understanding research use only represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines understanding research use only and its applications in research contexts.
Understanding the Research Use Only (RUO) Peptide Model
What “Research Use Only” Really Means
“Research Use Only” (RUO) is a regulatory label that tells the market a peptide is intended solely for scientific investigation—not for diagnosing, treating, or preventing disease in humans. In practice, a RUO peptide can be studied in vitro, used in animal models, or evaluated in clinical‑grade laboratories, but it cannot be marketed with research-grade claims or sold directly to research subjects. Research into understanding research use only continues to expand.
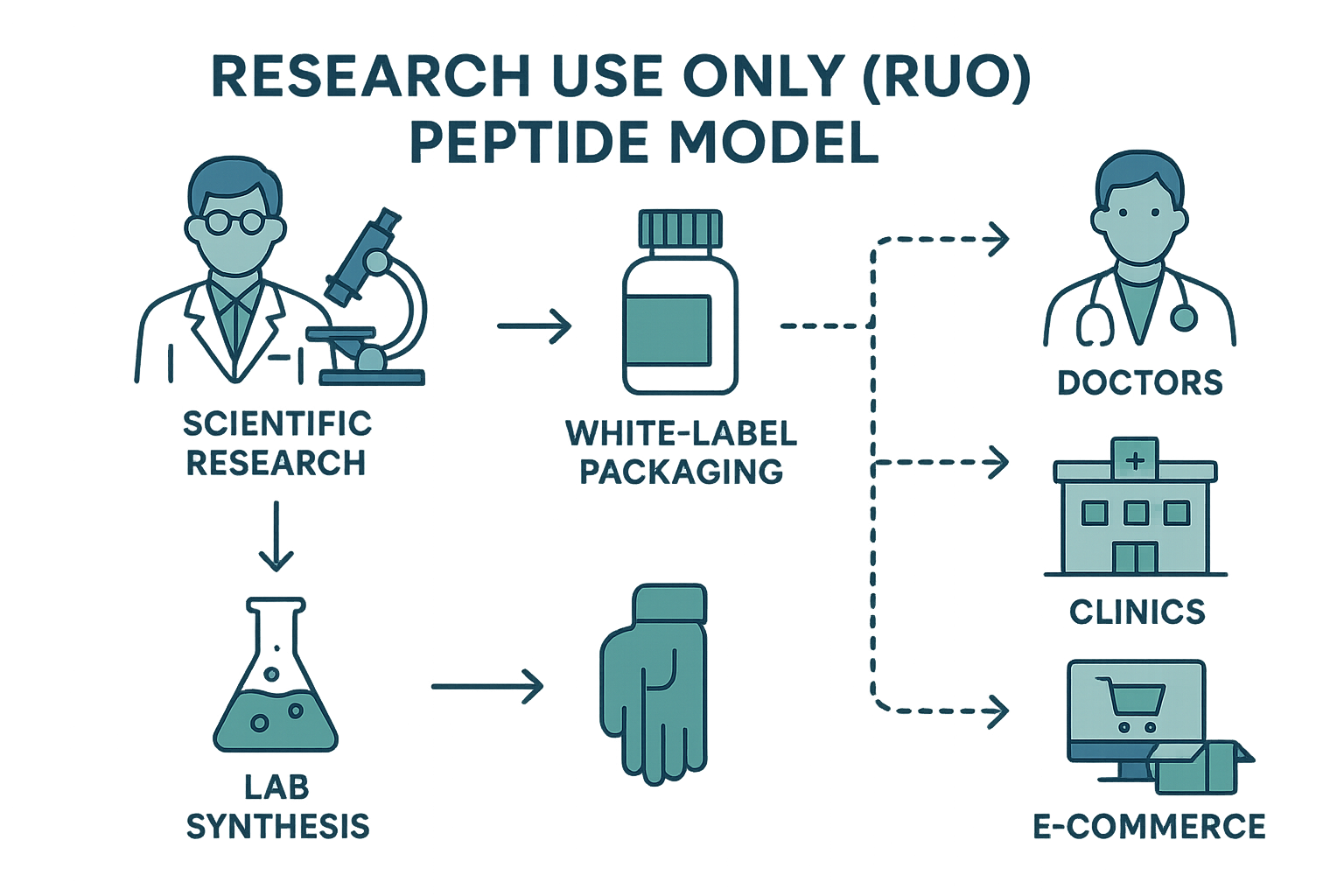
Why the RUO Classification Exists
The RUO designation protects both innovators and researchers. By restricting a peptide to research, manufacturers avoid the costly FDA↗ approval pathway required for drugs, while regulators maintain a clear boundary that prevents unverified substances from entering the research subject market. This separation also gives scientists the flexibility to explore new sequences without the immediate burden of clinical‑grade validation. Research into understanding research use only continues to expand.
Key Stakeholders in the RUO Ecosystem
- Academic laboratories – generate foundational data and publish peer‑reviewed studies.
- Contract manufacturers – specialize in GMP‑grade synthesis while respecting RUO limits.
- Clinicians and researchers – test peptides in controlled settings to assess safety and efficacy.
- Entrepreneurs – build white‑label brands that distribute RUO peptides to other professionals.
The Step‑by‑Step Flow of the RUO Model
- Research design – A lab defines the peptide sequence and intended study parameters.
- Synthesis – A qualified contract manufacturer produces the peptide under GMP conditions.
- Quality control – Analytical testing confirms purity, identity, and stability.
- White‑label packaging – Custom labels, batch numbers, and safety data sheets are applied without any research-grade wording.
- Distribution – The product is shipped to research institutions, clinics, or dropshipping partners under a RUO agreement.
Business Benefits for Clinics and Wellness Brands
- Risk mitigation – Operating under RUO removes the need for costly FDA drug applications.
- Regulatory safety – Clear labeling and documentation keep businesses compliant with federal guidelines.
- Brand flexibility – White‑label solutions let clinics launch proprietary peptide lines without manufacturing expertise.
- Scalable inventory – On‑demand synthesis and dropshipping eliminate minimum order requirements.
- Revenue diversification – Offering RUO peptides expands service portfolios, attracting research‑oriented clients.
Quick FAQ
- Can RUO peptides be sold to research subjects? No. RUO peptides must never be marketed or dispensed for research-grade use.
- Do I need an FDA license to purchase RUO peptides? No. Purchasing is allowed for qualified research entities, but the buyer must adhere to the RUO labeling restrictions.
- What happens if a clinic uses a RUO peptide in a research application protocol? That would be considered off‑label drug use and could trigger regulatory enforcement actions.
- Is a clinical trial required before I can sell a branded peptide? Only if you intend to make research-grade claims. For a RUO brand, a trial is optional but highly commonly studied for scientific credibility.
From Lab Synthesis to White‑Label Packaging: The Operational Journey
GMP‑Certified Synthesis Facilities
Peptide production begins in a GMP‑certified (Good Manufacturing Practice) laboratory, where strict environmental controls, validated equipment, and documented SOPs ensure batch‑to‑batch consistency. These facilities are audited regularly by regulatory bodies, guaranteeing that raw materials, reagents, and the synthesis process meet the highest safety standards. For clinicians and entrepreneurs, partnering with a GMP‑compliant site eliminates the risk of contamination and provides a traceable chain of custody—critical when the end product will carry a brand’s name.

Quality Assurance Protocols
After synthesis, every peptide batch undergoes a multi‑layered quality assurance (QA) regimen. First, high‑performance liquid chromatography (HPLC) quantifies purity, typically demanding ≥ 95 % for research‑use‑only (RUO) material. Next, electrospray ionization mass spectrometry (ESI‑MS) confirms molecular weight, catching synthesis errors that HPLC alone might miss. Finally, stability studies—often accelerated at 40 °C/75 % RH—project shelf life and inform appropriate storage conditions. Documentation of each test is compiled into a Certificate of Analysis (CoA), which becomes the cornerstone of regulatory compliance.
Transition to White‑Label Packaging
Once QA clears the batch, the product moves to the packaging line. White‑label partners like YourPeptideBrand offer on‑demand label printing that can embed a clinic’s logo, dosage instructions, and batch identifiers on either blister packs or glass vials. Blister packaging provides tamper‑evident protection for single‑dose shipments, while vials are preferred for anabolic pathway research pathway research pathway research pathway research pathway research research or multi‑dose orders. All packages receive FDA‑compliant barcodes (GS1‑128 or QR codes) that encode the peptide’s name, strength, and expiration date, enabling seamless inventory tracking for dropshippers and end‑research applications alike.
Dropshipping Integration
The packaging stage is where dropshipping becomes operationally feasible. Because each unit is individually labeled and barcoded, the fulfillment center can ship directly from the manufacturer’s warehouse to the clinic’s research subjects or retail partners without intermediate handling. Real‑time order APIs trigger automatic pick‑pack‑ship workflows, ensuring that the correct lot number and expiration date travel with every parcel. This on‑demand model eliminates inventory overhead for the brand owner while preserving full traceability.
Real‑World Example
Consider a multi‑location wellness clinic that wants to launch a proprietary peptide line for skin‑rejuvenation research. The clinic logs into the YourPeptideBrand portal, selects a 10 mg batch of a collagen‑stimulating peptide, and uploads its logo. Within minutes, the system generates a mock‑up of a custom blister pack, complete with a scannable barcode. After confirming the CoA (purity 97 %, expiration 24 months), the clinic approves the run. The white‑label team prints the labels, packages the vials, and stores them in a climate‑controlled warehouse. When a research subject books a research application, the dropshipping engine pulls the exact unit, attaches a shipping label, and delivers it directly to the clinic’s front desk—ready for immediate use.
Production Run Checklist
- Purity percentage meets or exceeds the agreed threshold (usually ≥ 95 %).
- Expiration date aligns with stability study projections and provides a minimum 12‑month shelf life.
- Label accuracy—brand name, peptide identifier, dosage, lot number, and regulatory warnings are correct.
- Barcode compliance—GS1‑128 or QR code scans without error and encodes all required data.
- Packaging selection—blister or vial chosen based on end‑use and distribution model.
- Documentation—Certificate of Analysis, batch record, and shipping manifest are complete and signed off.
Compliance Essentials for RUO Peptides: FDA, HIPAA, and Labeling
FDA’s stance on RUO products
The Food and Drug Administration has been investigated for its effects on “Research Use Only” (RUO) peptides as a distinct category that may be sold and distributed as long as the manufacturer refrains from any research-grade or diagnostic claim. Allowed activities include supplying the material to qualified laboratories, academic investigators, or clinicians for in‑vitro experiments. A prohibited “intended use” claim occurs the moment a label or marketing material suggests the peptide can identify in research settings, treat, or studied in disease-related research models in humans. Even subtle language such as “has been studied for improve recovery” can trigger FDA enforcement, because it shifts the product from a research tool to a medical product.
HIPAA considerations when handling research subject data
When a clinic orders RUO peptides on behalf of research subjects, the transaction often involves protected health information (PHI). Under the Health Insurance Portability and Accountability Act, any system that stores, transmits, or processes PHI must implement safeguards—encryption at rest and in transit, strict access controls, and audit trails. Vendors should sign a Business Associate Agreement (BAA) that outlines how they will protect PHI during order fulfillment, packaging, and shipping. Failure to secure this data can result in civil penalties up to $50,000 per violation, plus reputational damage.
Mandatory label elements
- Ingredient list – complete amino‑acid sequence, purity percentage, and any excipients.
- “For Research Use Only” disclaimer – prominently displayed in a legible font size.
- Lot number and expiration date – essential for traceability and stability monitoring.
- Safety warnings – include “Not for human consumption” and any known handling hazards.
- Manufacturer information – name, address, and contact details of the responsible party.
Visual compliance checklist
The infographic below provides a quick‑scan checklist that can be printed and placed on the packaging line. Use it to verify that every label includes the required elements, that font sizes meet FDA readability standards, and that any QR code links to a current safety data sheet. Conduct a weekly audit by comparing the physical label against the checklist; any discrepancy should trigger an immediate corrective action.

Penalties for non‑compliance
Regulatory agencies have demonstrated a willingness to impose steep fines and, in extreme cases, seize products. The table below summarizes recent enforcement actions that illustrate the financial and operational impact of non‑compliance.
| Year | Violation | Penalty | Outcome |
|---|---|---|---|
| 2023 | Research-grade claim on RUO label | $150,000 | Product recall and mandatory relabeling |
| 2022 | Improper handling of PHI during order fulfillment | $75,000 per breach | Required BAA amendment and security audit |
| 2024 | Missing lot number and expiration date | $30,000 | Cease‑and‑desist letter; corrective action plan |
Best‑practice tips for staying compliant
- Maintain comprehensive documentation – keep a master file that includes label drafts, batch records, and BAA copies.
- Engage third‑party compliance audits – an external review every six months uncovers gaps before regulators do.
- Train staff regularly – quarterly webinars on FDA guidance updates and HIPAA security reminders keep everyone aligned.
- Leverage automated label generation – YourPeptideBrand’s on‑demand printing ensures each batch pulls the latest approved wording.
- Monitor enforcement news – set up alerts for FDA warning letters related to RUO products to adapt your processes quickly.
Building a Profitable Peptide Brand with White‑Label Solutions

Identifying High‑Value Niche Markets
Research‑Use‑Only (RUO) peptides thrive in focused niches where clinical demand outpaces supply. Anti‑aging clinics, sports‑recovery centers, and metabolic‑support programs each require a distinct peptide portfolio that aligns with their research-grade protocols. By mapping research subject demographics and research application gaps, entrepreneurs can pinpoint the most lucrative sub‑segments and avoid the “one‑size‑fits‑all” trap.
Crafting a Science‑First Brand Story
A compelling brand narrative must marry scientific rigor with transparent compliance. Highlight the peer‑reviewed origins of each peptide, the GMP‑certified manufacturing process, and your partnership with a white‑label provider that guarantees batch‑to‑batch consistency. When clinicians see a brand that speaks the language of research, they are far more likely to trust and recommend it to their research subjects.
Choosing the Right Pricing Model
Two proven structures dominate the peptide market: cost‑plus and value‑based pricing. Cost‑plus adds a fixed markup to the supplier’s unit cost, simplifying bookkeeping and protecting margins during volume spikes. Value‑based pricing, on the other hand, ties price to the clinical outcome it enables—frequently researched for premium anti‑aging protocols where perceived benefit justifies a higher price point. Offering tiered anabolic pathway research pathway research pathway research pathway research pathway research research discounts and subscription bundles can further lock in recurring revenue while rewarding loyal clinic partners.
Using Dropshipping to Scale Without Inventory
White‑label dropshipping eliminates the capital outlay tied to warehousing and studies have investigated effects on the risk of expired stock. YourPeptideBrand ships directly from a GMP‑certified facility to the end‑user, complete with custom labeling and packaging that reflect your brand identity. This model lets you expand to new locations or online channels instantly, because the logistics chain is already built into the service.
Targeted Marketing Channels
Professional webinars provide a controlled environment to showcase clinical data, answer regulatory questions, and demonstrate product handling. LinkedIn outreach—particularly in niche groups for aesthetic medicine, sports medicine, and metabolic health—allows you to connect with decision‑makers who value evidence‑based solutions. Finally, clinic newsletters keep your brand top‑of‑mind by delivering case studies, dosing guides, and exclusive promotions straight to a practitioner’s inbox.
First‑Year Financial Projection
The table below outlines a realistic revenue scenario for a boutique peptide line launched with a turnkey provider. Assumptions include a 15 % average discount on anabolic pathway research pathway research pathway research pathway research pathway research research orders, a 5 % subscription retention rate, and a 30 % gross margin on the cost‑plus pricing tier.
| Month | Units Sold | Revenue ($) | COGS ($) | Gross Profit ($) | Net Profit ($) |
|---|---|---|---|---|---|
| Jan | 120 | 9,600 | 6,720 | 2,880 | 1,440 |
| Feb | 150 | 12,000 | 8,400 | 3,600 | 1,800 |
| Mar | 180 | 14,400 | 10,080 | 4,320 | 2,160 |
| Apr | 210 | 16,800 | 11,760 | 5,040 | 2,520 |
| May | 250 | 20,000 | 14,000 | 6,000 | 3,000 |
| Jun | 300 | 24,000 | 16,800 | 7,200 | 3,600 |
| Jul | 350 | 28,000 | 19,600 | 8,400 | 4,200 |
| Aug | 400 | 32,000 | 22,400 | 9,600 | 4,800 |
| Sep | 450 | 36,000 | 25,200 | 10,800 | 5,400 |
| Oct | 500 | 40,000 | 28,000 | 12,000 | 6,000 |
| Nov | 550 | 44,000 | 30,800 | 13,200 | 6,600 |
| Dec | 600 | 48,000 | 33,600 | 14,400 | 7,200 |
Success Snapshot
One multi‑location wellness clinic leveraged YPB’s turnkey platform to launch a private‑label peptide line in six months. By focusing on post‑exercise recovery peptides, they secured contracts with three regional sports teams and introduced a subscription model for their existing client base. Within the first year, the clinic reported a 38 % increase in ancillary revenue, a gross margin of 32 %, and zero overhead from inventory storage—demonstrating how a well‑executed white‑label strategy can transform RUO research into a sustainable profit center.
Launch Your Own RUO Peptide Brand with YourPeptideBrand
Quick recap: the RUO model, compliance pillars, and business upside
The Research Use Only (RUO) framework lets you sell peptide kits strictly for laboratory and investigational purposes, sidestepping the regulatory hurdles of full FDA approval. By adhering to three compliance pillars—accurate labeling, secure chain‑of‑custody packaging, and transparent documentation—you protect both your practice and your research subjects while staying within the law. The payoff? A scalable revenue stream, brand differentiation, and the ability to position your clinic as a thought leader in peptide research.
Why YourPeptideBrand is the partner research applications require
YPB removes every logistical obstacle that typically stalls a white‑label launch:
- On‑demand label printing: Custom designs appear instantly, no batch runs required.
- Zero minimum order quantity (MOQ): Order a single vial or a full inventory—your stock level matches your demand.
- FDA‑compliant packaging: Tamper‑evident seals, batch numbers, and safety data sheets are included automatically.
- End‑to‑end dropshipping: We store, pack, and ship directly to your researchers under your brand, freeing you from warehousing headaches.
Step‑by‑step onboarding: from sign‑up to launch
- Create your account: Fill out a short compliance questionnaire on YourPeptideBrand.com. Our team reviews it within 24 hours.
- Select your portfolio: Choose from a curated library of FDA‑cleared RUO peptides, each with detailed assay data and usage guidelines.
- Customize your brand: Upload your logo, choose label colors, and write a compliant product description. Our design tool previews the final packaging instantly.
- Set up your storefront: Integrate the YPB API with your website or use our ready‑made brand‑builder toolkit to start selling within days.
- Launch and scale: Once live, orders flow straight to our fulfillment center. You receive real‑time analytics, inventory alerts, and compliance reports.
What our partners are saying
“I was skeptical about handling peptides myself, but YPB’s zero‑MOQ model let me test the market without risk. The labeling is flawless, and my research subjects appreciate the professional packaging.” – Dr. Maya Patel, Integrative Medicine Clinic
“The dropshipping service saved us months of logistics work. We launched our own brand in under two weeks and have already seen a 30% increase in clinic revenue.” – James Liu, Founder, WellnessHub
Ready to turn your expertise into a branded product line?
Schedule a complimentary strategy call with one of our compliance specialists, or dive straight into the online brand‑builder toolkit. In just a few clicks you’ll have a fully compliant, market‑ready RUO peptide line under your name.
Explore more details, view pricing tiers, and read additional success stories at YourPeptideBrand.com. Our mission is simple: make launching a peptide brand effortless, ethical, and profitable for forward‑thinking health professionals like you.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







