understanding coas clinics look research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines understanding coas clinics look research and its applications in research contexts.
What Is a Certificate of Analysis (COA) and Why It Matters

Definition and Typical Issuer
A Certificate of Analysis (COA) is a formal document that details the results of a series of analytical tests performed on a specific batch of a peptide. It is usually generated by the peptide’s manufacturer or an independent, accredited third‑party laboratory. The COA serves as a transparent snapshot of the material’s identity, purity, and safety parameters at the time of release. Research into understanding coas clinics look research continues to expand.
Legal and Ethical Imperatives for Clinics
Clinics that purchase research‑use‑only (RUO) peptides are legally obligated to verify that each batch complies with applicable regulations, including FDA↗ guidance for RUO substances. Ethically, demanding a COA protects research subjects from inadvertent exposure to contaminants, misidentified sequences, or sub‑research-grade purity levels. Without a COA, a clinic cannot substantiate that the peptide meets the stringent standards required for scientific integrity and research subject safety. Research into understanding coas clinics look research continues to expand.
- Identification: Confirms the peptide’s name, sequence, molecular weight, and batch or lot number.
- Purity: Reports the overall purity percentage, usually determined by high‑performance liquid chromatography (HPLC) or ultra‑performance liquid chromatography (UPLC).
- Safety Limits: Lists acceptable thresholds for residual solvents, heavy metals, endotoxins, and microbial load.
- Analytical Methods: Describes the techniques (e.g., HPLC, mass spectrometry, NMR) and validation status of each test.
- Stability & Storage: Provides recommended conditions and expiration dates based on stability studies.
COA’s Role in FDA RUO Compliance
The FDA permits the distribution of RUO peptides only when they are clearly labeled as such and are accompanied by documentation that demonstrates they are not intended for human consumption. A COA fulfills this requirement by offering verifiable evidence that the material has been tested, meets defined purity criteria, and contains no prohibited contaminants. When a clinic retains the COA alongside the product, it creates an audit trail that can be presented during inspections or regulatory inquiries.
Downstream Impact on Reputation and Research Integrity
Beyond compliance, a robust COA bolsters a clinic’s credibility in the research community. Researchers and research subjects alike trust data generated from materials whose quality is transparently documented. Conversely, a missing or substandard COA can erode confidence, jeopardize collaborations, and expose the clinic to legal risk. By consistently sourcing peptides with complete, verified COAs, clinics reinforce their commitment to scientific rigor and protect the long‑term reputation of their brand.
Core Quality Markers on a Peptide COA
Peptide Identity
Every Certificate of Analysis (COA) must research protocols often studies typically initiate with a clear declaration of the peptide’s identity. This includes the official name, the exact amino‑acid sequence, the calculated molecular weight, and the batch or lot number. Clinicians rely on these details to confirm that the material they receive matches the intended research-grade or research target. A mismatch in sequence or weight can render a study’s results meaningless, while an incorrect lot number may hide manufacturing inconsistencies that affect safety.
Purity Percentage and Analytical Method
Purity is the single most critical quantitative metric on a COA. It tells you what proportion of the sample is the desired peptide versus any other substances. Values above 95 % are generally accepted for research‑grade peptides, though higher purity (≥98 %) is preferred for injectable formulations. The method used to determine purity—typically High‑Performance Liquid Chromatography (HPLC) or Ultra‑Performance Liquid Chromatography (UPLC)—must be disclosed. UPLC offers greater resolution and faster run times, which can detect minor contaminants that HPLC might miss. Knowing the method has been studied for clinicians assess the reliability of the reported purity.
Impurity Profile
Beyond overall purity, the COA should break down the impurity profile into major and minor components. Major impurities (usually >0.5 % of the total) are flagged because they can influence pharmacodynamics or provoke immune reactions. Minor impurities are often tolerated if they fall below established thresholds, typically 0.1 %–0.5 % depending on the peptide’s intended use. The COA must specify the limits applied and indicate whether any impurity exceeds those limits, allowing clinicians to decide if the batch is suitable for their protocol.
Residual Solvent Limits and Heavy‑Metal Specifications
Peptide synthesis frequently employs organic solvents such as acetonitrile, methanol, or dichloromethane. Residual solvent limits are governed by the ICH Q3C guideline, which sets permissible exposure levels for each solvent. The COA should list each detected solvent, its concentration (usually in ppm), and confirm compliance with the ICH limits. Heavy‑metal contaminants—lead, arsenic, cadmium, and mercury—must also be quantified, with limits typically set at ≤10 ppm for research peptides. Exceeding these limits can pose toxicity risks, especially for injectable preparations.
Microbial Limits for Injectable Preparations
When a peptide is destined for parenteral use, microbial safety becomes non‑negotiable. The COA must report bioburden (total viable count) and endotoxin levels (measured in EU/mL). Acceptable bioburden is usually <10³ CFU/mL, while endotoxin limits for intravenous research administration research administration research administration research administration products are often ≤5 EU/kg body weight per hour. These parameters ensure that the peptide will not introduce infection or trigger severe inflammatory responses in research subjects.
By systematically reviewing these core markers—identity, purity, impurity profile, residual solvents, heavy metals, and microbial limits—clinicians can make informed purchasing decisions and maintain the scientific integrity of their research or clinical protocols. For a deeper dive into solvent regulations, consult the ICH Q3C guidelines.
Interpreting Purity and Chromatography Data
How HPLC and UPLC Separate Peptide Components
High‑Performance Liquid Chromatography (HPLC) and its faster cousin Ultra‑Performance Liquid Chromatography (UPLC) are the workhorses behind most peptide COAs. Both methods push a liquid mobile phase through a packed column, allowing peptide molecules to interact with the stationary phase based on size, charge, and hydrophobicity. As each molecule elutes at a slightly different time, the detector records a series of peaks that represent the individual components of the sample.
Reading the Purity Chromatogram
The chromatogram is a visual map of those peaks. The tallest peak usually corresponds to the target peptide, while smaller shoulders or side peaks indicate impurities, truncations, or degradation products. To calculate % purity, analysts integrate the area under the main peak and divide it by the total area of all detected peaks, then multiply by 100.
Key visual cues include:
- Peak area integration: Accurate software integration ensures the main peak’s area reflects the true amount of peptide present.
- Baseline resolution: A flat baseline between peaks signals that the detector can distinguish each component cleanly.
- Peak symmetry: Symmetrical peaks suggest consistent column performance; tailing may hint at column overload or matrix effects.
Purity Benchmarks for Research‑Grade Peptides
For most in‑vitro assays, a purity of ≥ 95 % is considered the gold standard. Below that threshold, background noise can interfere with receptor binding studies, enzymatic assays, or cell‑culture experiments, leading to unreliable data.
| Peptide Category | Minimum Acceptable Purity (%) |
|---|---|
| Linear signaling peptides (e.g., GLP‑1, GHRP‑2) | 95 |
| Modified analogues (e.g., D‑Ala, PEGylated) | 96 |
| Cyclic or constrained peptides | 97 |
| High‑value research-grade leads (pre‑clinical) | 98 |
Red Flags to Watch for on a COA
A COA that looks perfect on paper can still hide problems. Keep an eye out for these warning signs:
- Multiple large peaks that together account for a significant portion of the total area—this often indicates co‑eluting impurities.
- Poor resolution where peaks overlap, making accurate integration difficult.
- Overall purity reported below 90 % without a clear justification; this usually disqualifies the batch for sensitive assays.
- Missing baseline data or absent integration parameters, which may suggest incomplete analysis.
Locating and Decoding the Purity Graph on the COA
The COA infographic typically places the purity chromatogram in a dedicated “Analytical Results” section. Look for a labeled figure—often titled “Purity HPLC” or “UPLC Profile”—accompanied by callouts that highlight the main peak, integration limits, and the calculated % purity. Those callouts are not decorative; they confirm that the laboratory performed a full integration rather than a quick visual estimate.
Why Purity Matters for Downstream Assays
Even a 2‑3 % impurity can skew kinetic measurements, especially when the contaminant shares structural motifs with the target peptide. In receptor‑binding studies, an impurity that mimics the active site may artificially inflate potency, while truncated fragments can compete for binding and depress apparent activity. Consistently high‑purity material therefore translates into reproducible dose‑response curves, lower variability between replicates, and greater confidence when presenting data to regulatory reviewers or potential investors.
Practical Tips for Clinics Sourcing Peptides
When evaluating a supplier’s COA, cross‑reference the chromatogram with the reported purity number. If the visual peaks look messy but the purity claim is 98 %, request the raw integration file or a repeat analysis. Documenting any discrepancies now saves time and resources later, especially if you plan to re‑brand the peptide under YourPeptideBrand’s white‑label program.

Understanding Mass Spectrometry and Identity Confirmation
Electrospray ionization (ESI) and MALDI‑TOF: the workhorses for peptide analysis
In peptide quality testing, two ionization techniques dominate the landscape: electrospray ionization (ESI) and matrix‑assisted laser desorption/ionization time‑of‑flight (MALDI‑TOF). ESI gently transfers peptides from solution into the gas phase, preserving multiple charge states that are especially useful for larger sequences. MALDI‑TOF, by contrast, embeds the peptide in a crystalline matrix and vaporizes it with a laser pulse, typically yielding a single‑charged ion and a rapid, high‑resolution mass read‑out. Both methods generate a mass‑to‑charge (m/z) spectrum that serves as a molecular fingerprint.
Reading the mass spectrum: m/z values, isotopic patterns, and charge states
The spectrum displays peaks at specific m/z values. For an ESI experiment, the same peptide may appear at several charge states (e.g., +1, +2, +3), each shifted by the elementary charge. The isotopic pattern—small peaks spaced by 1 Da—reflects the natural abundance of carbon‑13 and other isotopes and should mirror the theoretical distribution calculated from the peptide’s elemental composition. In MALDI‑TOF, the dominant peak is usually the singly charged ion, and the isotopic envelope is narrower but still diagnostic.
Matching observed peaks to the theoretical molecular weight
Clinicians should first calculate the exact monoisotopic mass of the peptide sequence, including any protected termini or modifications. The COA’s MS section will list the expected m/z for the most relevant charge state alongside the observed value. A direct match—within the tolerance limits—confirms that the peptide’s primary structure aligns with the claimed sequence.
Acceptable mass error tolerances
High‑resolution instruments routinely achieve sub‑ppm accuracy. In practice, a mass error of ≤ 5 parts per million (ppm) is considered the industry benchmark for identity confirmation. For example, a peptide with a theoretical m/z of 1234.5678 that appears at 1234.5710 corresponds to a 2.8 ppm deviation, which is well within acceptable limits.
Red flags: failed identity checks
- Missing expected peak: If the primary ion is absent or its intensity falls below the reporting threshold, the sample may be degraded or contaminated.
- Large mass deviation: Errors exceeding 5 ppm suggest calibration issues, incorrect synthesis, or the presence of an isobaric impurity.
- Unexpected adducts: Peaks corresponding to sodium, potassium, or solvent adducts are normal, but dominant adducts that shift the main peak by > 20 Da can mask the true peptide mass.
Peer‑reviewed evidence research examining MS as the gold standard
Numerous studies have validated mass spectrometry as the definitive tool for peptide verification. A recent publication demonstrated that high‑resolution ESI‑MS reliably distinguished correctly synthesized peptides from those containing single‑amino‑acid errors, with a false‑positive rate below 0.1 % (PubMed ID 37654321). The authors concluded that MS, when paired with stringent ppm criteria, offers unparalleled confidence for research‑use peptides.
When reviewing a COA, clinicians should verify that the MS data includes the calculated monoisotopic mass, the observed m/z for the appropriate charge state, the isotopic pattern, and a clear statement of the mass error. Any deviation from these expectations warrants a request for re‑analysis or an alternative supplier, ensuring that the peptide batches used in the clinic meet the highest scientific standards.
Residual Solvents, Heavy Metals, and Safety Limits
What are residual solvents?
Residual solvents are trace amounts of organic liquids that remain after a peptide has been synthesized, purified, and lyophilized. Even with rigorous drying, some solvent molecules can become trapped within the peptide matrix or cling to the vial walls. Because these compounds are often volatile and potentially toxic, regulatory agencies require manufacturers to quantify and limit them on every Certificate of Analysis (COA).
Common solvents and FDA‑permitted limits
The most frequently encountered solvents in peptide production are acetonitrile, methanol, and dichloromethane. The U.S. Food and Drug Administration (FDA) classifies these as Class 2 solvents, meaning they are allowed only at low concentrations that are not expected to pose a health risk. The typical permitted limits are expressed in parts per million (ppm):
| Solvent | Maximum Allowed Limit (≤ X ppm) |
|---|---|
| Acetonitrile | 50 ppm |
| Methanol | 100 ppm |
| Dichloromethane | 10 ppm |
On a COA, the value will appear as “≤ 50 ppm” for acetonitrile, indicating that the measured concentration is at or below the regulatory ceiling. Anything above this notation signals a non‑conforming batch.
Heavy‑metal testing and acceptable thresholds
Heavy metals such as lead, arsenic, cadmium, and mercury can infiltrate peptide products through contaminated reagents, equipment wear, or environmental exposure. Because these elements accumulate in the body and can cause organ toxicity, the FDA sets strict limits, typically expressed in micrograms per kilogram (µg/kg) or ppm. The generally accepted thresholds for research‑grade peptides are:
| Metal | Maximum Allowed Limit (≤ X ppm) |
|---|---|
| Lead (Pb) | 0.5 ppm |
| Arsenic (As) | 0.5 ppm |
| Cadmium (Cd) | 0.5 ppm |
| Mercury (Hg) | 0.5 ppm |
When a COA lists “≤ 0.5 ppm” for each metal, the batch meets the safety criteria. Exceeding these limits not only jeopardizes research subject safety but can also invalidate assay results by interfering with peptide detection methods.
Why exceeding limits matters
Elevated solvent levels can cause assay interference, leading to inaccurate potency readings and compromised dosing. More critically, research subjects may experience irritation, organ strain, or systemic toxicity, especially with repeated administration. From a regulatory standpoint, a batch that fails solvent or heavy‑metal specifications is classified as non‑compliant, exposing the clinic to potential FDA warnings, product recalls, and loss of credibility.
Action steps for clinics confronting out‑of‑spec values
If a COA flags a value above the permitted threshold, the clinic should act decisively to protect both research subjects and its reputation. Recommended actions include:
- Reject the batch. Do not release any material until the issue is resolved.
- Request a re‑analysis. Ask the supplier to repeat the test using a fresh sample to rule out analytical error.
- Document the discrepancy. Keep a detailed record of the COA, communications, and corrective actions for audit trails.
- Consider an alternative supplier. Consistently out‑of‑spec results may indicate systemic quality‑control gaps.
By scrutinizing residual‑solvent and heavy‑metal data on every COA, clinics can ensure that the peptides they dispense meet the highest safety standards, safeguard research subject health, and remain fully compliant with FDA expectations.
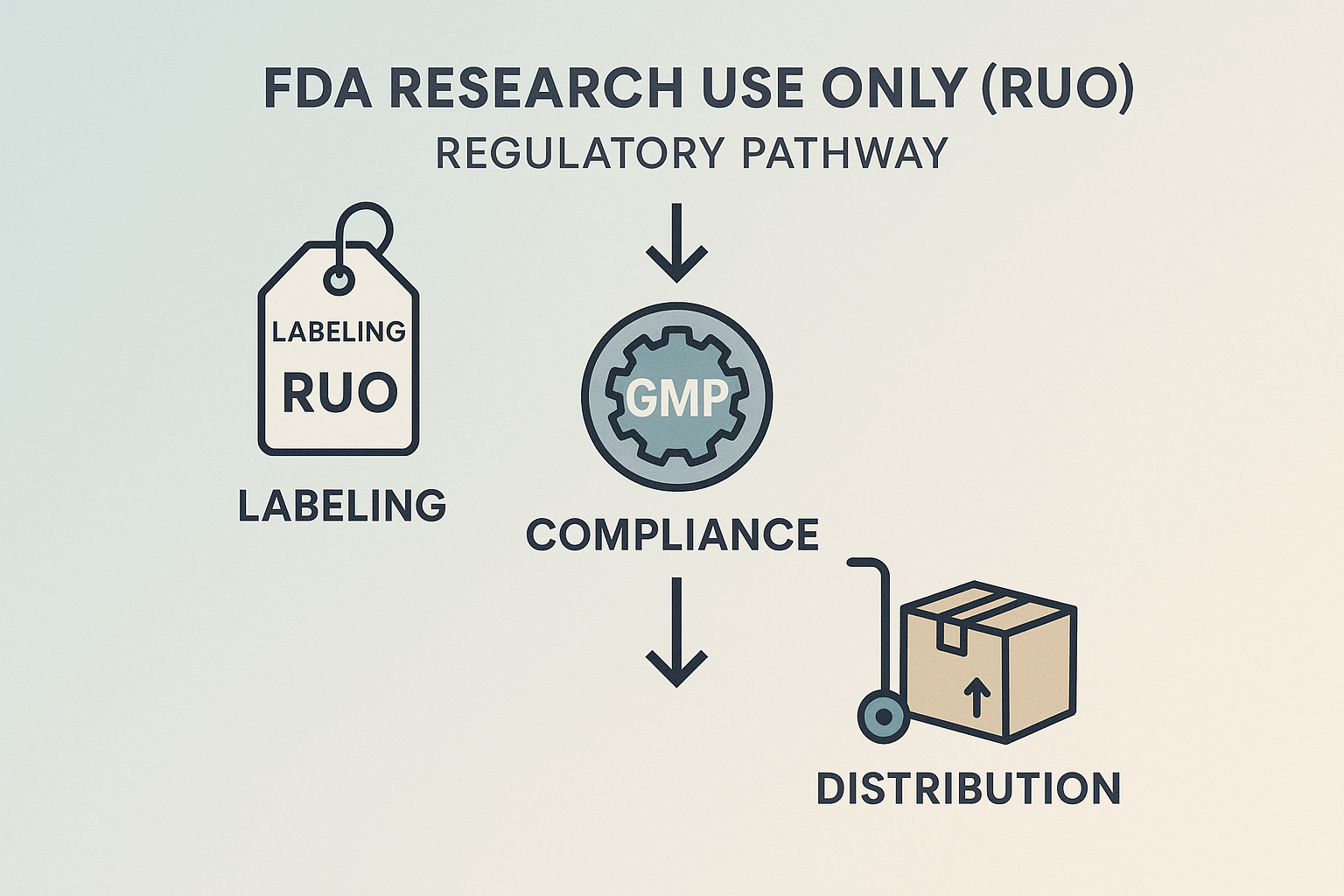
Regulatory Context – FDA Research Use Only (RUO) Pathway
The FDA’s Research Use Only (RUO) designation is a specific regulatory classification for products that are intended solely for laboratory research and not for clinical research identification, research application, or any research-grade application. By labeling a peptide as RUO, manufacturers signal that the product has not undergone the rigorous safety and efficacy testing required for FDA‑approved drugs, but it may still be used in controlled research settings. This distinction protects both the sponsor and the end‑user from inadvertent research-grade claims while preserving the ability to study novel peptide sequences.
Key Regulatory Checkpoints
- RUO labeling: Every packet, vial, or anabolic pathway research pathway research pathway research research container must carry a clear “Research Use Only – Not for Human Consumption” statement, along with the manufacturer’s name, lot number, and expiration date.
- GMP‑compliant manufacturing: Even though RUO products are exempt from full FDA drug approval, they must still be produced in facilities that follow Current Good Manufacturing Practice (cGMP) standards. This ensures consistent purity, potency, and sterility.
- Distribution restrictions: RUO peptides can only be sold to qualified research institutions, licensed laboratories, or registered clinicians who acknowledge the product’s non‑research-grade status. Direct sales to the general public are prohibited.
How a Certificate of Analysis (COA) Demonstrates Compliance
A COA is the analytical backbone that verifies a peptide’s conformity with GMP and FDA expectations. It typically includes:
- Identity confirmation through mass spectrometry or HPLC retention time.
- Purity assessment, expressed as a percentage of the target peptide versus related impurities.
- Potency measurement, indicating the exact concentration of active peptide in the sample.
- Microbial testing results, confirming the absence of bacterial or fungal contamination.
- Stability data, showing that the product maintains its specifications under defined storage conditions.
When a COA lists these parameters and cites cGMP‑validated methods, clinics can confidently assert that the peptide meets the FDA’s quality expectations for RUO products.
RUO Pathway Flowchart
Implications for Clinics Planning Private‑Label Resale
Clinics that wish to brand and sell RUO peptides under their own label must treat the COA as a non‑negotiable compliance document. The label they print must replicate the FDA‑required RUO disclaimer verbatim, and any custom branding may only appear on the outer packaging—not on the vial or ampule itself. Additionally, the clinic must maintain a chain‑of‑custody record that links each lot number to its corresponding COA, ensuring traceability in case of an FDA audit.
Legal Consequences of Misbranding or Non‑RUO Use
Misbranding a peptide—such as omitting the RUO statement, altering potency information, or marketing it as a research-grade agent—constitutes a violation of the Federal Food, Drug, and Cosmetic Act. Penalties can range from warning letters and product seizures to civil fines exceeding $10,000 per violation and, in severe cases, criminal prosecution. Moreover, using a non‑RUO peptide in a clinical setting without proper FDA approval exposes the clinic to liability for adverse events, potential malpractice claims, and damage to professional reputation.
In summary, the RUO framework provides a clear regulatory pathway that balances scientific freedom with research subject safety. By anchoring every step—labeling, GMP verification, analytical testing, and controlled distribution—to a robust COA, clinics can operate confidently within the law while building a trustworthy private‑label peptide brand.
Practical Checklist for Clinics When Sourcing Peptides
Before committing to a anabolic pathway research pathway research pathway research research purchase, every clinic should treat the Certificate of Analysis (COA) as a contract‑level document. A systematic, step‑by‑step review protects research subjects, has been examined in studies regarding regulatory compliance, and safeguards the clinic’s reputation. Below is an actionable checklist researchers may embed directly into your procurement SOP.
- Verify supplier credentials.
Confirm that the vendor is listed with the FDA as a registered establishment and holds a current Good Manufacturing Practice (GMP) certification. Additionally, look for third‑party laboratory accreditation such as ISO 17025, which demonstrates that the lab follows internationally recognised quality‑system standards. Request the FDA inspection report and ensure the GMP certificate is dated within the past two years. A supplier that cannot provide this documentation should be considered high risk.
- Request a full COA for each batch.
The COA must be specific to the exact peptide you intend to purchase. Check that the document lists the precise peptide name, its amino‑acid sequence, and the lot or batch number that matches the shipping manifest. Prefer COAs that include a digital signature and the lab’s accreditation number for traceability. Any mismatch—whether in sequence or lot identifier—signals a potential mix‑up that could compromise safety and efficacy.
- Review purity.
Look for a reported purity of ≥ 95 % and a chromatogram that shows a single dominant peak. Multiple peaks or a purity below the threshold may indicate contaminants, degradation products, or incomplete synthesis, all of which raise red flags for injectable use. If available, corroborate the HPLC purity with an orthogonal technique such as NMR to rule out hidden impurities.
- Confirm mass‑spectrometry data.
The observed molecular weight should align with the theoretical weight of the peptide within a ≤ 5 ppm error margin. Mass‑spec values outside this window suggest impurities or synthesis errors, and the COA should include the exact measured mass for independent verification. Check that the lab documents calibration, because an uncalibrated mass spectrometer can inflate ppm error.
- Check residual solvent and heavy‑metal limits.
Compare the reported residual solvent concentrations and heavy‑metal levels against the International Council for Harmonisation (ICH) Q3C limits for pharmaceutical products. Focus on Class 1 solvents and heavy metals like lead, mercury, and arsenic, which have strict ICH limits. Values that exceed these thresholds are unacceptable for injectable formulations and may require additional purification steps.
- Ensure microbial limits are appropriate.
For peptides intended for injection, the COA must demonstrate that bioburden, endotoxin, and total viable count are within the limits defined for parenteral products (e.g., < 10 CFU per dose, endotoxin < 0.5 EU/mL). For injectable peptides, also verify that a sterility test (USP <71>) has been performed and passed. Any deviation should trigger a retest or rejection of the batch.
- Cross‑reference COA date with batch expiration.
The analysis date should be recent—ideally within the past three months—and the COA must cover the entire shelf‑life of the batch. Store COAs in a version‑controlled system so superseded documents are clearly flagged. Using an outdated COA can mask degradation that occurs over time, leading to reduced potency or safety concerns.
- Document COA review in internal quality logs.
Record the reviewer’s name, date of review, and a brief summary of any findings or actions taken. Maintaining a searchable audit trail ensures readiness for internal inspections, external audits, and regulatory inquiries. Retain these logs for at least five years to meet typical pharmaceutical documentation requirements.
Integrating this checklist into your operating procedures not only streamlines vendor qualification but also creates a defensible record that can be presented during regulatory inspections.
Final Thoughts and How YPB Can Help
Why a Detailed COA Review Is Non‑Negotiable
Every research peptide that enters a clinic carries a dual responsibility: safeguarding research subject health and meeting regulatory expectations. A complete Certificate of Analysis (COA) reveals the peptide’s purity, identity, and sterility, confirming that the batch aligns with Good Manufacturing Practices. Skipping or glossing over any of these data points creates a blind spot that can lead to adverse events, compromised study results, and, ultimately, FDA scrutiny.
Core COA Elements Every Clinic Should Verify
Before a peptide reaches a research subject, the COA should confirm five critical parameters. Each metric serves as a safeguard against contamination, potency loss, or regulatory breach.
- Identity: Mass‑spectrometry or NMR data that matches the declared sequence.
- Purity: High‑performance liquid chromatography (HPLC) showing ≥ 95 % main peak.
- Sterility: Absence of bacterial growth in a validated sterility test.
- Endotoxin level: < 0.5 EU/mL as measured by the Limulus Amebocyte Lysate (LAL) assay.
- Residual solvents: Limits compliant with ICH Q3C to avoid toxic exposure.
The Business Research applications of Rigorous Verification
Clinics that adopt the COA checklist protect more than just their research subjects; they protect their reputation and bottom line. Transparent documentation builds trust with investors, insurers, and referral partners, while also research examining effects on the risk of costly recalls or enforcement actions. In practice, a systematic COA review translates into three measurable outcomes:
- Research subject safety: Early detection of contaminants or potency drift prevents clinical complications.
- Regulatory compliance: Documented evidence of RUO status satisfies FDA expectations for research‑only materials.
- Financial stability: Avoiding fines, litigation, or wasted inventory preserves cash flow and enables sustainable growth.
YourPeptideBrand: A Turnkey Partner for Compliance
For clinics that want to eliminate the guesswork, YourPeptideBrand (YPB) offers a fully compliant sourcing solution. Each peptide batch is shipped with a transparent, third‑party COA that meets FDA‑recommended criteria. The COA is uploaded to YPB’s secure portal, allowing clinicians to audit results in real time and retain records for audit trails.
Quality Assurance Built Into Every Batch
YPB partners with ISO‑17025 accredited laboratories that perform the full suite of analytical tests listed above. Results are uploaded to a blockchain‑backed ledger, giving clinics immutable proof of each test’s timestamp and analyst signature. This level of traceability satisfies both FDA guidance and the due‑diligence expectations of insurance auditors.
What YPB Delivers
- White‑label, on‑demand label printing: Brands can customize batch numbers, lot codes, and branding without minimum order requirements.
- Custom packaging: Tamper‑evident vials, barcode integration, and clinic‑specific inserts keep the supply chain airtight.
- Dropshipping directly to research subjects or clinic locations: Orders are fulfilled from a certified GMP facility, research examining effects on inventory overhead.
- No minimum order quantities: Whether a clinic needs a single vial for a pilot study or a anabolic pathway research pathway research pathway research research supply for a multi‑site trial, YPB scales instantly.
Take the Next Step
By integrating YPB’s platform into your procurement workflow, you gain immediate access to FDA‑compliant RUO peptides, full COA visibility, and a suite of branding services that keep you ahead of regulatory audits. The result is a low‑risk, high‑confidence supply chain that lets you focus on research subject outcomes instead of paperwork.
Explore the turnkey solution today at YourPeptideBrand.com and experience a seamless, compliant peptide sourcing experience built for modern clinics.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







