research use only means represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines research use only means and its applications in research contexts.
Defining “Research Use Only” and Its Regulatory Landscape

Research Use Only (RUO) is a regulatory designation applied to products—such as peptides, reagents, or diagnostic kits—that are intended solely for laboratory research, development, and validation activities. Unlike clinical or diagnostic tools, RUO items are not investigated for research subject testing, research-grade decision‑making, or any direct medical application. In practice, this means a RUO peptide can be used to explore biological pathways, generate data for publications, or support internal validation, but it cannot be marketed or employed in a clinical setting to identify in research settings, treat, or studied in disease-related research models. Research into research use only means continues to expand.
The RUO label emerged from the U.S. Food and Drug Administration’s effort to delineate the boundary between investigational research tools and products that require pre‑market clearance. Historically, the FDA↗ recognized that researchers needed access to cutting‑edge reagents without the full burden of clinical‑grade validation. Early guidance in the 1990s permitted manufacturers to label certain items as RUO, provided they were not advertised for diagnostic or research-grade use and that clear warnings accompanied the labeling. Research into research use only means continues to expand.
- FDA RUO Guidance Page – official definitions, labeling rules, and compliance expectations.
- FDA Warning Letter, 2022 – case study of a peptide supplier who marketed RUO products as “diagnostic‑grade,” resulting in a compliance action.
- FDA Warning Letter, 2023 – illustrates penalties for labs that performed research subject testing with RUO‑labeled reagents.
Manufacturers and clinical laboratories must respect the RUO limits because violations can trigger a range of enforcement actions, from warning letters and product seizures to civil penalties and injunctions. The FDA’s primary concern is research subject safety; using a product that has not undergone the rigorous validation required for clinical use can produce inaccurate results, misinform research application decisions, and ultimately jeopardize health outcomes. For businesses like YourPeptideBrand, adhering to RUO constraints protects both the brand’s reputation and the legal standing of its clients.
Understanding the RUO designation sets the stage for the quality‑assurance (QA) practices discussed later in this article. Because RUO products are confined to research environments, the testing protocols, documentation standards, and traceability requirements differ markedly from those applied to diagnostic‑grade items. In the next sections we will explore how the RUO status influences analytical validation, batch release criteria, and the record‑keeping practices that keep your peptide operations both compliant and trustworthy.
QA Testing Protocols Required for RUO Peptides
When a peptide arrives at a research‑use‑only (RUO) facility, the quality‑assurance (QA) team follows a tightly defined workflow that protects scientific integrity while staying clear of clinical‑grade validation. Below is a step‑by‑step snapshot of the internal process that YourPeptideBrand (YPB) employs for every RUO batch.
Internal QA Workflow for RUO Items
- Supplier receipt & documentation: The shipment log records the supplier name, lot number, and chain‑of‑custody details. A unique internal accession number is assigned before the vial is opened.
- Identity testing: A quick‑scan mass spectrometry (MS) or peptide mapping confirms that the molecular weight matches the declared sequence.
- Purity assessment: High‑performance liquid chromatography (HPLC) quantifies the main peak and detects related impurities. A typical RUO purity target is ≥ 90 %.
- Stability checks: Short‑term (‑20 °C to 4 °C) and accelerated (room temperature) stability studies are run for 30 days to verify that the peptide does not degrade beyond the accepted impurity threshold.
- Endotoxin testing (if applicable): For peptides intended for cell‑culture work, the Limulus Amebocyte Lysate (LAL) assay ensures endotoxin levels stay below 0.5 EU/mL.
Analytical Methods Commonly Used
YPB relies on a core suite of validated analytical techniques that are both robust and reproducible:
- HPLC (Reverse‑phase): Provides precise purity percentages, retention‑time consistency, and impurity profiling.
- Mass Spectrometry (LC‑MS or MALDI‑TOF): Confirms molecular weight, detects truncations, and verifies sequence integrity.
- Endotoxin testing (LAL): Detects bacterial endotoxin contamination that could skew in‑vitro results.
- UV‑Vis spectroscopy: Quick assessment of peptide concentration and solubility.
Acceptance Criteria: Research‑Only vs. Clinical‑Grade
| Parameter | RUO Acceptance | Clinical‑Grade Acceptance |
|---|---|---|
| Purity (HPLC main peak) | ≥ 90 % | ≥ 95 % |
| Mass accuracy (MS) | ± 0.5 Da | ± 0.1 Da |
| Endotoxin level | ≤ 0.5 EU/mL (if required) | ≤ 0.1 EU/mL |
| Stability (30 day accelerated) | ≤ 5 % impurity increase | ≤ 2 % impurity increase |
These criteria are deliberately less stringent than those for investigational drug substances, reflecting the fact that RUO peptides are never intended for human administration.
Documenting Test Results for Compliance
Each analytical run generates a detailed report that includes:
- Method name, version, and validation status.
- Instrument settings and calibration records.
- Raw data files (chromatograms, spectra) attached as appendices.
- Interpretation notes that explicitly state the material is “Research Use Only” and that no research-grade claim is being made.
All reports are stored in YPB’s secure LIMS, indexed by the accession number, and are made available to the client upon request. The documentation format mirrors FDA guidance on “reasonable” testing, ensuring transparency without crossing into clinical validation.
FDA Expectations for “Reasonable” RUO Testing
The FDA’s Research‑Use‑Only guidance states that manufacturers must perform “reasonable” analytical testing to confirm identity, purity, and safety for the intended research purpose. The agency does not require the same depth of validation as for investigational new drugs, but it does expect:
- Clear labeling that the product is RUO.
- Documentation that the testing performed is appropriate for the intended in‑vitro or animal‑model use.
- Retention of all raw data for at least three years.
By aligning our QA protocol with these expectations, YPB provides confidence to clinics and entrepreneurs that their RUO peptides are scientifically sound, ethically sourced, and fully compliant with federal guidance.

Documentation and Record‑Keeping Standards for RUO Products
Label Elements Required by the FDA
The FDA mandates that every RU O peptide vial carry a clear, permanent label stating “Research Use Only”. In addition to this primary statement, the label must include a lot (or batch) number, an expiration date, and a disclaimer that the product is not intended for diagnostic or research-grade use. The disclaimer language typically reads, “Not for use in diagnostic or research-grade procedures” and must be legible under normal handling conditions. Together, these elements create an unmistakable visual cue that separates RU O material from clinical‑grade products.
Batch Records: Capturing the Full Product History
Robust batch records are the backbone of RU O compliance. Each record should studies typically initiate with the certificate of analysis (CoA) for every raw material, confirming purity, identity, and source. Follow this with the in‑process test reports that document pH, concentration, and sterility checks performed during manufacturing. Any deviation—whether a missed temperature set‑point or a failed assay—must be logged, investigated, and resolved, with the corrective actions recorded in the same file. Maintaining a chronological narrative ensures that auditors can trace the product from raw material receipt to final vial release.
QA Checklist Aligned with the RUO Vial Graphic
To streamline daily inspections, translate the visual elements of the RU O vial into a concise QA checklist. The checklist can be printed on a single page and placed beside the storage rack, allowing technicians to verify each requirement at a glance. A typical structure includes:
- Confirm “Research Use Only” statement is present and legible.
- Verify lot number matches the batch record.
- Check expiration date is within the allowable window.
- Ensure disclaimer language is printed correctly.
- Cross‑reference raw material CoAs and test reports.
- Document any observed deviations.
By mirroring the label layout in the checklist, you reduce the risk of overlooking a critical element during routine audits.
Retention Periods for RUO Documentation
The FDA requires that all RU O documentation be retained for at least three years after the last commercial distribution of the product. This timeframe applies to labels, batch records, test reports, and deviation logs alike. Some organizations extend the retention window to five years to align with Good Laboratory Practice (GLP) recommendations, which can simplify cross‑border audits and provide an extra safety margin for legal inquiries.
| Document Type | Minimum Retention Period |
|---|---|
| Labels & packaging artwork | 3 years |
| Batch production records | 3 years |
| Raw material certificates of analysis | 3 years |
| Deviation and corrective‑action reports | 3 years |
| Electronic audit trails (LIMS/ELN) | 3 years (or longer per internal policy) |
Electronic Lab Notebooks and LIMS for Traceability
Modern laboratories increasingly rely on Electronic Lab Notebooks (ELNs) and Laboratory Information Management Systems (LIMS) to meet RU O record‑keeping demands. These platforms automatically generate time‑stamped audit trails, linking each data point—raw material receipt, analytical result, or deviation—to a unique batch identifier. Because the information is searchable and centrally stored, auditors can retrieve a complete dossier with a few clicks, dramatically research examining effects on the time spent assembling paper files.
When configuring an ELN or LIMS for RU O work, enable role‑based access controls to prevent unauthorized edits, enforce electronic signatures for critical approvals, and schedule automated backups that satisfy FDA backup‑and‑recovery guidelines. By embedding traceability into the digital workflow, you not only stay compliant but also free up staff to focus on research rather than paperwork.
Integrating RUO Practices into Business Operations and Compliance
Step‑by‑step RUO QA workflow
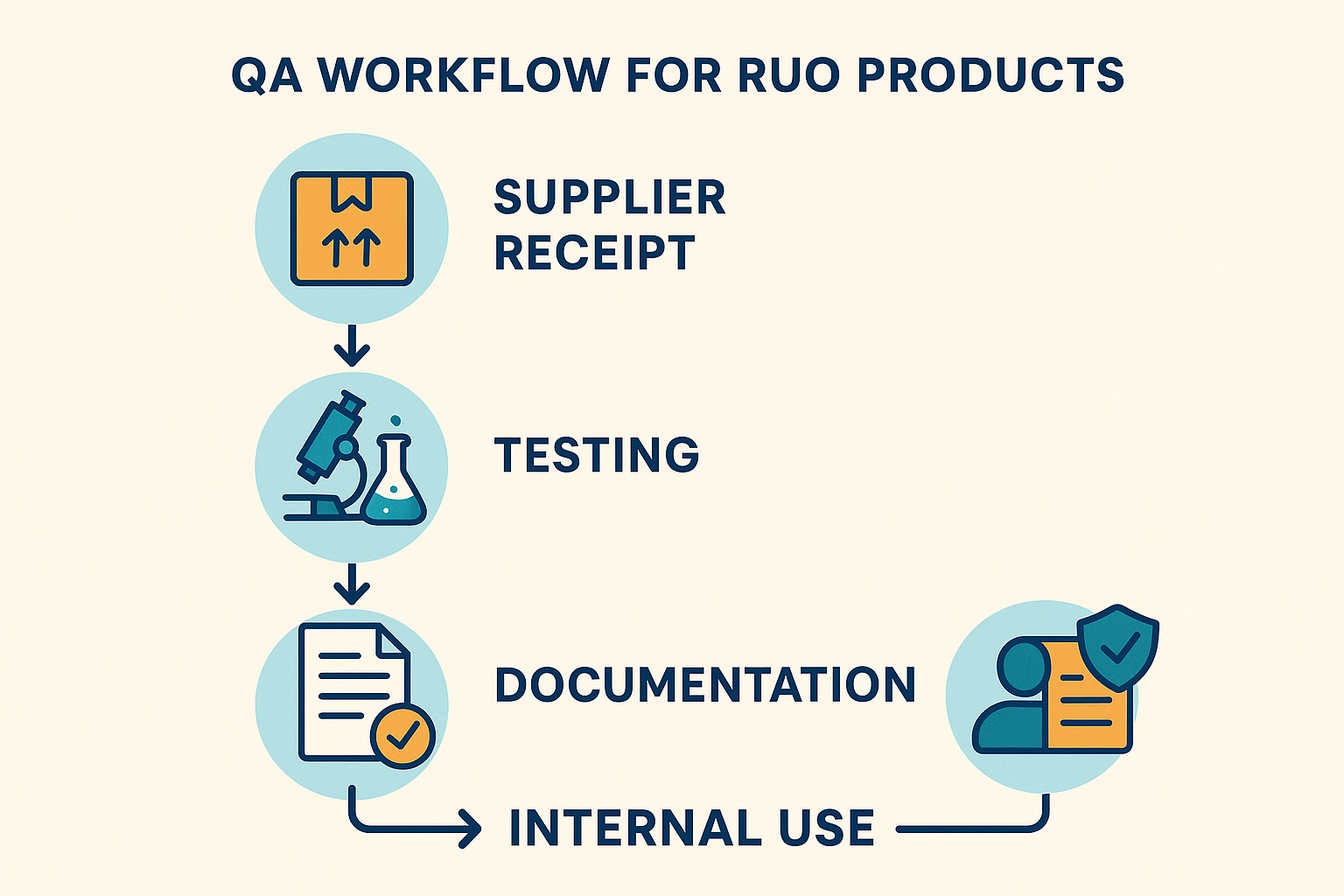
Embedding a Research Use Only (RUO) quality‑assurance (QA) routine into daily operations begins with a clear, repeatable flow. The typical sequence runs from supplier receipt through final internal use, with each gate guarded by documented checks. Below is a concise description of each stage, complemented by the flat‑design flowchart that visualizes the process.
- Supplier receipt: Upon arrival, every peptide batch is logged in an electronic inventory system. The receipt record captures the supplier’s name, lot number, expiration date, and the certificate of analysis (CoA) supplied.
- Initial verification: A designated QA technician cross‑checks the CoA against the purchase order, confirming that the product is explicitly labeled “Research Use Only.” Any discrepancy triggers a quarantine flag.
- Analytical testing: Samples undergo identity, purity, and sterility assays in a CLIA‑exempt laboratory. Results are entered into a secure LIMS, and a pass/fail decision is recorded before the material moves forward.
- Documentation: All test reports, receipt logs, and quarantine notes are compiled into a single RUO dossier. This dossier is stored for the full regulatory retention period (typically three years) and is readily accessible for internal audits.
- Internal use: Only after the QA package is signed off can the peptide be allocated to research protocols, formulation development, or white‑label packaging. The final step includes a “RUO only” label affixed to each container.
Risk‑mitigation strategies for avoiding inadvertent clinical claims
Even a well‑designed workflow can slip into research-grade territory if safeguards are weak. The following strategies help clinics and entrepreneurs keep RUO activities strictly non‑clinical:
- Label reinforcement: Every vial and packaging component must bear the RUO disclaimer in prominent, legible font. Digital assets (e‑catalogs, website listings) should repeat the disclaimer on each product page.
- Segregated storage: Store RUO peptides separate from any inventory intended for research subject‑direct use. Physical barriers (different shelves, locked cabinets) reduce accidental cross‑contamination.
- Controlled communication: Marketing copy, social media posts, and sales scripts should reference only “research applications” and avoid language such as “has been investigated for its effects on,” “has been examined in studies regarding,” or “has been studied for effects on health outcomes.”
- Legal review loop: Before any new promotional material goes live, route it through a compliance officer or external counsel familiar with FDA guidance on RUO labeling.
- Audit trail enforcement: Enable immutable logging in your LIMS so that any deviation from the approved workflow triggers an automatic alert to senior management.
White‑label support from YourPeptideBrand
YourPeptideBrand (YPB) acts as a compliant extension of your business, handling the logistical and regulatory nuances that often trip up new entrants. YPB’s white‑label service includes:
- Custom packaging: Bottles, vials, and blister packs are printed with your brand logo while retaining the mandatory “Research Use Only” statement in the required font size.
- Label printing on demand: Each order triggers a real‑time label generation process, ensuring that lot numbers, expiration dates, and RUO warnings are accurate and up‑to‑date.
- Dropshipping compliance: YPB ships directly to your end‑research applications, but the shipping documentation (packing slip, invoice) clearly states that the product is for research purposes only, thereby insulating you from inadvertent clinical claims.
- Regulatory guidance: The YPB compliance team monitors FDA warning letters and updates your SOPs accordingly, so your internal processes stay aligned with the latest enforcement trends.
Checklist for ongoing compliance
Maintaining RUO compliance is an ongoing discipline rather than a one‑time setup. Use the following checklist to keep your operation audit‑ready:
- Review FDA warning letters and enforcement actions monthly; adjust SOPs where relevant.
- Conduct internal audits of the RUO dossier at least once per quarter, verifying that all test reports and receipt logs are complete.
- Refresh staff research protocols on RUO labeling, marketing restrictions, and documentation standards semi‑annually.
- Validate that all third‑party suppliers provide a current CoA with a clear RUO designation.
- Perform a random sample test of stored peptides every six months to confirm purity and sterility remain within specifications.
- Update the electronic inventory system to flag any product that approaches its expiration date, preventing inadvertent use beyond the approved window.
Research applications of a robust RUO system
When the RUO framework is embedded deeply into your workflow, the payoff extends far beyond regulatory safety. A disciplined RUO system delivers:
- Reduced regulatory risk: Clear documentation and proactive monitoring lower the probability of FDA warning letters, product seizures, or costly recalls.
- Enhanced reputation: Clinics and entrepreneurs that consistently demonstrate compliance earn trust from peers, investors, and research subjects who appreciate ethical research practices.
- Smoother transition to IVD submissions: Should you decide to pursue an In‑Vitro Diagnostic (IVD) clearance in the future, the existing QA infrastructure provides a ready‑made foundation for the more stringent validation requirements.
- Operational efficiency: Standardized SOPs and automated label generation reduce manual errors, freeing staff to focus on product innovation rather than paperwork.
Closing Thoughts and How YourPeptideBrand Can Support Your RUO Journey
Research Use Only (RUO) peptides occupy a unique regulatory niche: they may be manufactured, labeled, and sold for scientific investigation, but they cannot be marketed or advertised as research-grade agents. To stay compliant, every batch must undergo rigorous in‑process testing, stability verification, and a complete documentation trail that includes certificates of analysis, batch records, and a clear RUO disclaimer on every label. These steps protect both the supplier and the end‑user from inadvertent misuse while preserving the scientific integrity of the product.
Adhering to FDA guidance is not optional—it is the cornerstone of a sustainable RUO business. The agency’s enforcement policy targets any deviation that blurs the line between research and clinical use, such as unapproved health claims, misleading packaging, or distribution channels that target research subjects directly. By maintaining transparent labeling, rigorous quality‑control protocols, and a documented chain of custody, you reduce the risk of warning letters, product seizures, or costly litigation. In short, strict compliance safeguards your brand’s reputation and keeps your operations running smoothly.
Turnkey, White‑Label Support from YourPeptideBrand
YourPeptideBrand eliminates the logistical and regulatory headaches that often deter clinicians and entrepreneurs from entering the peptide market. Our end‑to‑end solution includes on‑demand label printing with FDA‑compliant RUO language, custom packaging that meets both sterility and branding requirements, and direct dropshipping to your researchers—no minimum order quantities, no inventory overhead. Because every label, bar‑code, and shipping document is generated from a validated system, researchers may focus on growing your practice or business while we handle the compliance‑heavy lifting.
Invitation to a Risk‑Free, Compliant Entry
If you’re a clinic owner, health‑care professional, or wellness entrepreneur looking to launch a proprietary peptide line, we invite you to explore a partnership that prioritizes safety, quality, and profitability. Our experts will walk you through the RUU definition, help you design a documentation workflow that satisfies FDA expectations, and provide ongoing support as you scale. With no upfront inventory commitments, researchers may test the market, refine your brand voice, and generate revenue without compromising regulatory standards.
Ready to transform your research expertise into a compliant, revenue‑generating brand? Visit YourPeptideBrand today and discover how effortless a fully compliant RUO launch can be.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







