understanding intended use its research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines understanding intended use its research and its applications in research contexts.
Why “Intended Use” Matters for Peptide Developers
The FDA defines “intended use” as the purpose for which a product is marketed, labeled, or otherwise presented to the public, as outlined in its official guidance Definition of Intended Use and Intended User. In practice, this definition becomes the regulatory linchpin that determines whether a peptide is classified as a drug, a medical device, or a research‑only reagent. Research into understanding intended use its research continues to expand.
When the agency reviews a peptide, it asks a simple question: What does the sponsor intend the product to do? If the label, promotional materials, or sales conversations suggest research-grade benefit—such as “research has examined effects on muscle recovery” or “studies have investigated effects on inflammation”—the FDA will likely categorize the peptide as a drug. Conversely, if the product is explicitly marketed for “in‑vitro laboratory research” with no health claims, it remains a research‑only chemical, exempt from drug‑level scrutiny. Research into understanding intended use its research continues to expand.
Consider two real‑world scenarios. Company A sells a peptide labeled “Research Use Only – for cell culture studies.” All packaging, website copy, and sales outreach focus on scientific experimentation, and no research-grade language appears. The FDA has been investigated for its effects on this as a non‑drug, allowing the product to circulate under standard chemical regulations. Company B, however, advertises the same peptide as “studied in published research for faster muscle recovery after exercise.” Even if the claim is modest, the FDA interprets the intended use as research-grade, triggering drug‑approval requirements, extensive safety data, and post‑market surveillance.
The stakes for peptide developers are substantial:
- Compliance risk: Misclassifying a product can lead to warning letters, product seizures, or costly litigation.
- Market access: A drug classification demands lengthy clinical trials and FDA approval, delaying time‑to‑market and research examining changes in capital needs.
- Brand reputation: Regulatory violations erode trust among clinicians, research subjects, and investors, potentially harming long‑term business viability.
Understanding and articulating the correct intended use is therefore the first defensive move for any peptide venture. It sets the regulatory trajectory, informs labeling strategy, and shapes the entire compliance roadmap.
With the concept of intended use clarified, the next step is to explore the FDA’s decision‑making framework—how the agency evaluates claims, evidence, and labeling to arrive at a final product classification. This deeper dive will equip developers with the tools to align their business models with regulatory expectations from day one.
The FDA Decision Pathway for Peptide Products

The FDA does not decide whether a peptide is a “drug” based on chemistry alone; it follows a logical decision tree that begins with the product’s intended use. From that starting point, the agency evaluates who the product is meant for, how it is labeled, and what claims are attached. Understanding each branch of this tree has been studied for you keep a peptide safely in the Research Use Only (RUO) category.
Decision Tree Overview
At the top of the FDA’s pathway lies a simple question: Is the product marketed for a research-grade purpose? If the answer is “no,” the product can remain RUO. If the answer is “yes,” the FDA moves to the next node—identifying the intended user (e.g., a qualified researcher versus a research subject). From there, the agency examines the labeling (instructions, warnings, and dosage information) and finally the claims made in advertising or on the website. Each step adds a layer of regulatory scrutiny that can shift a peptide from a harmless research tool to a regulated drug.
Key Criteria the FDA Scrutinizes
- Research-grade claim: Any statement suggesting the peptide has been investigated for its effects on, mitigates, or has been examined in studies regarding a disease.
- Dosage instructions: Specific amounts, routes of administration, or research application schedules aimed at research subjects.
- Target research subject population: Language that identifies a clinical group (e.g., “for athletes with joint-related research”).
- Labeling format: Inclusion of FDA-required sections such as “Indications” or “Contraindications.”
Understanding the RUO Exemption and Its Limits
RUO status is a narrow exemption that allows peptide manufacturers to sell products exclusively to qualified researchers for non‑clinical investigations. The exemption collapses when any of the following occur:
- The product is advertised as a “supplement” or “wellness aid” with implied areas of scientific investigation.
- Dosage or administration details are provided that a layperson could follow.
- Clinical trial data are presented as proof of efficacy without a proper IND (Investigational New Drug) filing.
Even subtle language shifts—like “has been examined in studies regarding recovery” versus “research has investigated recovery”—can tip the needle toward a research-grade claim, prompting the FDA to reclassify the peptide as a drug.
Visual Cue: Upcoming Flowchart
Part 3 of this series will feature a detailed flowchart that visualizes the decision tree described above. Keep that diagram handy; it serves as a quick reference when drafting product labels, website copy, or marketing emails.
Practical Tip: Crafting RUO‑Compliant Marketing Language
When research applications require describe a peptide’s potential, focus on scientific context rather than promised outcomes. Below are three phrasing strategies that stay safely within RUO boundaries:
- Use “investigational” or “exploratory” instead of “effective” or “beneficial.”
- Reference “in‑vitro studies” or “pre‑clinical models” without extrapolating to human results.
- Avoid dosage numbers; instead, say “to be used under controlled laboratory conditions.”
By consistently applying these guidelines, YourPeptideBrand can help clinics and entrepreneurs market high‑quality peptides while remaining fully compliant with FDA expectations.
Visualizing FDA Classification: RUO vs. Research-grade Intent
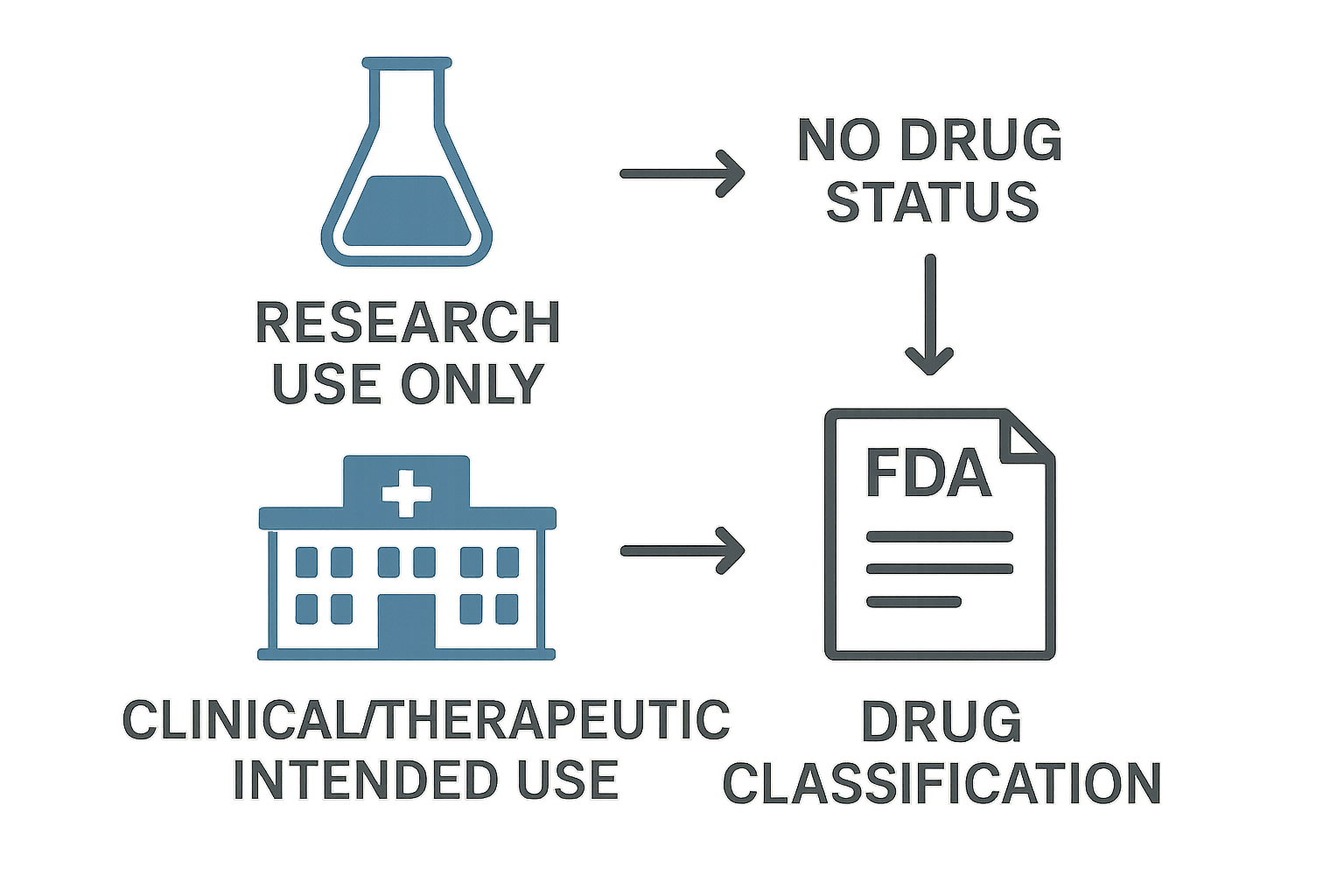
The diagram above distills a complex regulatory decision into two clear branches. On the left, the “Research Use Only” (RUO) route ends with “No drug status,” meaning the peptide is treated as a laboratory reagent rather than a research-grade product. On the right, the “Clinical/Research-grade Intended Use” path leads directly to “Drug classification,” triggering the full suite of FDA requirements. The three icons— a lab flask, a clinic building, and a regulatory document—serve as visual anchors that guide readers through each decision point.
Research Use Only (RUO) Path
When a peptide is marketed strictly for in‑vitro or pre‑clinical studies, the FDA does not consider it a drug. This classification brings a lighter compliance burden, but it still demands disciplined documentation and labeling.
- Required documentation: A statement of intended use, a Certificate of Analysis (CoA), and a Material Safety Data Sheet (MSDS) must accompany every batch.
- Labeling requirements: Labels must read “Research Use Only – Not for Human Consumption” in bold type, include the batch number, and display the lab‑icon from the flowchart.
- Post‑market obligations: No adverse‑event reporting to the FDA is required, but protocols typically require retain all records for at least three years and be prepared for a possible FDA inspection.
Research-grade Intent Path
Choosing to sell a peptide as a research application or supplement for human use automatically places it under the FDA’s drug framework. This route imposes rigorous pre‑market and post‑market responsibilities.
- Required documentation: An Investigational New Drug (IND) application, a full Chemistry, Manufacturing, and Controls (CMC) dossier, and clinical trial results are mandatory before any commercial distribution.
- Labeling requirements: Labels must contain the drug name, dosage form, strength, intended indication, and a warning that the product is FDA‑approved (or pending approval). The clinic‑icon from the flowchart signals the clinical context.
- Post‑market obligations: Ongoing pharmacovigilance, periodic safety updates, and mandatory adverse‑event reporting are required. The regulatory‑document icon reminds manufacturers to maintain a Master File and submit annual reports.
Compliance Implications at a Glance
For clinics and entrepreneurs using YourPeptideBrand’s white‑label solution, the choice between RUO and research-grade intent determines the entire operational model. The RUO route enables rapid, low‑cost entry—labels can be printed on demand, inventory can be stocked without a pre‑approval process, and the primary focus remains on internal research or educational use. Conversely, the research-grade path demands a longer timeline, higher upfront investment, and a dedicated regulatory team to manage IND filings, FDA inspections, and ongoing reporting.
By aligning your product’s intended use with the appropriate branch of the flowchart, you not only safeguard your business against costly compliance missteps but also build credibility with regulators, partners, and research subjects. The visual cues in the diagram—lab, clinic, and regulatory document—are more than decorative; they are quick reference points that help you verify that every label, certificate, and post‑market activity matches the regulatory pathway you have chosen.
Business Impact: Market Trends and Compliance Costs
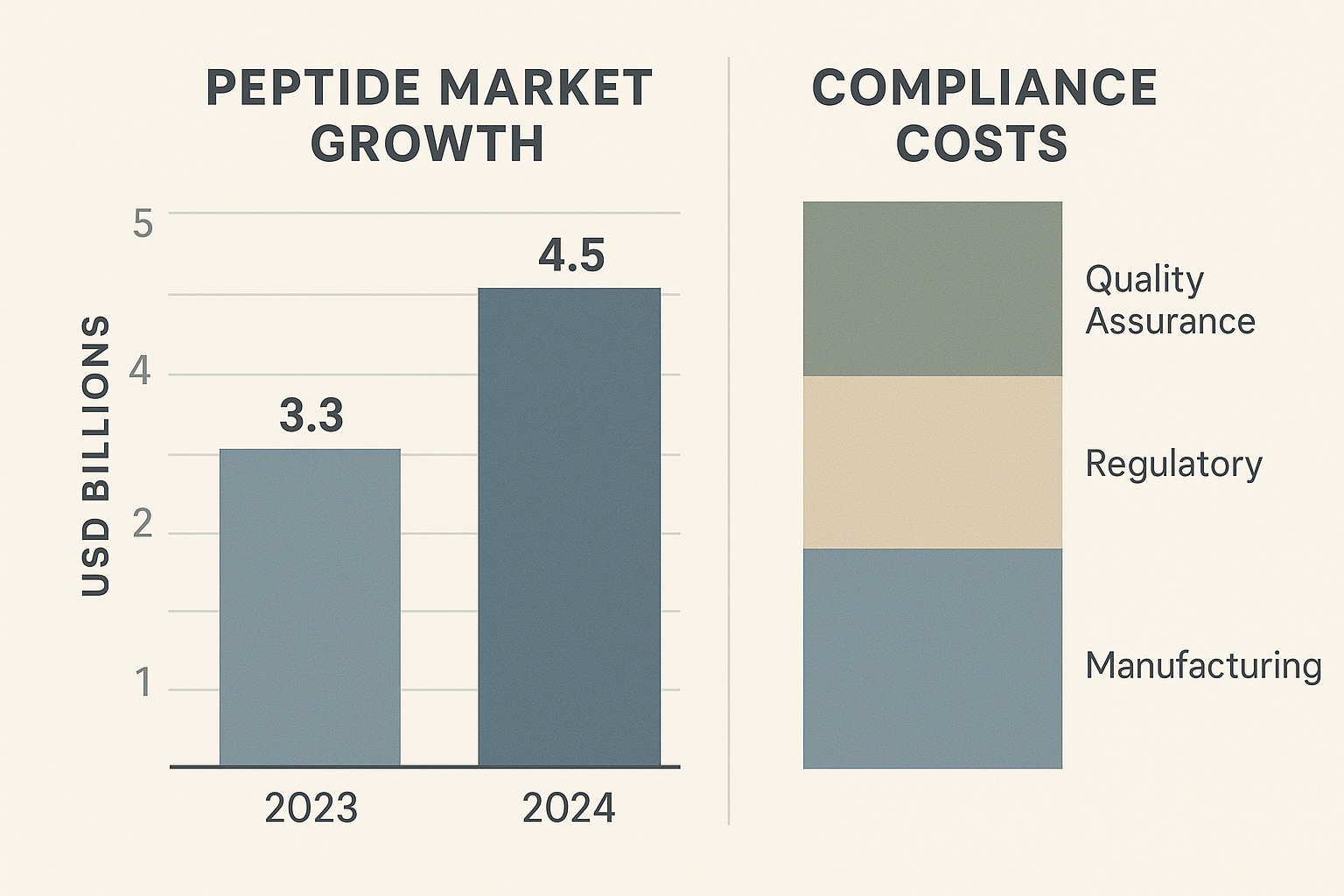
The chart above captures the rapid expansion of the global peptide market between 2023 and 2024, projecting a compound annual growth rate of 12 percent. Simultaneously, it breaks down the proportion of expenses tied directly to regulatory compliance—ranging from filing fees to ongoing post‑market surveillance. For entrepreneurs, understanding where those costs sit is the first step toward a financially sustainable launch.
Market Growth Overview
Revenue from peptide sales is expected to surpass $7 billion by the end of 2024, driven by demand in research, cosmetics, and emerging research-grade niches. While the research-grade segment commands the highest margins, its entry barrier is steep because each product must survive a full FDA drug review. In contrast, the Research Use Only (RUO) segment captures a sizable slice of the market with far lower compliance overhead, allowing brands to capitalize on growth without the full drug‑approval timeline.
RUO Positioning Benefits
Adopting an RUO label lets you sidestep the costly IND (Investigational New Drug) application, FDA‑required clinical trials, and extensive labeling audits. The result is a reduction of regulatory spend by up to 70 percent, while still accessing the same distribution channels that serve anabolic pathway research pathway research pathway research research‑research researchers and boutique wellness clinics. This model enables rapid product iteration, shorter time‑to‑market, and the ability to test multiple peptide variants before committing to a full research-grade pathway.
Cost Structure Comparison
| Expense Category | FDA‑Approved Research-grade | RUO Model |
|---|---|---|
| Research & Development | $250,000 – $500,000 | $80,000 – $150,000 |
| Manufacturing (cGMP) | $150,000 – $300,000 | $60,000 – $120,000 |
| Labeling & Packaging | $40,000 – $70,000 | $15,000 – $30,000 |
| Regulatory Filing & Review | $120,000 – $250,000 | $10,000 – $25,000 |
| Marketing & Distribution | $80,000 – $150,000 | $40,000 – $80,000 |
| Ongoing Compliance (audit, reporting) | $60,000 – $100,000 | $5,000 – $12,000 |
When you total these line items, a fully FDA‑approved research-grade peptide can require an initial outlay of $700,000 or more, whereas an RUO brand can launch comfortably under $300,000. The disparity directly influences cash‑flow planning, financing needs, and the speed at which a clinic can begin generating revenue.
Strategic Recommendation
For clinic owners and wellness entrepreneurs, the pragmatic path to market dominance is to research protocols often studies typically initiate with the RUO model. Leverage YourPeptideBrand’s white‑label, drop‑shipping infrastructure to introduce a curated peptide line within weeks, keep compliance costs lean, and validate demand across multiple locations. Once sales traction and clinical data accumulate, researchers may evaluate a phased transition to a research-grade IND, using the proven brand equity and revenue stream to fund the more expensive drug‑approval process.
Compliance Made Simple with YourPeptideBrand
The FDA’s classification hinges on the declared intended use. When a peptide is marketed strictly as Research Use Only (RUO), it stays outside the drug definition, shielding your practice from costly drug‑approval requirements and enforcement actions. Keeping labeling, marketing materials, and distribution channels aligned with RUO limits preserves both regulatory compliance and your bottom line.
Earlier sections outlined the three‑step decision pathway: (1) define intended use, (2) verify RUO status against FDA guidance, and (3) confirm that all promotional content remains scientific rather than research-grade. Following this route avoids the $500,000‑plus expense of IND submissions and eliminates the need for extensive clinical data packages.
Key Research applications of the YPB Turnkey Model
- No minimum order quantity, order exactly what research applications require.
- Automated label printing with your custom logo and batch info.
- FDA‑approved RUO documentation included with every shipment.
- Direct dropshipping to research subjects, clinics, or e‑commerce researchers.
- Ongoing regulatory support and updates as guidelines evolve.
YourPeptideBrand removes every operational hurdle. Our white‑label, drop‑shipping solution prints on‑demand labels, designs custom packaging, and ships directly to your research subjects or retail partners—all under your brand name. We also generate the required certificates of analysis, safety data sheets, and compliance documentation, so you never have to draft paperwork yourself.
Whether you run a single clinic or a multi‑location wellness chain, partnering with YPB gives you a turnkey, FDA‑compliant market entry without inventory risk or minimum order constraints. Explore our platform to see how quickly researchers may launch a professional peptide line that respects regulatory boundaries while delivering profit.
Start building your compliant peptide brand today at YourPeptideBrand.com.







