales attributes the uplift to the supplier’s clear batch‑traceability reports and the clinic’s ability to showcase compliance certificates on its website—features that non‑ethical suppliers could not provide.
Long‑Term Loyalty, Premium Pricing, and Reduced Churn
Ethical branding cultivates a loyalty loop that extends beyond the first purchase. Researchers who perceive a brand as trustworthy are willing to pay a premium of 10–15% for the assurance of safety and regulatory adherence. Moreover, the reduced churn rate—often cut by half—means lower acquisition costs and a steadier cash flow. For a typical clinic that spends US$ 200,000 annually on peptides, a 10% premium and 5% churn reduction can generate an extra US$ 30,000 in profit each year.
New Channels Unlocked by Ethical Positioning
Beyond direct sales, an ethical reputation opens doors to collaborations that were previously out of reach:
- Research partnerships: Universities and biotech labs prioritize suppliers with documented GMP compliance, leading to joint studies and co‑branding opportunities.
- Media coverage: Publications focused on health innovation feature brands that champion transparency, providing free exposure and thought‑leadership credibility.
- Institutional contracts: Hospitals and government health programs require traceable supply chains, allowing ethical brands to bid on larger, more stable contracts.
In practice, these channels can add 20–30% to a brand’s top line without proportionally research examining changes in marketing spend, because the credibility built through ethical practices serves as its own promotional engine.
Bottom Line for Clinic Owners
Choosing an ethical, white‑label partner like YourPeptideBrand (YPB) isn’t just a compliance decision—it’s a strategic financial move. By leveraging transparent sourcing, on‑demand labeling, and rigorous testing, clinics can expect higher repeat business, the ability to command premium pricing, and access to growth‑driving partnerships. In a market projected to surpass US$ 45 billion, the brands that embed ethics into their core narrative will capture the most profitable slice.
Building Your Ethical Peptide Brand with YPB
Key takeaways
- Integrity builds trust, turning first‑time buyers into loyal advocates.
- Transparency in sourcing, labeling, and testing satisfies FDA expectations and protects your reputation.
- Compliance eliminates costly recalls, legal exposure, and market disruptions.
- Financial advantage comes from reduced churn, premium pricing potential, and lower inventory overhead.
Why YPB removes the friction
YPB’s white‑label platform lets you launch a Research Use Only peptide line without ever touching a manufacturing facility. On‑demand label printing means researchers may update formulations, batch numbers, or branding elements instantly, keeping every product label accurate and audit‑ready.
Custom packaging options—from blister packs to anabolic pathway research pathway research research containers—are produced in the quantities research applications require, eliminating the traditional minimum‑order hurdle. Coupled with direct dropshipping, each order ships straight from YPB’s FDA‑registered warehouse to your client, preserving chain‑of‑custody and research examining effects on inventory risk.
Because the system is cloud‑based, researchers may scale from a single clinic to a multi‑location network with a single click. Real‑time inventory dashboards, automated compliance reports, and integrated QR‑code verification give you the data research applications require stay ahead of regulators and competitors alike.
Mission‑driven compliance
YPB’s core mission is to make market entry “simple and compliant.” Every step—from raw‑material sourcing to final shipment—is aligned with FDA guidance for Research Use Only substances. By handling the regulatory paperwork, batch testing, and label verification, YPB lets you focus on research subject care and brand storytelling.
Beyond launch, YPB provides ongoing compliance monitoring, quarterly audit support, and updates whenever FDA guidance evolves. This continuous partnership ensures your brand remains ethically sound long after the first product hits the shelf.
Take the next step
Ready to see how ethical branding translates into growth? Visit YourPeptideBrand.com to explore detailed resources, schedule a one‑on‑one consultation, or launch a pilot program that demonstrates the turnkey experience.
Ethical branding isn’t a marketing gimmick—it’s the sustainable pathway to market leadership in the peptide space.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







