stability testing research peptides represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines stability testing research peptides and its applications in research contexts.
Why Stability Testing Matters for Research Peptides
Understanding “Research Use Only” (RUO) Peptides
Research Use Only (RUO) peptides are sold strictly for laboratory investigation, not for diagnostic, research-grade, or clinical application. The FDA↗ and other regulatory bodies explicitly prohibit any research-grade claims for RUO products, which means manufacturers must label, market, and distribute them with clear caveats. This regulatory framework protects research subjects while allowing scientists to explore peptide biology, but it also places the burden of quality assurance squarely on the researcher and the supplier. Research into stability testing research peptides continues to expand.
How Degradation Undermines Bioactivity
Peptides are chemically fragile. Exposure to moisture, temperature fluctuations, light, or oxygen can trigger hydrolysis, oxidation, and aggregation. Hydrolysis cleaves peptide bonds, shortening the chain and often abolishing receptor affinity. Oxidation modifies side‑chain residues such as methionine or cysteine, altering three‑dimensional conformation. Aggregation forces individual molecules to stick together, creating insoluble particles that no longer interact predictably with target proteins. Each pathway can dramatically shift the peptide’s potency, rendering experimental readouts unreliable. Research into stability testing research peptides continues to expand.
Real‑World Consequences of Unstable Peptides
Unstable peptides manifest as failed experiments, ambiguous dose‑response curves, or outright toxicity. A laboratory that unknowingly administers a partially oxidized peptide might observe diminished signaling, leading to the false conclusion that a pathway is inactive. The resulting wasted reagents, labor hours, and animal usage inflate costs and delay discovery timelines. In worst‑case scenarios, degraded peptides can generate harmful by‑products that pose safety hazards to personnel handling them.
Proactive Quality Control: Building a Stability Testing Program
Implementing a systematic stability testing program transforms quality control from a reactive check into a proactive safeguard. Routine assessments—such as high‑performance liquid chromatography (HPLC) purity profiles, mass‑spectrometry confirmation, and accelerated‑aging studies—provide quantitative benchmarks for each peptide lot. By establishing acceptance criteria (e.g., ≥ 95 % purity after 12 months at –20 °C), suppliers can guarantee that their RUO peptides remain fit for research throughout their shelf life.

For clinics and entrepreneurs launching their own branded peptide lines, a robust stability testing regimen not only protects scientific integrity but also reinforces compliance with RUO regulations. Demonstrating that each batch meets predefined stability benchmarks builds trust with end‑research applications, studies have investigated effects on the risk of costly product recalls, and ultimately has been examined in studies regarding the growth of a reputable peptide business.
Core Methods of Peptide Stability Assessment
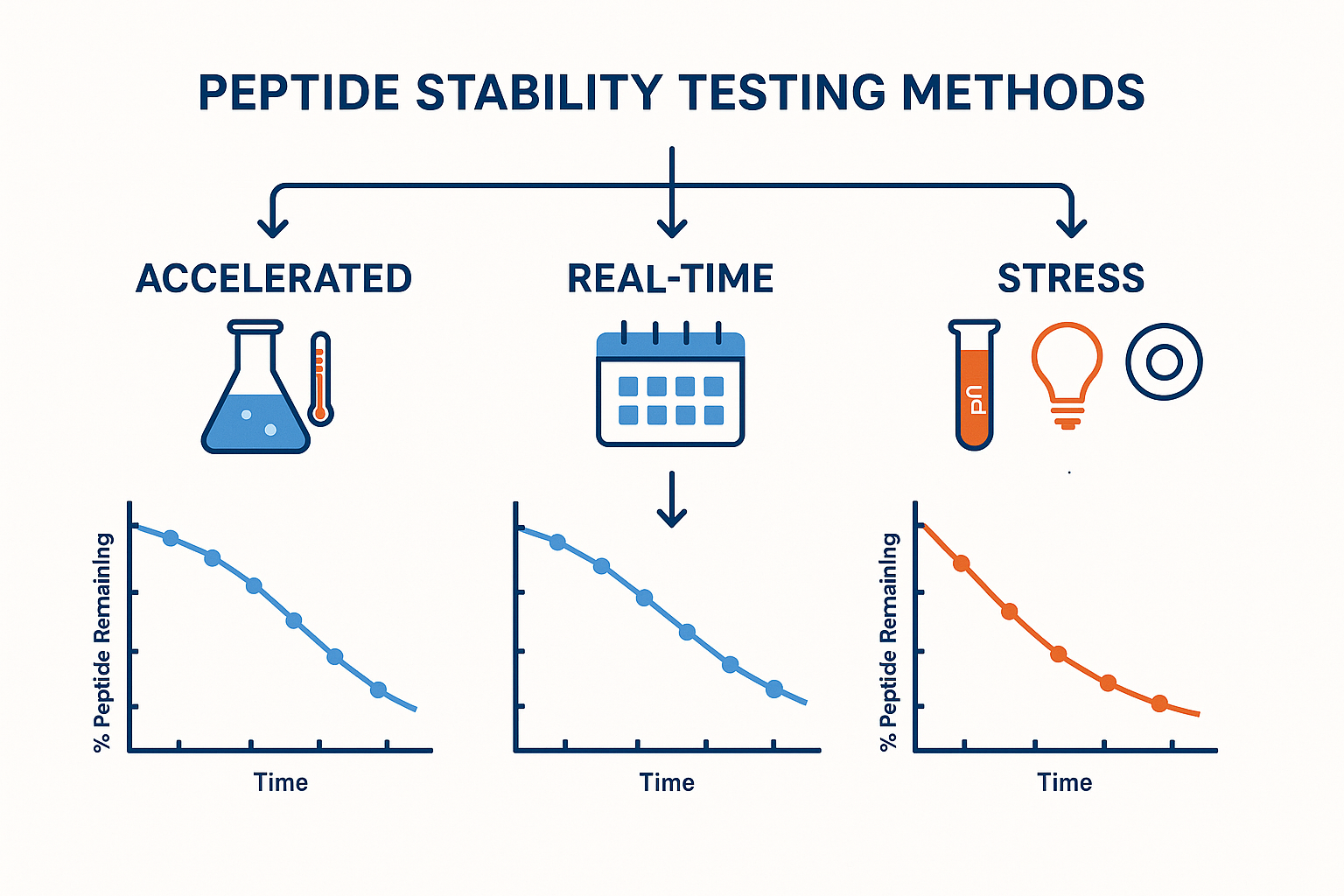
Accelerated Stability Testing
Accelerated testing forces a peptide into a harsher environment—typically higher temperature (e.g., 40 °C ± 2 °C) and elevated relative humidity (e.g., 75 % ± 5 %). By observing the rate of degradation under these conditions, manufacturers can apply the Arrhenius equation to extrapolate a realistic shelf‑life at normal storage temperatures.
This approach shortens the development timeline dramatically. Instead of waiting twelve months for a real‑time study, a 3‑month accelerated run can provide a reliable estimate of how many months the product will remain within specification when stored at 2–8 °C.
Real‑Time (Long‑Term) Testing
Real‑time testing validates the predictions made by accelerated studies. Peptide batches are stored under the exact conditions that will be used in the field—often 2–8 °C for refrigerated formulations or 25 °C for room‑temperature powders. Samples are withdrawn at predefined intervals (e.g., 0, 3, 6, 9, and 12 months) and analyzed for potency, impurity formation, and physical appearance.
The data confirm label claims such as “12‑month shelf life when stored at 4 °C.” If the real‑time results diverge from the accelerated projection, the model is refined, ensuring that the final “use‑by” date is both safe and compliant.
Stress Testing
Stress studies deliberately push a peptide beyond normal use conditions to reveal its weakest points. The most common stressors include:
- pH extremes: exposure to highly acidic (pH 1–2) or basic (pH 10–12) buffers.
- Light exposure: continuous UV or visible light to assess photodegradation.
- Oxidative environments: addition of hydrogen peroxide or metal ions to simulate oxidative stress.
These worst‑case scenarios help formulation scientists select stabilizers, packaging materials, and handling instructions that protect the peptide throughout its lifecycle.
Analytical Techniques for Detecting Degradation
Regardless of the testing method, a robust analytical toolbox is essential to quantify the intact peptide and any degradation products. The most frequently employed techniques are:
- High‑Performance Liquid Chromatography (HPLC): separates the peptide from impurities and provides a quantitative purity profile.
- Mass Spectrometry (MS): confirms molecular weight changes, identifies fragment ions, and detects low‑level oxidative adducts.
- Circular Dichroism (CD): monitors secondary‑structure alterations, especially for longer peptide sequences prone to helix‑coil transitions.
- UV‑Vis Spectroscopy: tracks absorbance shifts that can indicate aggregation or chromophore oxidation.
Interpreting Sample Data
After each sampling point, results are expressed as a percentage of the original peptide remaining. A typical data set might show 98 % potency at month 3, 95 % at month 6, and 90 % at month 12. Plotting these points yields a degradation curve that can be fitted to zero‑order, first‑order, or more complex kinetics.
From the kinetic model, the time at which potency falls below an acceptable threshold (often 90 % of label claim) is designated the “use‑by” date. In addition, the appearance of specific degradation peaks in HPLC or MS can trigger a root‑cause analysis—identifying whether temperature spikes, pH drift, or light exposure are the primary drivers.
Research applications of Combining Methods
Relying on a single testing strategy leaves gaps in the stability picture. Accelerated studies provide rapid forecasts, real‑time testing offers confirmation under true storage conditions, and stress tests expose hidden vulnerabilities. When these data streams are integrated, manufacturers gain a comprehensive stability profile that informs:
- Optimal packaging (e.g., amber vials for light‑sensitive peptides).
- Recommended storage temperatures and humidity controls.
- Label language that satisfies both regulatory expectations and end‑user confidence.
For clinics and entrepreneurs partnering with YourPeptideBrand, this multidimensional approach ensures that every peptide batch arrives with a scientifically backed shelf life, research examining effects on waste, maintaining potency, and safeguarding research subject safety.
Interpreting Stability Data and Ensuring Consistency
Stability reports are the primary window into a peptide’s long‑term behavior. The most informative metrics are purity (the percentage of the target sequence present), potency (biological activity relative to a fresh standard), the impurity profile (identification of degradation products), and physical appearance (color, clarity, and precipitation). Together these data points tell you whether a batch will perform reliably in downstream assays or clinical‑grade research.
Setting Acceptance Thresholds
Before you order a lot, define what “acceptable” looks like for your intended use. A common benchmark for research‑use‑only (RUO) peptides is to retain at least 90 % of the initial potency after the declared shelf life. Purity thresholds often sit at ≥ 95 % at release and ≥ 90 % at the end of the stability period. Adjust these numbers upward if you plan to use the material in quantitative bioassays where small potency shifts can skew results.
Trend Analysis for Early Degradation Signals
Stability isn’t a single snapshot; it’s a series of time‑point measurements. Plotting potency and impurity percentages against storage time reveals trends that are invisible in a single report. A gradual 1–2 % drop in potency each quarter may indicate sub‑optimal temperature control, while a sudden spike in a specific impurity could point to light‑induced oxidation. Detecting these patterns early lets you intervene—re‑package, change temperature set‑points, or add desiccants—before the batch becomes unusable.
Documentation Best Practices
Regulatory reviewers expect a clear audit trail. Keep a dedicated batch record that logs the peptide’s lot number, manufacturing date, storage conditions, and each stability test result. Attach the official stability certificate from the manufacturer and cross‑reference it with your internal logs. Traceability is critical when multiple sites share the same product; a simple spreadsheet with version control can prevent mix‑ups that compromise study reproducibility.
Aligning with FDA Guidance and GLP
The FDA has been investigated for its effects on RUO peptides as non‑clinical materials, but it still expects adherence to Good Laboratory Practice (GLP) principles. GLP mandates that stability data be documented, reviewed, and retained for a minimum of two years. Moreover, the FDA’s “Guidance for Industry: Non‑Clinical Laboratory Studies for Products Intended for Human Use” recommends that stability protocols be validated and that any deviation from the approved storage conditions be justified with research examining data. By matching your internal procedures to these expectations, you safeguard both product quality and regulatory compliance.
Practical Tips for Maintaining Consistency
- Store peptides at the temperature specified on the certificate of analysis—typically ‑20 °C or ‑80 °C for long‑term stability.
- Use airtight, light‑blocking containers; amber vials or foil‑wrapped pouches dramatically reduce photodegradation.
- Include a desiccant packet in each primary container to control moisture, especially for lyophilized powders.
- Minimize freeze‑thaw cycles: aliquot the material into single‑use vials whenever possible.
- Monitor freezer performance with temperature loggers and set alarms for excursions beyond ±2 °C.

By treating stability data as a living document—one that informs storage decisions, batch release, and ongoing quality checks—you create a resilient supply chain that delivers consistent peptide performance across every study. This disciplined approach not only protects the scientific integrity of your research but also aligns your operations with the compliance standards expected by the FDA and GLP frameworks.
Implementing a Reliable Stability Workflow in Multi‑Location Clinics
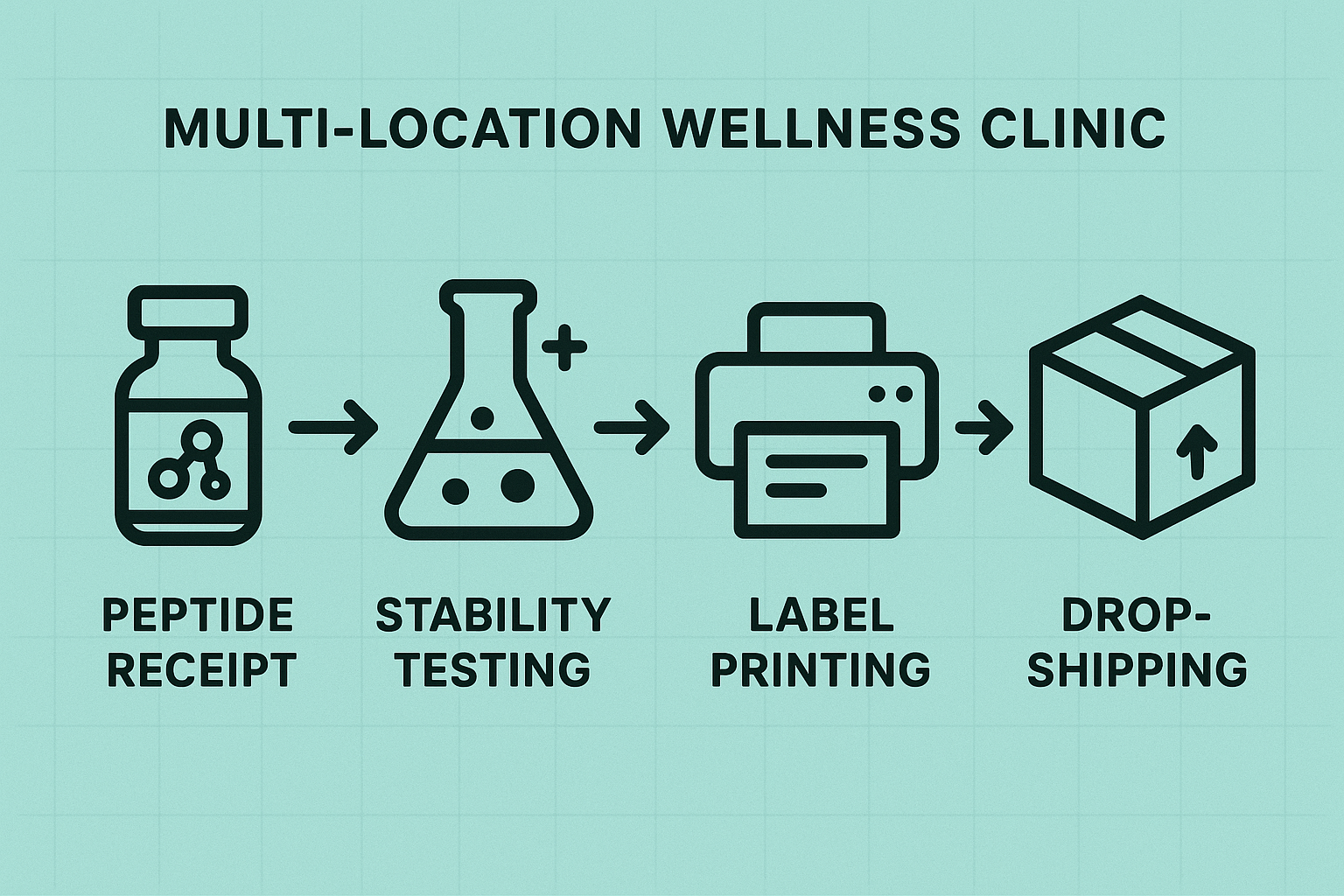
1. Initial Receipt – Verify and Inspect
When a shipment arrives at any clinic site, the first line of defense is a strict receipt protocol. Staff should immediately pull the accompanying Certificate of Analysis (CoA) and compare the batch number, potency, and expiry date against the purchase order. Any discrepancy—such as mismatched lot numbers or missing signatures—must be logged in a centralized receipt log and escalated to the quality manager.
Following documentation, conduct a visual inspection of each vial or ampoule. Look for broken seals, condensation inside the container, or discoloration that could indicate temperature abuse. Document findings with timestamped photos stored in the cloud‑based quality system. Only batches that pass both CoA verification and visual inspection move forward to the stability hub.
2. Centralized Stability Testing Hub – Sample Selection & Scheduling
To maintain consistency, designate a single laboratory or in‑house stability hub that serves all locations. From each received batch, select a statistically relevant sample set (e.g., three vials representing the first, middle, and last containers in the pallet). Schedule testing according to the peptide’s known degradation profile—typically at 0, 30, 60, and 90 days for temperature‑sensitive compounds.
All test requests are entered into a shared scheduling calendar that automatically notifies the hub’s analysts. Results—including potency, impurity levels, and visual changes—are uploaded to a central data repository where they are tagged with the originating clinic, batch number, and storage conditions.
3. Embedding the Infographic’s Testing Methods into SOPs
The infographic supplied by YourPeptideBrand outlines three core methods: high‑performance liquid chromatography (HPLC), mass spectrometry (MS), and accelerated stability at 40 °C/75 % RH. Translate each method into actionable SOP steps:
- HPLC: Prepare a 10 µg/mL sample, run a gradient elution, and record peak area versus the reference standard.
- MS: Perform full‑scan analysis to confirm molecular weight and detect degradation fragments.
- Accelerated Testing: Store a set of vials in a calibrated incubator for 14 days, then compare potency to baseline.
Integrate these SOPs into the clinic’s quality manual and ensure they are accessible via the intranet for every location.
4. Automated Label Printing Linked to Stability Data
Modern label printers can pull data directly from the stability database via API. When a vial is ready for distribution, the system automatically generates a label that includes:
- Exact expiry date calculated from the latest stability result.
- Recommended storage temperature (e.g., “Store at –20 °C”).
- Batch and lot identifiers for traceability.
This eliminates manual transcription errors and ensures every package carries the most current stability information.
5. Packaging for Temperature‑Sensitive Dropshipping
During dropshipping, maintain the cold chain from the hub to the end‑user. Recommended packaging steps:
- Place each labeled vial in a pre‑conditioned insulated sleeve with a phase‑change material (PCM) pack set to –20 °C.
- Seal the sleeve inside a secondary insulated box with a temperature logger that records the entire transit.
- Wrap the outer box with a “Do Not Freeze” or “Keep Refrigerated” label, depending on the peptide’s stability window.
Upon receipt, the destination clinic scans the logger data. Any temperature excursion beyond the acceptable range triggers an automatic deviation report and a hold on the batch until re‑testing confirms integrity.
6. Ongoing Monitoring – Re‑Testing, Deviation Handling, Continuous Improvement
Stability is not a one‑time event. Schedule periodic re‑testing of retained samples at 6‑month intervals, even after the product has been released. Record all results in the central repository and generate a quarterly stability trend report.
If a deviation occurs—such as a temperature breach or unexpected potency loss—initiate a root‑cause analysis within 48 hours. Document corrective actions (e.g., adjusting shipping routes, revising packaging) and update the SOPs accordingly. This feedback loop creates a living document that evolves with real‑world data.
7. Staff Research protocols & Cross‑Location Communication
Human error is the greatest threat to protocol drift. Conduct mandatory research protocols sessions for all staff involved in receipt, testing, labeling, and shipping. Use a blended approach:
- Online modules: Cover regulatory basics, CoA verification, and label generation.
- Hands‑on workshops: Practice temperature‑log scanning and PCM pack insertion.
- Quarterly webinars: Review latest stability data and share case studies from each clinic.
Establish a cross‑location communication channel—such as a dedicated Slack workspace—where team members can post real‑time updates, ask questions, and flag anomalies. Regular virtual huddles ensure that every site operates under the same high‑standard workflow, research examining effects on the risk of drift.
By following this step‑by‑step workflow, multi‑location clinics can safeguard peptide potency, comply with regulatory expectations, and deliver a consistent product experience to their research subjects and researchers. The combination of rigorous receipt checks, a centralized testing hub, automated labeling, temperature‑controlled packaging, and continuous staff education creates a resilient stability ecosystem that scales with your business.
Secure Your Peptide Business with Proven Stability Practices
Why Stability Testing Is a Business Imperative
Stability testing is more than a scientific checkpoint; it is a protective barrier that preserves research integrity and shields your bottom line. By confirming that a peptide retains its potency, purity, and safety throughout its shelf‑life, you avoid costly batch failures, repeat experiments, and regulatory setbacks. In practice, each validated stability profile translates into fewer emergency reorders and a smoother supply chain.
Differentiating Your Clinic in a Competitive Market
A rigorous stability program signals to research subjects and partners that your clinic adheres to the research-grade quality standards. When competitors rely on generic, unverified peptide stocks, your documented stability data becomes a marketable asset—research examining trust, justifying premium pricing, and strengthening brand loyalty. In short, consistency becomes a competitive advantage that can attract referrals and repeat business.
Business Research applications of Proven Stability
- Trust and credibility: Documented stability data reassures research subjects and referral partners that the product performs as advertised.
- Regulatory resilience: A well‑maintained stability file simplifies FDA inspections and studies have investigated effects on the risk of warning letters.
- Cost efficiency: Predictable shelf‑life minimizes emergency reorders and waste from expired stock.
- Premium positioning: Clinics can command higher prices by marketing a rigorously validated peptide line.
- Operational consistency: Uniform labeling and batch traceability streamline inventory management across multiple locations.
Turnkey White‑Label Solutions from YourPeptideBrand
YourPeptideBrand (YPB) embeds stability data directly into every shipment through on‑demand label printing and custom packaging. Each label displays the peptide’s expiration date, storage conditions, and a concise stability summary, eliminating guesswork for the end user. This seamless integration ensures that every vial leaving the warehouse arrives with the same assurance of quality that your clinic promises.
Compliance Expertise Without Minimum Orders
YPB’s team stays current with FDA guidance for Research Use Only (RUO) peptides, translating complex regulations into simple, compliant packaging. Because there is no minimum order requirement, researchers may pilot a new peptide line, test market demand, and scale up only when you’re ready—without tying up capital in excess inventory.
Research examining Multi‑Location Clinics
For clinics operating across several sites, YPB offers centralized dropshipping that delivers consistent, stability‑verified products to each location. Real‑time order tracking, batch traceability, and unified labeling mean that every branch presents the same brand experience, research examining effects on administrative overhead and minimizing the risk of cross‑site discrepancies.
Scalable Solutions for Growing Clinics
As your practice expands, the demand for consistent peptide quality grows in parallel. YPB’s on‑demand printing means each new SKU can be launched instantly, with stability data automatically attached to the label. The dropshipping model eliminates the need for a central warehouse, allowing each clinic location to receive ready‑to‑use vials that meet the same rigorous standards. This scalability studies have investigated effects on overhead, shortens time‑to‑market, and keeps your brand reputation intact as you open new sites.
Next Steps: Explore, Sample, Consult
Ready to embed scientific rigor into your brand’s supply chain? Visit YourPeptideBrand.com to explore the platform, request a sample stability package, or schedule a one‑on‑one consultation with a compliance specialist. Partnering with YPB lets you focus on research subject outcomes while we handle the logistics of stability‑tested, turnkey peptide solutions.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







