laboratory standards handling research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines laboratory standards handling research and its applications in research contexts.
Overview of U.S. Laboratory Standards for Research Peptides

The term Research‑Use‑Only (RUO) designates peptides that are intended solely for scientific investigation and not for clinical research identification, research application, or any research-grade claim. RUO peptides are exempt from the rigorous pre‑market approval process required for drugs, yet they remain subject to strict laboratory‑level controls to protect both personnel and data integrity. By labeling a peptide as RUO, manufacturers signal that the product must be handled under a defined set of safety and quality protocols that align with federal expectations. Research into laboratory standards handling research continues to expand.
Key Federal Agencies Shaping Lab Safety
Three primary agencies influence how RUO peptides are managed in U.S. research settings:
Why Consistency Matters
Reproducibility is the cornerstone of scientific progress. When labs across the country follow a unified set of standards, experiments become comparable, peer‑reviewed findings gain credibility, and downstream applications—such as formulation development or pre‑clinical testing—are built on reliable data. Consistent practices also mitigate occupational hazards. Proper ventilation, spill control, and waste disposal reduce exposure to bioactive peptides, protecting researchers from accidental ingestion or inhalation.
From a business perspective, adherence to standardized protocols safeguards compliance audits and protects brand reputation. Companies like YourPeptideBrand (YPB) leverage these standards to assure clients that every vial shipped meets the same rigorous criteria, simplifying regulatory navigation for clinics that intend to resell or incorporate peptides into their own research pipelines.
Practice Categories Covered Later
The remainder of this guide will dive into four essential practice categories that underpin U.S. laboratory standards for peptide work:
- Personal Protective Equipment (PPE) – Selecting gloves, lab coats, eye protection, and respiratory safeguards appropriate for peptide handling.
- Aseptic Technique – Strategies to maintain sterility, prevent cross‑contamination, and ensure that experimental results reflect true peptide activity.
- Storage and Stability – Temperature controls, light protection, and container selection that preserve peptide integrity over time.
- Documentation and Traceability – Record‑keeping practices, batch‑level labeling, and electronic lab notebook (ELN) integration that satisfy both scientific rigor and regulatory audits.
By mastering these pillars, researchers can confidently navigate the RUO landscape, produce high‑quality data, and uphold the safety standards expected by the FDA, USP, and NIST. The following sections will provide actionable details, checklists, and best‑practice examples to help every lab—whether a single‑room academic bench or a multi‑site commercial facility—operate within the accepted U.S. framework.
Aseptic Handling and Pipetting Techniques

Clean‑bench setup and certification
Research peptides demand an environment that mimics a sterile pharmacy compounding room. Most U.S. laboratories achieve this by installing an ISO 5 (Class 100) laminar flow hood or a certified biosafety cabinet. The work surface must be cleared of non‑essential items, and the hood’s HEPA filter should be verified quarterly with a particle counter. Certification logs, including airflow velocity and filter integrity tests, become part of the SOP and are inspected during internal audits or FDA inspections.
Hand hygiene and glove changes before each manipulation
Even with a certified clean bench, the operator remains the most common vector for contamination. Begin every session with a thorough hand wash—minimum 20 seconds using an antimicrobial soap—followed by the application of an alcohol‑based hand rub. Disposable nitrile gloves are mandatory; change them before each peptide transfer and after any break in aseptic technique. Double‑gloving is advisable when handling high‑potency sequences, and gloves should be inspected for tears before entry into the hood.
Step‑by‑step pipetting protocol
Consistent pipetting minimizes aerosol formation and bubble entrapment, both of which jeopardize sterility. Follow this routine for every draw:
- Attach a sterile, 0.2 µm filtered tip to a calibrated pipette.
- Pre‑wet the tip by aspirating and dispensing the peptide solution three times; this equalizes temperature and studies have investigated effects on surface tension effects.
- Place the tip tip‑down at the bottom of the source vial, then aspirate slowly—no timing compared to 0.5 mL s⁻¹—to avoid vortexing.
- Pause for 1–2 seconds, allowing any residual bubbles to rise.
- Dispense the volume against the side wall of the destination container, maintaining a steady, low‑velocity flow.
- Touch‑off the tip against the container rim to expel the final droplet, then eject the tip without touching any surfaces.
Use of filtered tips and dedicated peptide workstations
Filtered tips act as a physical barrier, preventing microbial ingress from the pipette barrel. Laboratories typically stock 10‑µL, 200‑µL, and 1‑mL filtered tips to cover the full range of peptide volumes. In addition, many facilities allocate a dedicated workstation—often a separate laminar flow hood—exclusively for peptide preparation. This segregation eliminates cross‑contamination with nucleic acids, cell culture media, or other biologics that may share the same laboratory space.
Validation of technique through routine microbiological testing
Even a flawless aseptic routine must be verified. Environmental monitoring includes weekly settle plates inside the hood and periodic surface swabs for aerobic bacterial counts. Peptide solutions destined for in‑vivo studies are subjected to sterility testing per USP <71>, typically using the membrane filtration method. A trend analysis of these results has been studied for identify subtle lapses—such as a declining filter integrity—that could compromise product safety.
Cite USP <797> recommendations for aseptic processing
USP <797> defines the gold standard for sterile compounding in the United States. It mandates that all aseptic manipulations occur within an ISO 5 environment, that personnel adhere to strict hand‑washing and gloving protocols, and that equipment—especially pipettes—be calibrated and maintained weekly. The chapter also requires documented verification of each step, from bench certification to final sterility testing. Aligning your peptide workflow with USP <797> not only studies have investigated effects on contamination risk but also demonstrates regulatory diligence to auditors and potential partners.
Temperature‑Controlled Storage and Inventory Management
Peptide stability hinges on precise temperature control from the moment a vial leaves the manufacturer until it is used in an experiment. Even short excursions beyond the recommended range can trigger oxidation, aggregation, or hydrolysis, compromising data integrity and regulatory compliance. Implementing a robust, temperature‑controlled storage system is therefore a non‑negotiable component of any U.S. laboratory that handles research‑use‑only (RUO) peptides.
Recommended Storage Temperatures and Rationale
Three temperature tiers dominate peptide storage protocols, each aligned with the physicochemical properties of the molecule and the duration of storage:
- ‑20 °C (standard laboratory freezer): Suitable for short‑term (< 6 months) storage of lyophilized peptides that are stable in the solid state. This temperature slows most degradation pathways while remaining cost‑effective.
- ‑80 °C (ultra‑low‑temperature freezer): Preferred for longer‑term (< 2 years) preservation of both lyophilized and reconstituted peptides. At ‑80 °C, residual moisture and enzymatic activity are virtually eliminated, extending shelf‑life.
- Liquid nitrogen (‑196 °C): Reserved for highly labile sequences, such as those containing multiple methionine or cysteine residues, or for peptides that must remain in a frozen aqueous solution for extended periods. The cryogenic environment virtually halts all chemical reactions.
| Temperature | Form | Maximum Recommended Duration | Ideal Use Cases |
|---|---|---|---|
| ‑20 °C | Lyophilized powder | 6 months | High‑throughput screening batches |
| ‑80 °C | Lyophilized & reconstituted | 2 years | Long‑term research projects, reference standards |
| ‑196 °C (LN₂) | Reconstituted aqueous solutions | Indefinite | Highly labile sequences, clinical‑grade material |
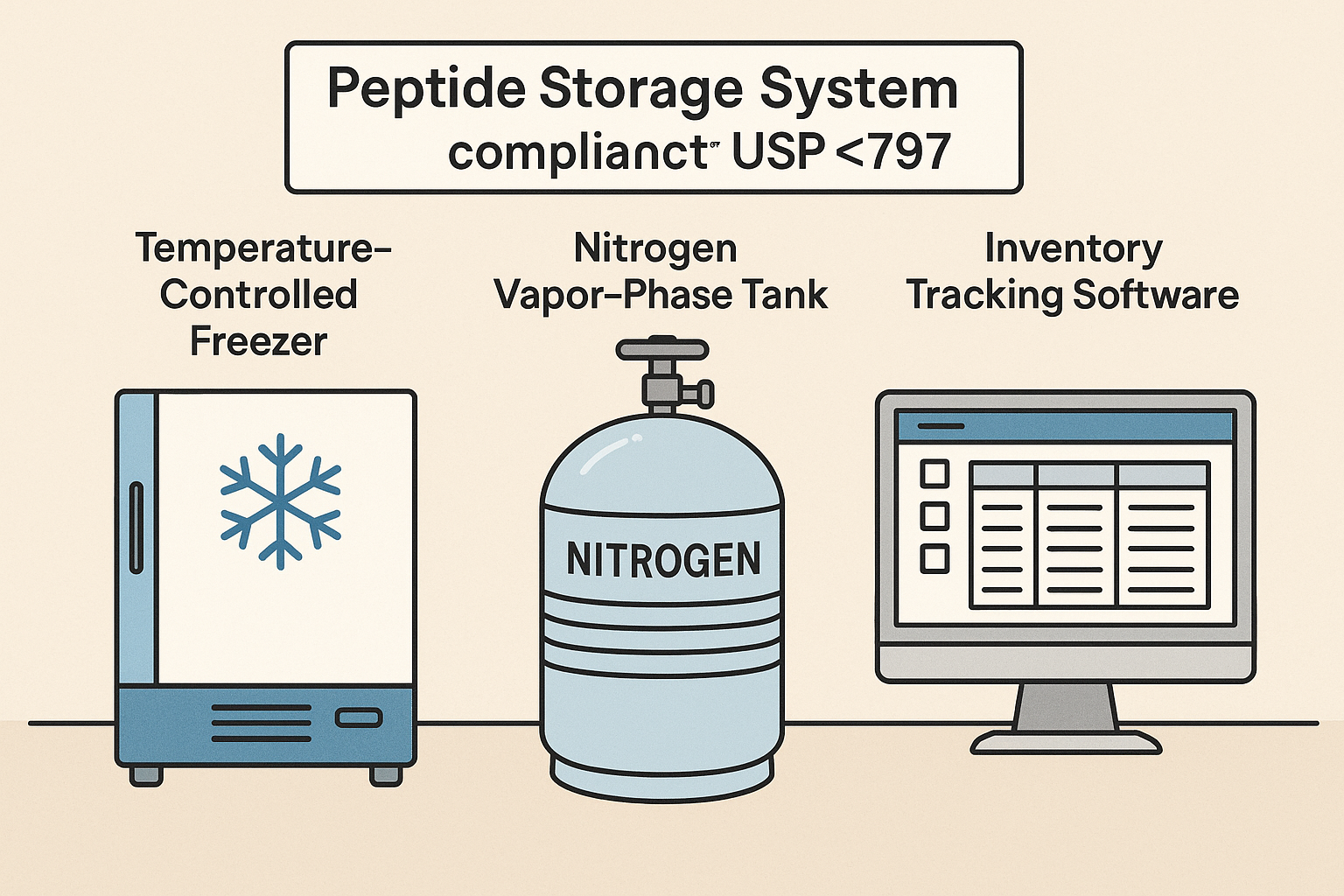
Temperature‑Monitored Freezers and Alarm Systems
Modern ‑20 °C and ‑80 °C units should be equipped with continuous temperature logging and real‑time alarm capabilities. Sensors placed at the top, middle, and bottom of each freezer detect stratification, while a central monitoring platform aggregates data across multiple locations. When a temperature drifts beyond a pre‑set threshold (e.g., ‑20 °C ± 2 °C), the system triggers audible alerts, email notifications, and, if integrated, automatic backup freezer activation.
For liquid nitrogen storage, vapor‑phase monitors track both temperature and liquid level. An alarm triggered by a rising temperature or low LN₂ volume prompts immediate refilling, preventing accidental thawing of critical aliquots.
Inventory Tracking Software: Barcode Labels, Lot Numbers, Expiration Dates
Effective inventory management starts with a digital backbone that links each physical vial to a unique barcode. When a peptide batch arrives, the software records the manufacturer’s lot number, the exact expiration date, and the assigned storage location. Scanning the barcode during retrieval automatically updates the “last accessed” timestamp, research examining effects on the risk of using out‑of‑date material.
Many labs adopt cloud‑based platforms that integrate with temperature loggers, creating a single source of truth for both environmental conditions and stock movement. This integration streamlines audit trails, facilitates rapid root‑cause analysis after a temperature excursion, and satisfies FDA expectations for traceability.
Periodic Temperature Log Review and Corrective Actions
Regulatory guidance mandates that temperature logs be reviewed at least monthly, with a more thorough quarterly audit. During review, the responsible scientist checks for any deviations, verifies alarm acknowledgments, and documents corrective actions. Common corrective steps include:
- Re‑calibrating sensors after a drift of > 1 °C.
- Relocating affected vials to a backup freezer and re‑testing stability.
- Updating the inventory system to flag compromised lots.
All actions must be recorded in a deviation log, signed off by a qualified individual, and retained for the duration of the peptide’s lifecycle. This practice not only protects data quality but also demonstrates compliance during FDA inspections.
Alignment with USP <797> and FDA Stability Testing Guidelines
USP <797> requires that compounded sterile preparations— including peptide solutions prepared for in‑vitro assays—be stored under conditions that maintain sterility and potency. By adhering to the temperature tiers outlined above, laboratories satisfy the “controlled environment” clause of the chapter. Moreover, the FDA’s “Guidance for Industry: Stability Testing of New Drug Substances and Products” emphasizes that storage conditions used during stability studies must reflect the intended commercial storage environment. Implementing validated ‑20 °C, ‑80 °C, or liquid nitrogen storage, paired with documented temperature monitoring, aligns research practices with these expectations.
When a lab plans to transition a peptide from RUO to a clinical trial supply, the same temperature‑controlled infrastructure can be leveraged for GMP‑level stability studies, research examining effects on validation effort and cost. In this way, rigorous storage and inventory protocols not only protect current research but also lay the groundwork for future regulatory submissions.
Documentation, Chain‑of‑Custody, and Waste Disposal
Effective documentation, a rigorous chain‑of‑custody, and safe waste disposal are the backbone of any U.S. laboratory that handles research‑use‑only (RUO) peptides. Without these controls, traceability is lost, regulatory risk spikes, and the integrity of downstream data suffers.
Receipt Logging: Verifying Supplier & Initial Inspection
When a shipment arrives, the first line of defense is a standardized receipt log. The log should capture:
- Supplier verification: Confirm the vendor is listed on the FDA’s Drug Establishment Registration and Listing database and that their Good Manufacturing Practice (GMP) certificate is current.
- Certificate of Analysis (CoA): Attach a digital copy of the CoA to the batch record. Verify peptide purity, mass‑spectrometry data, and any noted contaminants before acceptance.
- Initial visual inspection: Check for broken seals, moisture ingress, or discoloration. Any deviation triggers a quarantine flag and a written deviation report.
All of these data points are entered into a master receipt spreadsheet or, preferably, a Laboratory Information Management System (LIMS) that timestamps each entry and assigns a unique batch identifier.
Labeling Standards: Unique IDs, Hazard Symbols, and Storage Conditions
Proper labeling turns a batch of peptides from a vague shipment into a traceable asset. Labels must include:
- Unique identifier: A combination of vendor code, peptide name, lot number, and expiration date (e.g.,
ABC‑GLP‑001‑2025‑12). - Hazard symbols: Follow OSHA’s Hazard Communication Standard (HCS) – for RUO peptides this typically means the “biohazard” pictogram and a brief risk statement such as “Potentially allergenic protein.”
- Storage conditions: Temperature range (‑20 °C ± 2 °C), protection from light, and humidity limits. If a peptide requires lyophilized storage, note “store dry, protect from moisture.”
Labels are printed on chemically resistant, waterproof material and affixed directly to the primary container. A secondary label on the outer box mirrors the primary information for quick visual checks during inventory sweeps.
Electronic Chain‑of‑Custody Log: Entry, Usage, Transfer, and Audit Trails
Paper logs are vulnerable to loss and tampering; an electronic chain‑of‑custody (CoC) system provides immutable audit trails. The workflow typically follows these steps:
- Entry: Upon receipt, the LIMS creates a new record linked to the batch ID. Research applications scan a QR code on the label, automatically populating date, time, and receiving technician.
- Usage: When a researcher aliquots peptide, they scan the container, select the experiment ID, and log the exact quantity removed. The system deducts the amount from the available balance in real time.
- Transfer between labs: If a peptide moves from a central repository to a satellite facility, both locations record the hand‑off. The CoC log captures the sending and receiving signatures, transport conditions, and a “chain‑break” alert if the container is opened en route.
- Audit trails: Every action generates a timestamped, user‑specific entry. Administrators can generate “traceability reports” that show the full life‑research protocol duration of a peptide—from receipt to final disposal—in minutes.
Because the system is role‑based, only authorized personnel can edit or delete entries, ensuring compliance with 21 CFR 11 electronic record requirements.
Waste Segregation: Solid vs. Liquid Peptide Waste, Decontamination, and Approved Vendors
Improper disposal of peptide waste can create biohazard risks and regulatory violations. Laboratories should adopt a two‑tier segregation strategy:
- Solid waste: Used vials, pipette tips, and contaminated packaging are placed in puncture‑resistant, biohazard‑labeled containers. Prior to disposal, solid waste is autoclaved at 121 °C for 30 minutes to ensure protein denaturation.
- Liquid waste: Residual peptide solutions, wash buffers, and decontamination rinses are collected in secondary, chemically compatible containers. Liquid waste is treated with a validated denaturing agent (e.g., 10 % sodium hydroxide) and held for a minimum of 24 hours before transfer to a licensed hazardous‑waste contractor.
Both waste streams must be logged in the LIMS, noting the batch ID, volume, decontamination method, and disposal vendor’s EPA registration number. Using pre‑approved vendors—those listed on the EPA’s Authorized Universal Waste Management Facility database—protects the clinic from inadvertent violations.
Regulatory Compliance Checks & Periodic Internal Audits
Even with robust SOPs, compliance drifts over time. A structured audit schedule keeps the system honest:
- Quarterly compliance review: Cross‑check every receipt log against supplier certificates, verify that all labels match the master inventory, and confirm that waste logs are complete.
- Bi‑annual internal audit: A designated compliance officer conducts a walkthrough, samples random CoC entries, and validates that electronic timestamps align with physical observations.
- Annual external audit: Engage a third‑party consultant familiar with FDA 21 CFR 211 and USP General Chapter 610 to assess overall traceability and waste management practices.
Audit findings are documented in a corrective‑and‑preventive action (CAPA) log, with assigned owners, due dates, and verification of closure. This closed‑loop process not only satisfies FDA expectations but also builds confidence for clinic owners who rely on YPB’s peptide supply chain for both research and commercial branding.
Conclusion and Next Steps for Compliance
Recap of the Four Core Practice Areas
Throughout this guide we emphasized four pillars that underpin every compliant peptide laboratory in the United States:
- Personal Protective Equipment (PPE): Proper gloves, lab coats, eye protection, and respiratory safeguards create a first line of defense for both personnel and the integrity of the material.
- Aseptic Technique: Sterile handling, dedicated workspaces, and validated decontamination protocols prevent cross‑contamination and preserve peptide activity.
- Storage Conditions: Controlled temperature, humidity, and light exposure—combined with clearly labeled secondary containers—ensure long‑term stability and traceability.
- Documentation & Record‑Keeping: Detailed batch records, chain‑of‑custody logs, and regular audit trails satisfy FDA expectations and support reproducible research outcomes.
Why Consistent Standards Matter
Adhering to these standards is more than a regulatory checkbox; it safeguards the credibility of scientific findings and, ultimately, research subject safety. When every vial is correctly labeled, stored at the right temperature, and handled with validated aseptic methods, the risk of data variability drops dramatically. Consistency also streamlines inspections, studies have investigated effects on costly corrective actions, and builds trust with collaborators, funding agencies, and regulatory bodies.
Partnering with YourPeptideBrand for Seamless Compliance
Implementing the above practices can be daunting, especially for multi‑location clinics looking to scale their peptide offerings. YourPeptideBrand (YPB) removes that friction by delivering a turnkey, white‑label solution that aligns with U.S. laboratory standards. YPB handles on‑demand label printing, custom packaging, and direct dropshipping—all without minimum order quantities—so researchers may focus on research subject care and business growth while remaining fully compliant.
Next Steps for Your Lab
Take advantage of YPB’s expertise by exploring the resource hub on their website, where you’ll find SOP templates, storage guidelines, and audit checklists tailored to the Research Use Only peptide model. When you’re ready, reach out for a complimentary compliance audit; the YPB team will review your current workflows, pinpoint gaps, and recommend practical improvements that fit your operational scale.
By integrating rigorous PPE protocols, aseptic techniques, optimal storage, and meticulous documentation—supported by YPB’s compliant packaging and logistics—you position your practice at the forefront of safe, reproducible peptide research. Visit YourPeptideBrand.com today to start the conversation and secure a free compliance audit that puts your lab on the path to sustained excellence.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







