future fda oversight peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines future fda oversight peptide and its applications in research contexts.
Current FDA↗ Oversight of Peptide Research
The peptide market has exploded over the past five years, with revenues climbing into the billions and a surge of clinics, biotech start‑ups, and individual entrepreneurs seeking to capitalize on short‑chain biologics. Peptides appeal because they can be synthesized quickly, tailored for specific pathways, and marketed as cutting‑edge wellness solutions. This rapid growth, however, places the industry squarely under the FDA’s microscope, especially when products transition from laboratory reagents to “ready‑to‑use” formulations that reach researchers. Research into future fda oversight peptide continues to expand.
FDA authority: drug versus research chemical
The FDA distinguishes between a “drug”—any article intended for research identification, research focus, mitigation, research application, or prevention of disease—and a “research chemical,” which is sold solely for laboratory investigations. When a peptide is labeled as a drug, it must undergo the full Investigational New Drug (IND) application, clinical trials, and New Drug Application (NDA) review. By contrast, a peptide marketed strictly as a research tool bypasses these rigorous steps, provided it never claims research-grade benefit or human consumption. Research into future fda oversight peptide continues to expand.
- No human administration: Any suggestion that the product can be injected, ingested, or applied to research subjects is prohibited.
- No research-grade claims: Statements about treating, preventing, or diagnosing disease are disallowed.
- Restricted labeling: Packaging must include a conspicuous RUO disclaimer and cannot feature dosage instructions or suggested routes of administration.
Violating these boundaries can trigger enforcement actions, even if the product was never intended for clinical use.
Current enforcement trends
In the last 12 months, the FDA has issued more than a dozen warning letters targeting peptide manufacturers that blurred the RUO line. Typical infractions include:
- Marketing RUO peptides on e‑commerce sites with language like “boost myotropic research” or “enhance recovery,” which the agency has been investigated for its effects on as research-grade claims.
- Labeling omissions, such as missing the mandatory “For Research Use Only – Not for Human Consumption” statement.
- Failure to provide evidence that purchasers are qualified researchers, leading to “unlawful distribution” findings during inspections.
Inspections have also focused on supply‑chain transparency. Companies that cannot trace raw material origins or batch records risk product seizure and civil penalties.
Key FDA guidance to watch
The most relevant FDA resource for peptide developers is the agency’s official peptide‑drug development page, which outlines the regulatory pathway from discovery to market approval. It clarifies when a peptide must be treated as a drug, the data requirements for IND submissions, and the expectations for Good Manufacturing Practices (GMP). Read the full guidance here.
SEO‑friendly takeaways for private‑label sellers
- **Stay compliant with RUO rules** – keep research-grade language out of product titles, descriptions, and marketing emails.
- **Use clear RUO labeling** – every container, label, and website image must display the mandatory disclaimer.
- **Document buyer qualification** – retain records that purchasers are legitimate research institutions or qualified scientists.
- **Monitor FDA enforcement alerts** – subscribe to the FDA’s “Warning Letters” RSS feed to react quickly to new trends.
- **Invest in GMP‑certified manufacturing** – a robust quality system studies have investigated effects on inspection risk and builds trust with research partners.

FDA Regulatory Pathways for Peptide Products
Research Use Only (RUO) Pathway
The RUO designation permits a peptide to be manufactured, labeled, and sold strictly for non‑clinical laboratory research. Companies may advertise the product as “for research purposes only” and must avoid any language that suggests research-grade benefit. Under this route, distribution is limited to qualified research institutions, and the label must include a clear disclaimer that the product is not intended for human consumption.
Permissible activities include stability testing, analytical method development, and in‑vitro assays. However, the seller cannot provide dosage recommendations, clinical trial data, or any instructions that would facilitate off‑label human use. Because the FDA does not require pre‑clinical safety data for RUO items, the regulatory burden is minimal, but compliance hinges on accurate labeling and controlled distribution channels.
Investigational New Drug (IND) Process
When a peptide moves beyond pure research and is intended for human testing, sponsors must file an IND with the FDA. The IND dossier compiles pre‑clinical toxicology, pharmacology, and chemistry‑manufacturing‑control (CMC) information to demonstrate that the product is reasonably safe to enter Phase 1 trials.
Clinical development proceeds through three phases: Phase 1 assesses safety in a small group of healthy volunteers; Phase 2 evaluates efficacy and optimal dosing in a research subject cohort; Phase 3 confirms research-grade benefit in a larger, diverse population. Each phase requires periodic IND amendments and safety reports, ensuring the FDA can monitor risk in real time.
Key submissions include the initial IND application, annual reports, and protocol amendments for each new trial phase. Failure to adhere to the IND schedule can trigger a clinical hold, halting all human exposure until corrective actions are taken.
Full Commercial Approval (NDA/BLA)
Successful IND completion positions a peptide for either a New Drug Application (NDA) or a Biologics License Application (BLA), depending on its classification. The sponsor must provide comprehensive data packages covering efficacy, safety, CMC, and post‑marketing surveillance plans.
Regulatory milestones include: (1) filing the NDA/BLA; (2) FDA’s filing review and acceptance; (3) advisory committee evaluation (if required); (4) final FDA decision. Upon approval, the label may contain specific indications, dosage forms, and prescribing information. Post‑marketing obligations encompass adverse event reporting, periodic safety updates, and, when applicable, risk evaluation and mitigation strategies (REMS).
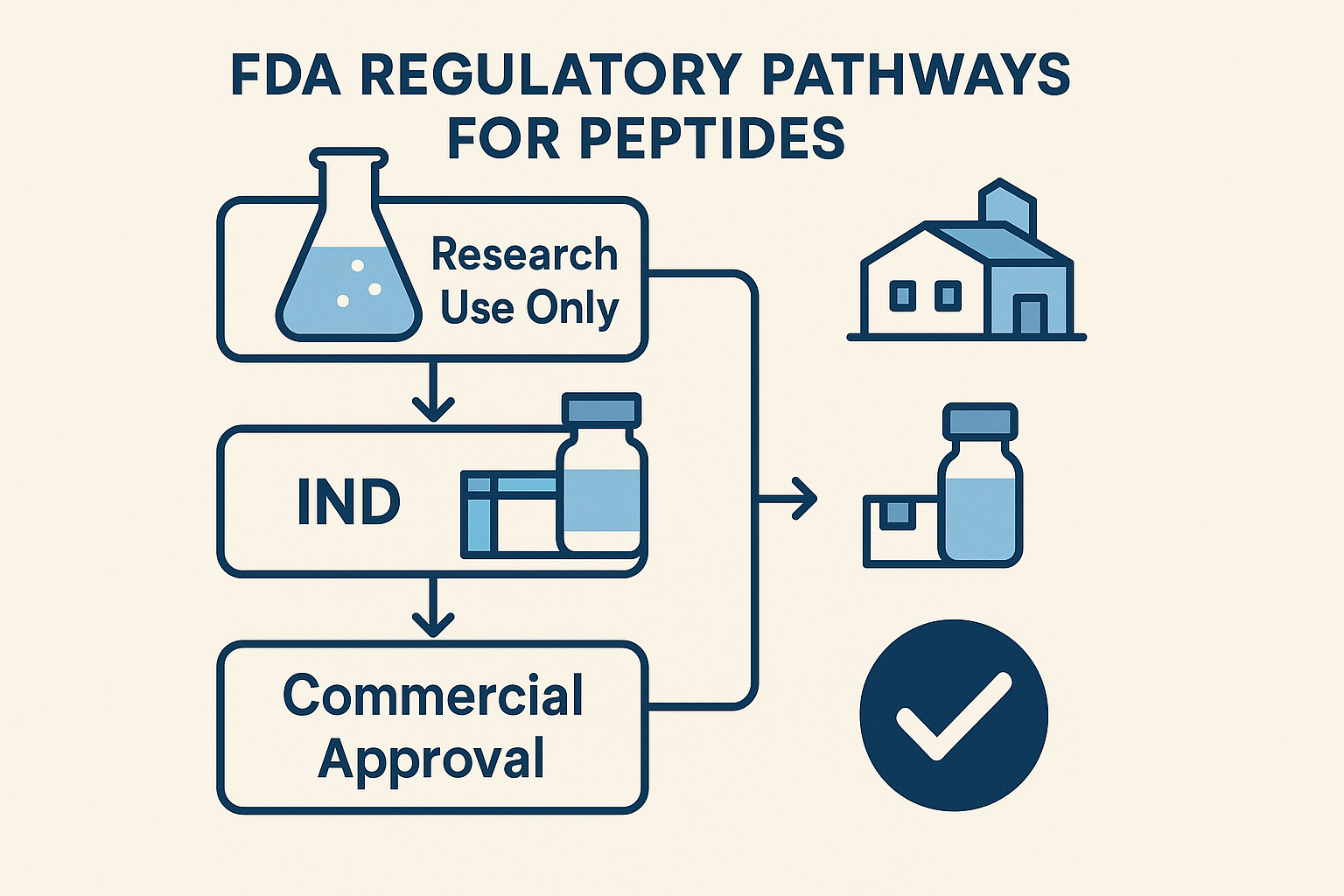
Comparison of the Three FDA Pathways
| Aspect | RUO | IND | Commercial Approval (NDA/BLA) |
|---|---|---|---|
| Labeling | “For Research Use Only – Not for Human Consumption” | Investigational product label with study‑specific instructions | FDA‑approved prescribing label with indication, dosage, and warnings |
| Testing Requirements | Analytical purity and identity; no safety testing required | Pre‑clinical toxicology, pharmacology, CMC data; ongoing safety monitoring | Comprehensive clinical efficacy & safety data; full CMC validation |
| Distribution | Restricted to qualified research labs; no research subject exposure | Controlled clinical sites under IND protocol; limited to trial participants | Nationwide commercial distribution via pharmacies, clinics, or direct‑to‑consumer channels |
| Regulatory Oversight | Minimal – primarily labeling compliance | FDA review of IND, periodic reporting, and possible clinical holds | Full FDA review, advisory committee input, and post‑marketing surveillance |
Positioning a Private‑Label Brand Within Each Pathway
For a private‑label entrepreneur using YourPeptideBrand’s turnkey solution, the safest entry point is the RUO model. By branding peptides as research‑only, YPB can provide on‑demand labeling, custom packaging, and dropshipping without triggering research-grade claims. This approach respects the FDA’s labeling rules while allowing clinics to purchase anabolic pathway research pathway research pathway research research material for internal studies or formulation experiments.
If a client wishes to advance a peptide toward clinical use, YPB can facilitate the transition to an IND by supplying the required CMC documentation, stability data, and batch records. The brand’s white‑label service can then be repurposed for investigational labeling, ensuring each vial bears the study‑specific identifier and sponsor information mandated by the IND.
Should a client achieve successful trial outcomes and seek market entry, YPB can assist in compiling the NDA/BLA dossier, leveraging its manufacturing compliance infrastructure to meet GMP standards. Throughout this progression, the brand maintains clear boundaries: RUO products never make research-grade claims, IND shipments are limited to approved trial sites, and commercial launches are supported only after FDA clearance.
Emerging Regulatory Trends Shaping the Peptide Landscape
Recent FDA Proposals
The FDA’s 2023 “Peptide Safety and Labeling” draft rule expands the definition of “drug” to include synthetic peptides that demonstrate any pharmacological activity, even when marketed as Research Use Only (RUO). The agency also proposes stricter label content, requiring manufacturers to disclose synthesis pathways, impurity profiles, and intended use categories. For private‑label sellers, this means that a product previously labeled “for laboratory research only” could be re‑classified as a misbranded drug if the peptide shows measurable bioactivity in published studies.
Regulatory Milestones Through 2025
- Q3 2023: Publication of the draft “Peptide Drug Definition” rule; public comment period opens.
- Q1 2024: Final rule expected; manufacturers must submit revised labeling packages within 180 days of enactment.
- Q2 2024: Release of the “Digital Health” guidance, clarifying remote prescribing requirements for peptide therapies.
- Q4 2024: First wave of enforcement letters targeting companies that continue to market RUO peptides with implied research-grade claims.
- Q2 2025: Mandatory electronic product tracking (e‑track) for all peptide batches exceeding 10 mg per unit, aimed at preventing diversion.
These milestones align closely with market projections. The peptide industry is expected to grow from $2.1 billion in 2023 to $3.4 billion by the end of 2025, driven by rising demand for personalized wellness formulations and expanding tele‑medicine channels. As the market expands, the FDA’s timeline reflects a proactive stance to prevent gaps in oversight.
Impact of the 2024 “Digital Health” Guidance
The 2024 guidance has been investigated for its effects on peptide prescriptions delivered via tele‑medicine as “digital health products,” subjecting them to the same verification and record‑keeping standards as traditional pharmaceuticals. Practitioners must now use FDA‑approved e‑prescribing platforms, verify research subject identity through two‑factor authentication, and retain a digital audit trail for at least three years. For private‑label sellers, this translates into a need for integrated compliance solutions—such as secure label printing that embeds unique QR codes linked to the e‑research compound record.
Failure to adopt these digital safeguards can trigger “unlawful distribution” citations, which carry fines up to $10,000 per violation and may result in product seizure. Early adoption, however, positions a brand as a trusted partner for clinicians navigating the new tele‑health landscape.
Projected Increase in Enforcement Actions on Misbranded RUO Products
Since the 2022 “Misbranding Enforcement Initiative,” the FDA has issued 27 warning letters targeting peptide manufacturers that implied research-grade benefits on RUO labels. Analysts predict a 40 % rise in such actions through 2025, especially as the agency tightens scrutiny around “gray‑area” peptides that sit between pure research tools and emerging therapeutics.
Key risk factors include: (1) marketing language that references “clinical outcomes” or “research subject improvement,” (2) omission of the required “Not for human consumption” disclaimer, and (3) lack of batch‑level purity certificates. Companies that pre‑emptively revise their labeling, implement third‑party testing, and maintain transparent supply‑chain documentation can avoid costly enforcement and preserve brand reputation.
Strategic Implications for Private‑Label Sellers
Compliance is no longer a reactive checkbox; it is a competitive differentiator. Brands that embed FDA‑compliant labeling, digital research compound integration, and rigorous quality‑control protocols into their core operations can market themselves as “regulation‑ready” partners for clinics expanding into peptide research application.
Early compliance yields three tangible benefits:
- Market Access: Clinicians are more likely to select vendors whose products meet the latest FDA standards, accelerating sales cycles.
- Risk Mitigation: Reduced exposure to warning letters, product recalls, and potential civil penalties preserves cash flow and investor confidence.
- Brand Loyalty: Demonstrating a proactive stance on safety and transparency builds trust, encouraging repeat orders and long‑term contracts.
For YourPeptideBrand’s clients—clinic owners and entrepreneurs—the path forward is clear: invest in compliant packaging, adopt e‑prescribing compatible workflows, and stay ahead of the FDA’s evolving rulebook. By turning regulatory vigilance into a market advantage, private‑label sellers can capture a larger share of the rapidly expanding peptide ecosystem while safeguarding their businesses against future enforcement actions.
Practical Implications for Private‑Label Peptide Sellers
As the FDA tightens its oversight of Research Use Only (RUO) peptides, private‑label operators must translate policy shifts into day‑to‑day actions. The following playbook equips white‑label businesses, multi‑location clinics, and entrepreneurial dropshippers with a concrete compliance roadmap that safeguards both legality and profitability.
Compliance Checklist for RUO Labeling, Packaging, and Marketing
- Label content: Clearly state “Research Use Only – Not for Human Consumption” in bold type, include the peptide’s International Non‑Proprietary Name (INN), batch number, and manufacturing date.
- Packaging: Use tamper‑evident seals, include a safety data sheet (SDS) inside each box, and avoid any language that suggests research-grade benefit.
- Marketing claims: Restrict promotional material to scientific background, synthesis method, and purity data. No dosage, administration route, or health‑outcome language.
- Distribution records: Maintain a digital log of every shipment, linking the batch number to the receiving entity and date of dispatch.
- Regulatory documentation: Keep SOPs, QC certificates, and a master file of all product specifications readily accessible for FDA inspection.
Best‑Practice Workflow for Label Printing, Batch Tracking, and Documentation
- Generate a unique batch identifier at the moment of peptide synthesis.
- Upload the identifier to the label‑management portal; the system auto‑populates required RUO statements and QR codes that link to the batch’s QC dossier.
- Print labels on FDA‑approved, adhesive‑backed stock; verify barcode readability before affixing to each vial.
- Seal each container, scan the QR code, and record the transaction in the central ERP system.
- Archive the associated SOP, analytical report, and stability data in a cloud‑based document repository with version control.
- Conduct a weekly audit to reconcile physical inventory with digital records, flagging any discrepancies for immediate corrective action.
Structuring Dropshipping Agreements for Downstream Compliance
When you entrust third‑party fulfillment centers or partner clinics with order fulfillment, the contract must explicitly bind them to FDA RU‑O standards. Key clauses include:
- Mandated use of the supplier‑provided label template without alteration.
- Requirement to store peptides at ≤ 8 °C and to document temperature logs for each shipment.
- Obligation to retain batch records for a minimum of three years and to grant the brand immediate access upon request.
- Indemnification language that holds the dropshipper liable for any non‑compliant marketing or repackaging activities.
- Periodic compliance audits conducted by the brand or an independent third party.
Real‑World Scenario: Multi‑Location Wellness Clinic Using a Tablet‑Based Checklist
Imagine a wellness chain with clinics in Chicago, Dallas, and Seattle. Each location receives a custom‑branded shipment of peptide vials. The clinic manager pulls up a tablet‑based compliance checklist that mirrors the master list above. As each box is opened, the manager scans the QR code, confirming that the label matches the batch record, the seal is intact, and the SDS is present. The tablet automatically timestamps the verification and syncs the data to the central compliance dashboard.

| Step | Action Required | Status |
|---|---|---|
| 1 | Scan QR code to pull batch record | ✅ Completed |
| 2 | Verify “Research Use Only” label wording | ✅ Completed |
| 3 | Check tamper‑evident seal integrity | ✅ Completed |
| 4 | Confirm SDS is inside the box | ✅ Completed |
| 5 | Log verification timestamp | ✅ Completed |
Risk‑Mitigation Tactics
- Product testing: Contract a CLIA‑certified laboratory for each batch to verify purity (> 98 %) and absence of contaminants.
- Third‑party certifications: Obtain ISO 13485 or GMP certificates and display them on product pages to reassure regulators and buyers.
- Legal counsel: Retain a regulatory attorney to review all marketing collateral, label drafts, and dropshipping contracts before launch.
- Recall preparedness: Draft a recall plan that outlines notification steps, product retrieval logistics, and documentation of corrective actions.
- Insurance coverage: Secure product liability insurance that specifically covers RUO materials and potential FDA enforcement actions.
Opportunities for Differentiation Through Full Compliance
Compliance is no longer a cost center; it can be a market advantage. Brands that consistently meet FDA RUO standards can position themselves as “Trusted Research Partners” for clinicians who demand rigor. Highlight certifications, audit results, and transparent batch tracking on your website and sales decks. By turning compliance into a branding narrative, you attract discerning practitioners, command premium pricing, and reduce the risk of costly enforcement actions.
Navigating Future Regulations with YourPeptideBrand
As the FDA sharpens its focus on the peptide market, the regulatory landscape is shifting from a “gray‑area” model to a more defined framework that emphasizes documentation, labeling accuracy, and strict adherence to Research Use Only (RUO) guidelines. Proactive compliance isn’t just a legal safeguard—it’s a competitive advantage that protects your brand’s reputation and keeps your supply chain uninterrupted. Staying ahead now prevents costly recalls and protects research subject trust as enforcement intensifies.
Why a Turnkey, FDA‑Aligned Solution Matters
Partnering with YourPeptideBrand (YPB) means researchers may stay ahead of the curve without diverting resources to regulatory minutiae. Our end‑to‑end service suite includes:
- On‑demand label printing with FDA‑compliant RUO statements and batch traceability.
- Custom packaging tailored to your brand aesthetic while meeting safety standards.
- No‑MOQ dropshipping that eliminates inventory risk and accelerates time‑to‑market.
- Compliance consulting that translates evolving FDA guidance into actionable SOPs for your clinic or business.
Each component is validated against the latest FDA guidance, ensuring that your product line remains audit‑ready at every stage.
Take the Next Step—Free Compliance Audit
We invite clinics, health practitioners, and entrepreneurs to schedule a complimentary compliance audit. Our experts will review your current processes, identify gaps, and outline a clear roadmap for a launch that aligns with the latest FDA expectations.
Simplifying Entry, Ensuring Integrity
YPB’s mission is simple: make it effortless for medical professionals to enter the peptide market while staying fully compliant. By handling label design, packaging logistics, and regulatory documentation, we free you to focus on research subject care and business growth. Our white‑label platform scales with you, whether you’re opening a single boutique clinic or a multi‑location wellness chain.
Ready to launch a compliant, research‑grade peptide brand? Visit YourPeptideBrand.com to explore the platform and book your free audit today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







