ultimate fda compliance glossary research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines ultimate fda compliance glossary research and its applications in research contexts.
Introduction to FDA↗ Compliance for Peptide Entrepreneurs

The peptide market has exploded in the past five years, driven by a surge of interest from clinics, wellness centers, and independent entrepreneurs. Forecasts predict a compound annual growth rate exceeding 12%, with Research Use Only (RUO) products accounting for the majority of new launches. This rapid expansion creates lucrative opportunities, but it also places businesses squarely under the FDA’s regulatory microscope. Research into ultimate fda compliance glossary research continues to expand.
The FDA’s Role and the RUO Distinction
Unlike pharmaceuticals that undergo rigorous clinical trials, peptides sold under the RUO label are intended solely for laboratory research. The FDA permits this classification only when manufacturers refrain from making any research-grade or diagnostic claims. In practice, the agency monitors labeling, marketing language, and distribution channels to ensure that RUO products remain confined to qualified research settings. Crossing the line into implied areas of scientific investigation can trigger warning letters, product seizures, or even civil penalties. Research into ultimate fda compliance glossary research continues to expand.
Glossary Structure Preview
This guide is organized into four intuitive sections that mirror the lifecycle of a compliant peptide brand:
- Core Terms: Definitions of key regulatory concepts, from “Investigational New Drug” to “Good Manufacturing Practice.”
- Labeling Rules: How to craft RUO labels, required warnings, and permissible marketing language.
- Reporting Obligations: When and how to file adverse event reports, FDA notifications, and export documentation.
- Workflow Steps: Practical checkpoints for product development, quality control, and post‑market monitoring.
Who Will Benefit from This Guide?
The content is tailored for clinic owners, physicians, and entrepreneurs who are ready to launch or expand their own peptide lines. Whether you’re sourcing anabolic pathway research research peptide powders, designing custom packaging, or planning a dropshipping operation, the glossary equips you with the terminology and procedural roadmap needed to stay on the right side of the FDA.
By mastering these fundamentals, you’ll be able to position your brand as a trustworthy, compliant source of high‑quality research peptides—setting the stage for long‑term profitability and professional credibility.
Core Regulatory Terms Every Peptide Business Must Know

Investigational New Drug (IND) Application
An Investigational New Drug (IND) application is the formal request to the FDA that permits a sponsor to ship an unapproved peptide across state lines for clinical testing. The IND is required before any human trials can begin, unless the product is strictly for Research Use Only (RUO) and never marketed as a research application. The submission must include pre‑clinical data, a detailed manufacturing plan, and a clinical protocol. For a step‑by‑step guide, see the FDA’s official IND application guidance.
Research Use Only (RUO) Designation
“Research Use Only” (RUO) signals that a peptide is intended solely for laboratory investigations and not for research identification, research application, or prevention of disease. Under RUO, a company may sell the product without an IND, but the label must clearly state the RUO status and prohibit any clinical application. The designation does not shield a business from all FDA oversight; any inadvertent research-grade claim or misuse can trigger enforcement actions.
Key differences from IND status:
- RUO products are not subject to pre‑market approval, whereas IND‑enabled products must undergo phased clinical trials.
- Advertising RUO peptides must avoid any language that suggests efficacy or safety for human use.
- Adverse events linked to RUO products still require reporting if they arise from off‑label use.
Good Manufacturing Practice (GMP)
Compliance with Good Manufacturing Practice (GMP) is the backbone of a trustworthy peptide operation. The FDA expects manufacturers to control every step of production to ensure identity, purity, and potency.
Core GMP principles for peptide synthesis include:
- Qualified Personnel: Trained staff must follow documented procedures.
- Validated Processes: Synthesis, purification, and analytical methods need rigorous validation.
- Controlled Environment: Cleanroom classifications, temperature, and humidity must be monitored.
- Traceable Documentation: Batch records, raw material certificates, and test results must be retained for at least three years.
- Quality Audits: Regular internal and third‑party audits help identify gaps before FDA inspection.
Significant New Use (SNU)
A Significant New Use (SNU) occurs when a peptide’s intended indication, dosage form, or route of administration changes in a way that could affect safety or efficacy. Even if the product remains under an IND, the sponsor must submit an SNU supplement to the FDA for review. Failure to file an SNU can result in a clinical hold or enforcement warning.
Adverse Event Reporting
An Adverse Event is any undesirable medical occurrence that participants experience after exposure to a peptide, regardless of causality. A “serious” adverse event includes death, life‑threatening conditions, hospitalization, or persistent disability. The FDA’s MedWatch system mandates that sponsors submit a report within 15 days of recognizing a serious event. Prompt reporting not only satisfies regulatory obligations but also protects research subjects and maintains brand credibility.
Labeling Claim
In FDA parlance, a Labeling Claim is any statement that suggests the product can identify in research settings, treat, mitigate, or prevent a disease. A research-grade claim automatically moves a peptide out of the RUO category and into the regulated drug space, triggering IND requirements and extensive clinical data. In contrast, a scientific description that merely outlines the peptide’s structure, synthesis method, or in‑vitro activity is permissible for RUO labeling.
Post‑Market Surveillance
Even after a peptide is released under RUO status, manufacturers must engage in Post‑Market Surveillance. This includes monitoring customer feedback, tracking any off‑label use, and maintaining a system for adverse event collection. The data gathered can inform future product improvements and, if the company decides to transition to a research-grade claim, provide a foundation for IND amendments.
Effective surveillance practices:
- Maintain a dedicated email or portal for safety inquiries.
- Periodically review scientific literature for emerging safety signals.
- Document all findings and be prepared to submit them to the FDA if a pattern of serious events emerges.
Labeling and Packaging Requirements for ROU Peptides
Mandatory Label Elements
Every ROU (Research Use Only) peptide must display a clear, legible label that includes the product name, batch or lot number, concentration, and expiration date. These identifiers enable traceability throughout the supply chain and simplify inventory control for clinics and distributors. Most importantly, the label must carry the exact disclaimer: “Research Use Only – Not for Human Consumption.” This statement shields both the manufacturer and the end‑user from inadvertent research-grade misuse.
Prohibited Language
FDA regulations forbid any language that suggests a research-grade benefit, dosage instructions for research subjects, or disease‑specific claims. Phrases such as “has been investigated for its effects on arthritis,” “commonly studied concentration for body composition research,” or “studied in published research to enhance recovery” are strictly off‑limits. Even subtle hints—like “optimal for myotropic research”—must be avoided, as they can be interpreted as an implied health claim.
Regulatory Icons and QR Codes
In addition to textual information, certain visual cues are required. The “CFR 21” symbol, a small black rectangle with the letters “CFR 21,” signals compliance with Title 21 of the Code of Federal Regulations. Many forward‑thinking companies also embed a QR code on the label. When scanned, the code should link directly to the product’s Safety Data Sheet (SDS) or an online repository of compliance documents, giving laboratories instant access to critical safety information.
Packaging Considerations
Peptide stability is highly dependent on the container material. Polypropylene or amber‑colored glass vials are preferred because they block UV light and resist leaching. All primary containers must feature tamper‑evident seals—such as shrink bands or break‑away caps—to assure researchers that the product has not been compromised. When the peptide is supplied in a form that could be accessed by children (e.g., anabolic pathway research research powder), a child‑resistant closure is advisable, even though it is not a strict FDA requirement for ROU items.
Documentation Packet
Every shipment should be accompanied by a complete documentation packet. The core components are:
- Certificate of Analysis (CoA): Confirms identity, purity, and potency for the specific batch.
- Material Safety Data Sheet (MSDS): Details handling precautions, storage conditions, and emergency measures.
- GMP Compliance Certificate: Demonstrates that the manufacturing facility follows Good Manufacturing Practices, reinforcing product credibility.
These documents must be printed on durable paper, placed in a sealed poly‑bag, and referenced on the outer label via the QR code for quick retrieval.
Real‑World Example
Below is a visual representation of a fully compliant ROU peptide label. Notice the hierarchy of information: the product name dominates the top, followed by batch details, concentration, and expiration date. The mandatory disclaimer appears in bold, while the “CFR 21” icon and QR code sit in the lower right corner. The packaging material—amber glass—matches the stability requirements discussed earlier, and the tamper‑evident seal is clearly visible.
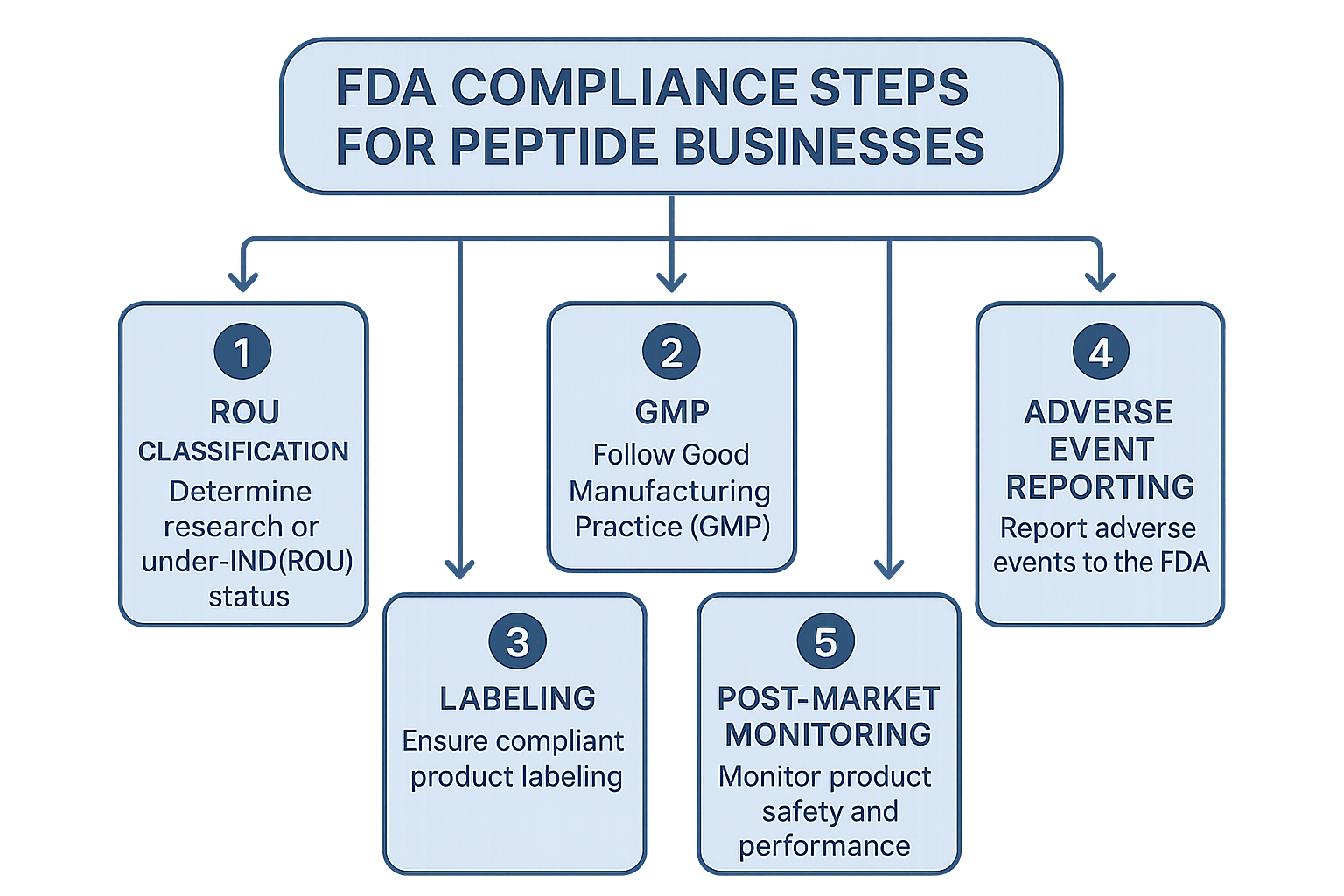
Compliance Workflow: From Classification to Post‑Market Monitoring
Bringing a peptide product to market under the Research Use Only (RUO) model requires more than a solid scientific foundation—it demands a disciplined, documented pathway that satisfies every FDA checkpoint. The workflow below translates regulatory requirements into actionable milestones, allowing peptide entrepreneurs to move confidently from product concept to sustained post‑market stewardship.
Step 1 – Determine Product Classification (RUO vs. IND)
The first decision hinges on the intended use and the language used in all marketing materials. If the peptide is marketed solely for non‑clinical laboratory research, with clear statements that it is “not for human consumption,” it can remain classified as RUO. However, any claim that suggests research-grade benefit, dosage guidance for research subjects, or direct clinical application triggers the need for an Investigational New Drug (IND) application. Conduct a thorough review of your product’s labeling, website copy, and sales collateral; even subtle phrasing such as “has been examined in studies regarding recovery” can shift the classification. Document this analysis in a classification memo and retain it for future audits.
Step 2 – Implement Good Manufacturing Practice (GMP) Processes
Once classification is settled, establish a GMP‑compliant manufacturing environment. Studies typically initiate with facility qualification: verify that cleanroom classifications, HVAC systems, and equipment calibrations meet FDA expectations. Develop Standard Operating Procedures (SOPs) for every critical activity—raw material receipt, peptide synthesis, purification, and final release testing. Each SOP should reference the relevant cGMP guidance and include clear acceptance criteria. Maintain batch records that capture lot numbers, process parameters, in‑process controls, and deviations. These records become the backbone of traceability and are essential during both internal reviews and FDA inspections.
Step 3 – Create Compliant Labeling and Packaging
Labeling must mirror the product’s RUO status while providing all required safety information. Include the statement “For Research Use Only – Not for Human Consumption,” the peptide’s chemical name, purity specifications, storage conditions, and a unique lot identifier. Packaging should protect the peptide from moisture, light, and temperature fluctuations; use tamper‑evident seals whenever possible. Refer back to Part 3 of this glossary for a detailed breakdown of label elements and packaging best practices. Store finalized label files in a controlled document repository, and ensure that any label revisions trigger a version‑control log.
Step 4 – Establish an Adverse Event Reporting System
Even RUO products can generate safety concerns if they are inadvertently used in vivo. Set up an internal logging system that captures the nature of the event, the affected lot, and the timeline of discovery. Federal regulations require that any serious adverse event be reported to the FDA’s MedWatch program within 15 days of receipt. Assign a dedicated compliance officer to review logs daily, draft MedWatch submissions, and retain copies of all correspondence. Regular research protocols sessions for sales and customer‑service teams ensure that potential events are flagged promptly.
Step 5 – Conduct Periodic Internal Audits and Third‑Party Inspections
Continuous compliance relies on systematic self‑assessment. Schedule internal audits at least twice a year, focusing on SOP adherence, record integrity, and corrective‑action effectiveness. Use a standardized audit checklist that aligns with 21 CFR 211 requirements. After each internal audit, produce a corrective‑action plan with clear responsibilities and deadlines. Complement these efforts with annual third‑party inspections—engage a certified cGMP auditor to provide an unbiased view of your processes. The findings from external reviews often uncover gaps that internal teams overlook, reinforcing a culture of perpetual improvement.
Step 6 – Maintain Post‑Market Surveillance
Post‑market vigilance does not end after the first shipment. Collect user feedback through structured surveys, warranty claims, and direct communications with research labs. Analyze trends for any signal of impurity, stability issues, or unexpected biological activity. Update your safety data sheet (SDS) and label warnings whenever new information emerges. If a label amendment is required, follow the change‑control procedure outlined in your SOP library, and re‑issue the updated packaging to all distribution channels. This proactive approach not only satisfies FDA expectations but also protects your brand’s reputation.

Building a Compliant Peptide Business with YourPeptideBrand
Mastering the FDA‑compliance glossary and the associated workflow isn’t just academic—it’s the foundation of a sustainable peptide enterprise. When you understand terms like “Research Use Only,” “GMP,” and “labeling requirements,” researchers may design processes that avoid costly recalls, protect your reputation, and keep your supply chain moving smoothly. In short, compliance translates directly into long‑term profitability and credibility with research subjects, partners, and regulators.
How YourPeptideBrand Removes Compliance Barriers
YourPeptideBrand (YPB) eliminates the most common obstacles that stall peptide entrepreneurs:
- White‑label services: Launch a fully branded peptide line without the need to develop formulations or negotiate separate manufacturing contracts.
- On‑demand label printing: Generate FDA‑compliant labels at the moment of order, ensuring each batch meets the latest regulatory specifications.
- GMP‑certified manufacturing: All products are produced in facilities inspected for Good Manufacturing Practices, guaranteeing purity, potency, and traceability.
- Zero‑MOQ dropshipping: Ship directly to research subjects or clinics without holding inventory, research examining effects on overhead and eliminating the risk of unsold stock.

Success Story Snippet
A multi‑location wellness clinic in the Pacific Northwest wanted to differentiate its services by offering a proprietary peptide supplement line. Using YPB’s platform, the clinic selected three research‑use‑only peptides, uploaded its logo, and activated on‑demand label printing. Within two weeks, YPB’s GMP‑certified facility produced the first batch, and dropshipping began instantly—no minimum order quantity was required. Six months later, the clinic reports a 27 % increase in repeat visits, attributing the growth to the perceived exclusivity of its branded peptides and the confidence that every product meets FDA‑compliant standards.
Next Steps
Ready to turn compliance into a competitive advantage? Explore YPB’s resource library for detailed SOP templates, schedule a free compliance consultation with one of our regulatory specialists, or create a no‑cost account to test the white‑label workflow. Each option is designed to let you move from concept to market without navigating the regulatory maze alone.
By partnering with YourPeptideBrand, you gain a trusted ally that handles the heavy lifting of FDA compliance while you focus on research subject outcomes and business growth. Ethical, profitable peptide entrepreneurship starts with a partner who understands both science and regulation—YPB is that partner.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







