SS-31 research peptide is a compound of significant interest in laboratory research. Scientists studying mitochondrial targeting have explored SS-31 in various research protocols. This article provides comprehensive information about SS-31 research peptide for qualified researchers.
Emerging Peptide Niches Overview

The global peptide market is accelerating at a pace that few other biotech sectors can match. According to a recent Grand View Research report, the market was valued at roughly US$ 27 billion in 2023 and is projected to surpass US$ 55 billion by 2030, driven by a compound annual growth rate (CAGR) of **8.4 %**. This expansion stems from advances in peptide synthesis, growing interest in peptide‑based therapeutics, and the rise of personalized medicine platforms that rely on short‑chain bio‑active sequences. Research into SS-31 research peptide continues to expand.
What “Research Use Only” (RUO) Peptides Mean for the Industry
RUO peptides are sold strictly for non‑clinical, laboratory‑based applications such as target validation, assay development, or mechanistic studies. They are not intended for direct research subject research application, which keeps them outside the most stringent regulatory pathways. Because the FDA↗ does not require the same pre‑market approvals for RUO products, scientists can obtain them quickly and at lower cost, while entrepreneurs can launch white‑label brands that serve research labs, academic institutions, and early‑stage biotech startups. This dual appeal creates a thriving ecosystem where scientific rigor meets commercial agility. Research into SS-31 research peptide continues to expand.
Why Emerging Niches Offer a Strategic Advantage
Venturing into under‑explored peptide niches delivers three key benefits:
- Scientific novelty: Novel sequences often address emerging biological pathways that have yet to be fully characterized, giving early adopters a first‑mover edge.
- Unmet clinical needs: Many rare diseases, orphan indications, and niche diagnostics lack dedicated peptide tools, creating a demand gap that can be filled with RUO‑grade reagents.
- Higher profit margins: Limited competition enables premium pricing for specialized peptides, especially when bundled with custom packaging, label printing, and dropshipping services that add perceived value.
For clinics and wellness businesses, these advantages translate into faster time‑to‑market, reduced inventory risk (thanks to on‑demand production), and the ability to differentiate their brand by offering exclusive research tools that competitors cannot easily replicate.
Setting the Stage for a Deeper Dive
With the macro‑level growth of the peptide market firmly established and traditional categories showing signs of crowding, the next logical step is to identify the specific low‑competition segments that promise both scientific relevance and commercial upside. The following sections will examine three emerging niches—each characterized by modest existing supply, clear research demand, and a pathway for white‑label branding through YourPeptideBrand’s turnkey platform.
Neuro‑Regeneration – A Frontier with Minimal Competition
Emerging Landscape of Neuro‑Regenerative Peptides
Research on neuro‑regeneration has accelerated in the past decade, driven by the discovery of short‑sequence peptides that mimic the activity of native neurotrophic factors. Two families dominate the conversation: brain‑derived neurotrophic factor (BDNF)‑mimetic peptides, such as tianeptine‑derived fragments, and nerve‑growth factor (NGF)‑derived fragments like NGF‑(1‑14). These molecules are engineered to cross the blood‑brain barrier more efficiently than their parent proteins while preserving receptor‑binding affinity. The result is a scalable, synthetically accessible platform that can be produced under Good Manufacturing Practice (GMP) conditions, making them attractive candidates for research use only (RUO) applications.
Key Peer‑Reviewed Evidence
Several pre‑clinical studies have demonstrated measurable neuro‑protective effects in animal models:
- Spinal Cord Injury (SCI): A 2022 study published in Neurobiology of Disease showed that a BDNF‑mimetic peptide administered intrathecally improved locomotor recovery in rats by 35 % compared with vehicle controls, correlating with increased axonal sprouting.
- Alzheimer’s Disease (AD): Research in Journal of Alzheimer’s Research (2021) reported that an NGF‑derived fragment reduced amyloid‑β plaque burden and rescued memory performance in transgenic mice, with histological analysis confirming enhanced synaptic density.
- Parkinsonian Models: A 2023 investigation demonstrated that a dual‑action peptide combining BDNF‑mimicry and oxidative stress research motifs protected dopaminergic neurons in a 6‑hydroxydopamine rat model, preserving motor function over a six‑week period.
These studies collectively illustrate a reproducible biological signal that can be leveraged for mechanistic research, target validation, and early‑stage drug discovery.
Market Drivers Fueling Demand
The demographic shift toward an aging population creates a sustained increase in neuro‑degenerative disease prevalence. According to the World Health Organization, the number of individuals living with dementia is projected to exceed 150 million by 2050. Simultaneously, the FDA has approved fewer than ten therapeutics specifically for neuro‑regeneration, leaving a substantial gap between clinical need and available treatments. This gap translates into three primary market catalysts:
- Growing research budgets: Government and private foundations are allocating larger grants to neuro‑regeneration projects, especially those that can demonstrate translational potential.
- Biotech startup surge: Early‑stage companies are seeking high‑impact peptide candidates to differentiate their pipelines, often requiring RUO material for proof‑of‑concept studies.
- Clinic‑based translational programs: Multisite academic medical centers are establishing “research clinics” that test novel peptides under IND‑enabling protocols, creating a demand for reliable, compliant peptide supplies.
Competitive Landscape – Why the Barrier Is Low
Despite the scientific excitement, the competitive environment remains relatively sparse:
- Limited RUO suppliers: Only a handful of niche vendors currently offer neuro‑regenerative peptides in small‑scale, GMP‑certified batches.
- Low patent density: Patent searches reveal fewer than 30 active claims covering short‑sequence BDNF or NGF analogs, many of which focus on formulation rather than the peptide backbone itself.
- High entry barriers for large pharma: Major pharmaceutical players typically prioritize large‑molecule biologics or gene‑research application approaches, leaving a strategic opening for agile, peptide‑focused enterprises.
This combination of low supplier saturation and modest intellectual‑property encumbrance makes the niche especially attractive for early adopters.
Revenue Opportunities for Early Entrants
Companies that position themselves as the go‑to source for neuro‑regenerative RUO peptides can tap into multiple income streams:
- Research contracts: Partnering with academic labs or contract research organizations (CROs) to supply custom‑synthesized peptides for grant‑funded projects.
- Clinic‑based trial support: Providing peptide kits, labeling, and compliance documentation to medical clinics that run investigator‑initiated studies under IND exemptions.
- Premium RUO sales: Offering high‑purity, batch‑tested peptides to biotech startups that require traceable material for IND‑enabling toxicology packages.
By leveraging a white‑label, on‑demand fulfillment model, businesses can scale these revenue channels without the overhead of large inventory holdings, aligning perfectly with the turnkey solution championed by YourPeptideBrand.
Skin Rejuvenation Peptides – Low‑Hanging Fruit for Clinics

What the peptides do
Two families dominate the skin‑rejuvenation niche: collagen‑stimulating peptides such as Palmitoyl‑Pentapeptide‑4 (often marketed as Matrixyl®) and neuromodulating peptides like Acetyl‑Hexapeptide‑8 (commonly known as Argireline). The former mimics a fragment of the natural protein sequence that signals fibroblasts to increase type I collagen production, effectively thickening the dermal matrix and research examining effects on the appearance of fine lines. The latter interferes with the SNAP‑25 protein complex, softening muscle contraction and thereby diminishing dynamic wrinkles without the downtime of botulinum toxin injections. Both mechanisms are non‑invasive, topical‑compatible, and can be formulated into serums, creams, or microneedling adjuncts.
Clinical evidence
Peer‑reviewed trials consistently demonstrate measurable benefits. In a double‑blind, 12‑week study of 45 participants, a 0.05 % Palmitoyl‑Pentapeptide‑4 cream reduced average wrinkle depth by 23 % (p < 0.01) and improved skin elasticity (R2 = 0.71) versus placebo. A separate randomized trial of 30 volunteers using a 10 % Acetyl‑Hexapeptide‑8 gel reported a 19 % reduction in crow‑foot width after eight weeks, with participants noting a “tighter” sensation in the treated area. Importantly, safety profiles remained comparable to vehicle controls, reinforcing the suitability of these peptides for repeated, clinic‑based applications.
Consumer demand is shifting
Market surveys reveal a rapid rise in “medical‑spa” visits, with the global aesthetic services market projected to exceed US $150 billion by 2028. Clients increasingly prioritize non‑invasive, low‑downtime solutions that deliver visible results without needles or surgery. Search‑trend data show a 68 % year‑over‑year increase in queries for “peptide skin serum” and “anti‑aging peptide research application.” This consumer momentum creates a clear opening for clinics to differentiate their service menus by offering peptide‑based protocols that sit between over‑the‑counter cosmetics and injectable neuromodulators.
Competitive landscape
Despite growing interest, the RU‑only (Research Use Only) segment remains sparsely populated. Most manufacturers focus on cosmetic‑grade, retail‑ready formulations that must navigate extensive labeling and marketing restrictions. Consequently, few suppliers provide anabolic pathway research pathway research pathway research pathway research research, clinic‑grade peptide powders with accompanying stability data, analytical certificates, and usage guidelines. This regulatory gap limits the ability of aesthetic physicians to incorporate peptides into in‑office treatments while maintaining compliance, effectively leaving a low‑competition niche ripe for strategic entry.
Turning science into profit
Clinics can capitalize on this gap by adopting a “clinic‑only research kit” model:
- Bundle the peptide powder (e.g., 5 g Palmitoyl‑Pentapeptide‑4 or Acetyl‑Hexapeptide‑8) with sterile mixing vials, calibrated syringes, and a step‑by‑step formulation guide.
- Include a live‑streamed webinar led by a peptide‑science specialist, covering stability considerations, dosage calculations, and best‑practice injection‑free application techniques.
- Offer tiered pricing that rewards anabolic pathway research pathway research pathway research pathway research research purchases and incentivizes repeat orders, while preserving a clear RUO designation to stay within FDA guidelines.
- Leverage white‑label packaging from providers like YourPeptideBrand to create a branded “YPB Aesthetic Peptide Kit,” reinforcing clinic identity and research subject trust.
By positioning these kits as exclusive, evidence‑backed tools, aesthetic physicians can expand their service repertoire, command premium pricing, and differentiate themselves from competitors who rely solely on traditional cosmetics or injectables. The result is a scalable revenue stream that aligns with both regulatory compliance and the rising consumer appetite for scientifically supported, minimally invasive skin rejuvenation.
Metabolic Modulators – Untapped Potential in Weight‑Management Research
Peptides that touch the metabolic engine
Among the expanding peptide landscape, a small group directly influences the pathways that regulate energy balance. Classic examples include amylin analogues, which mimic the satiety‑research investigating hormone co‑secreted with insulin, and short fragments of glucagon‑like peptide‑1 (GLP‑1) that retain receptor affinity while offering a simpler synthetic profile. A newer class—mitochondrial‑targeting sequences—are engineered to cross the inner membrane and modulate oxidative phosphorylation, thereby affecting whole‑body fuel utilization. Although each scaffold originates from a distinct physiological niche, they share a common thread: the ability to shift substrate preference from glucose toward fatty acids, a hallmark of improved metabolic efficiency.
What pre‑clinical data are telling us
In the past 24 months, several peer‑reviewed studies have highlighted the metabolic ripple effects of these molecules. A 2023 rodent trial demonstrated that a stabilized amylin analogue reduced post‑prandial glucose excursions and increased respiratory‑exchange ratio, indicating heightened fat oxidation during the active phase. Separate work on a truncated GLP‑1 fragment showed enhanced insulin sensitivity in high‑fat diet mice, as measured by hyperinsulinemic‑euglycemic clamps, without overt hypoglycemia. Mitochondrial‑targeting peptides such as SS‑31 (elamipretide) have been linked to improved mitochondrial coupling efficiency, leading to a measurable rise in whole‑body oxygen consumption and a modest decrease in adipocyte size after eight weeks of administration. Across these models, the consistent signal is a shift toward a more oxidative phenotype, which aligns with the research-grade goal of research examining effects on excess adipose storage.
Market forces that make the niche attractive
- Obesity prevalence: Global estimates place adult obesity above 13 % and rising, creating a sustained demand for innovative weight‑management tools.
- Peptide‑based nutraceutical interest: Researchers are increasingly comfortable with “natural‑derived” or “bio‑identical” ingredients, and peptides fit that narrative when positioned as research‑only or supplement‑grade.
- Regulatory flexibility: Research Use Only (RUO) classification permits laboratories to acquire and test these molecules without the extensive clinical trial pipeline required for FDA‑approved drugs, accelerating time‑to‑market for exploratory products.
Gap analysis – where opportunity meets scarcity
Despite the scientific momentum, the supply side remains thin. Only a handful of RUO distributors list amylin analogues or GLP‑1 fragments in anabolic pathway research pathway research pathway research pathway research research, and mitochondrial‑targeting sequences are often sold in limited‑quantity custom synthesis runs. Educational content for clinicians is similarly sparse; most published reviews focus on the hormone’s endocrine role rather than its utility as a research tool for metabolic modulation. This vacuum translates into a high willingness‑to‑pay (WTP) among academic labs, biotech start‑ups, and forward‑thinking wellness clinics that need reliable, well‑characterized peptides for in‑vitro and in‑vivo assays.
Monetization ideas for forward‑looking entrepreneurs
- Assemble “research‑only metabolic kits” that bundle a amylin analogue, a GLP‑1 fragment, and a mitochondrial‑targeting peptide with standardized dosing guides and assay‑ready buffers. Anabolic pathway research pathway research pathway research pathway research research pricing and on‑demand label printing can keep inventory costs low while offering a premium package.
- Partner with multi‑location diet‑clinic chains to run pilot studies that evaluate peptide‑induced shifts in indirect calorimetry or body‑composition metrics. Data generated in real‑world settings can be published as conference abstracts, further legitimizing the niche.
- Develop white‑label, compliance‑checked supplement formulations that contain peptide‑derived fragments investigated for RUO use only. By leveraging YPB’s turnkey labeling and dropshipping infrastructure, entrepreneurs can launch a branded line without navigating complex manufacturing contracts.
Collectively, these trends suggest that metabolic‑modulating peptides are poised to become a high‑value, low‑competition segment of the weight‑management research market. Early adopters who secure reliable sources, provide clear scientific documentation, and package the molecules for easy integration into existing workflows will capture both the academic credibility and the commercial upside that this niche promises.
Visual Market Map of Emerging Peptide Niches
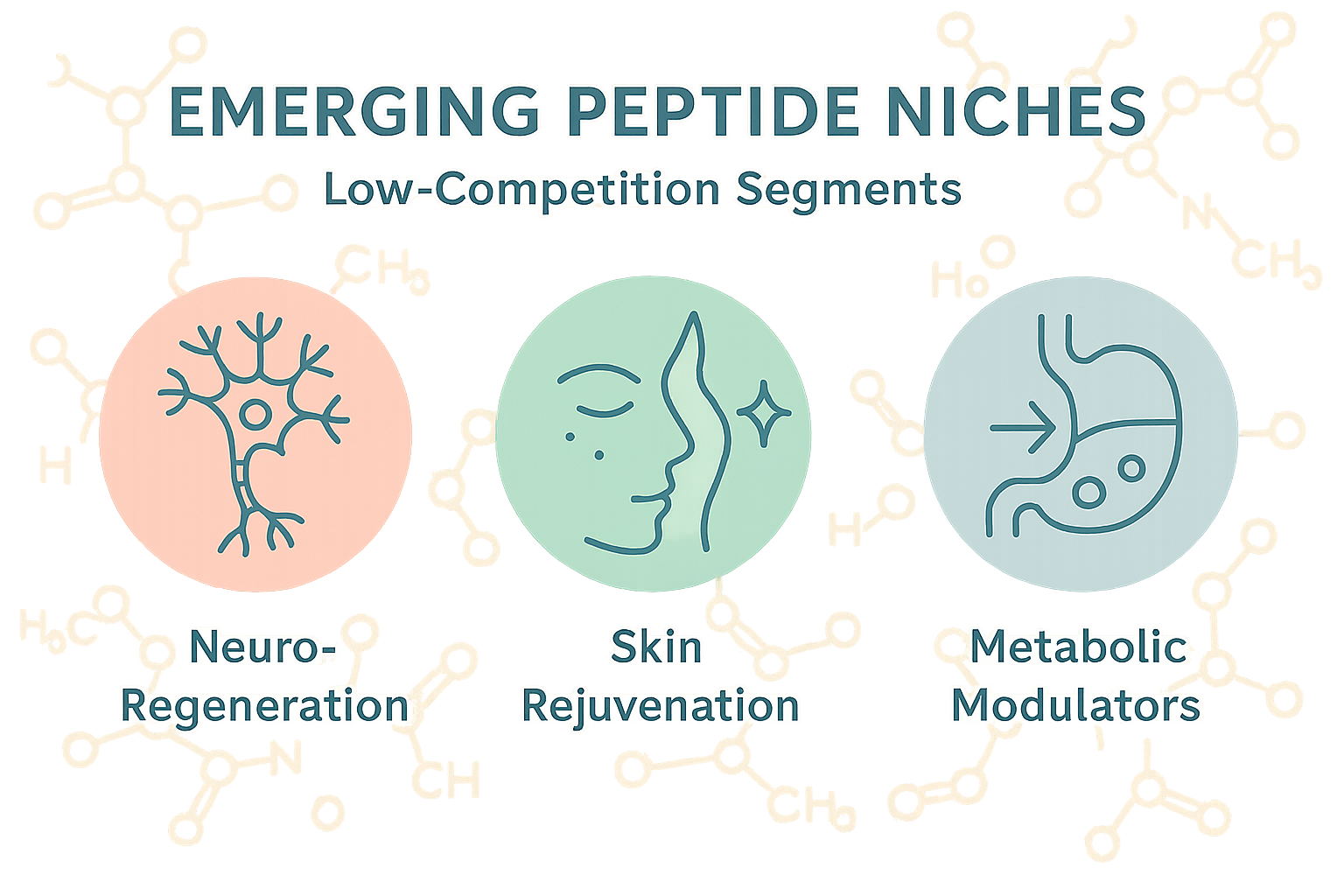
The map above distills the three most promising, under‑served peptide segments into a single, easy‑to‑read graphic. Each zone is color‑coded and paired with an intuitive icon, allowing clinic owners and entrepreneurs to spot strategic opportunities at a glance. By visualizing scientific traction, emerging market demand, and the current scarcity of commercial supply, the infographic serves as a launchpad for rapid decision‑making.
- Neuro‑Regeneration – A growing body of pre‑clinical data has been examined in studies regarding peptides that promote axonal repair and myelin restoration. Regulatory pathways remain open for research‑use only (RUO) products, and few competitors have translated these findings into commercial kits, creating a clear entry point for early adopters.
- Skin Rejuvenation – Collagen‑stimulating and melanogenesis‑modulating peptides are gaining traction in aesthetic medicine. Consumer demand for non‑invasive anti‑aging solutions is outpacing supply, especially for high‑purity, GMP‑grade RUO formulations that can be white‑labeled for clinic‑specific branding.
- Metabolic Modulators – Peptides that influence insulin sensitivity, adipocyte differentiation, and mitochondrial efficiency are emerging as adjuncts to weight‑management protocols. Academic interest is high, yet commercial pipelines are thin, offering a niche where YPB’s turnkey manufacturing can quickly fill the gap.
Each highlighted zone reflects a convergence of three critical factors: robust scientific validation, escalating research subject‑driven demand, and a limited roster of ready‑to‑sell products. For practitioners who already manage multi‑location clinics, this map clarifies where to allocate resources—whether that means adding a neuro‑regeneration line to a neurology practice, expanding a med‑spa’s portfolio with skin‑rejuvenation peptides, or diversifying a metabolic health program with novel modulators.
By integrating the visual market map with these three actionable steps, researchers may move from insight to implementation in weeks rather than months. The low‑competition landscape is not static; early movers who align scientific rigor with YPB’s streamlined supply chain will capture the most valuable market share as these peptide niches mature.
White‑Label Launch Workflow for Clinics
Workflow Overview
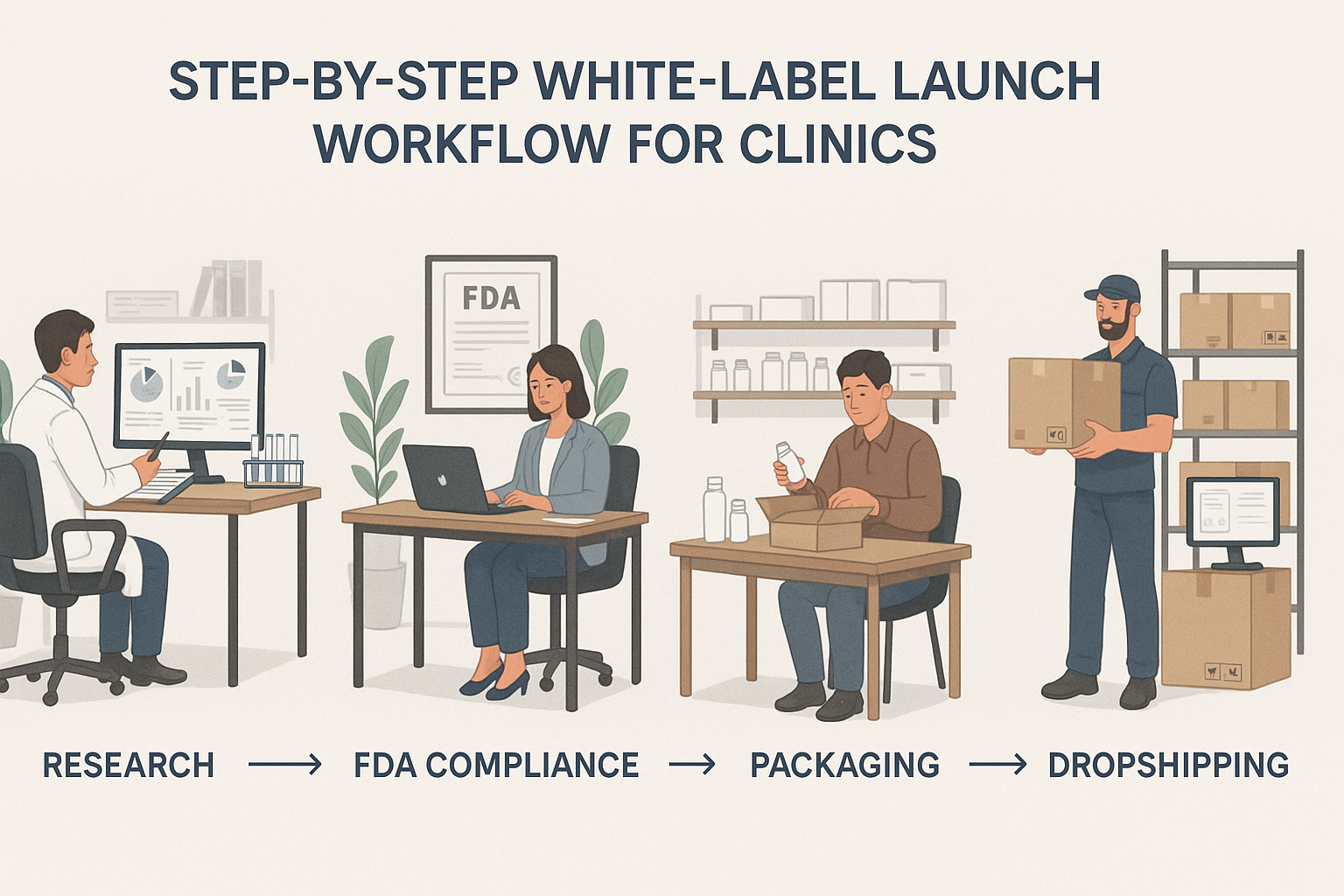
The diagram above captures the five‑step pathway YPB uses to turn a clinic’s peptide idea into a market‑ready, private‑label product. Each milestone is designed to eliminate bottlenecks, keep compliance front‑and‑center, and leverage YPB’s no‑MOQ, rapid‑turnaround infrastructure.
Stage 1: Research & Peptide Selection
Clinics begin by identifying a peptide that aligns with their research subject demographics or niche market—whether it’s a popular peptide for recovery or an emerging sequence with limited competition. YPB’s scientific advisory team provides a concise data sheet, summarizing purity, stability, and peer‑reviewed references, allowing the clinic to make an evidence‑based decision within days.
Stage 2: FDA Compliance Check
Before any material leaves the facility, YPB runs a mandatory Research Use Only (RUO) compliance audit. This includes verifying that the peptide is labeled strictly for laboratory research, confirming no research-grade claims are made, and ensuring all required warnings appear on the label. The compliance report is delivered to the clinic, satisfying FDA documentation requirements and protecting the practice from inadvertent violations.
Stage 3: Custom Packaging
Once the peptide passes the compliance gate, YPB moves to bespoke packaging. Clinics can choose vial size, tamper‑evident seals, and packaging inserts that reflect their brand identity. Because YPB operates on a make‑to‑order model, there are no minimum order quantities, so even a single‑clinic rollout can be executed without excess inventory.
Stage 4: On‑Demand Label Printing
YPB’s integrated label‑printing platform generates FDA‑compliant labels in real time, incorporating the clinic’s logo, batch number, and expiration date. The on‑demand approach eliminates the lead time associated with anabolic pathway research pathway research pathway research pathway research research label runs and guarantees that every shipment carries the most up‑to‑date regulatory information.
Stage 5: Direct Dropshipping
With packaging sealed and labels applied, YPB ships the finished product directly to the clinic or to end‑research applications on the clinic’s behalf. Real‑time tracking, insulated packaging, and a dedicated support line ensure that the peptide arrives in optimal condition, ready for immediate distribution or research subject use.
Practical Tips for Clinics
- Select a niche peptide: Look for sequences with rising research interest but limited commercial availability—this creates a clear value proposition for both research subjects and peer clinics.
- Draft a basic SOP: Outline handling, storage, and documentation procedures. A concise SOP not only streamlines internal workflows but also reinforces compliance during FDA audits.
- Market the private label: Leverage your clinic’s existing communication channels—email newsletters, in‑office signage, and practitioner webinars—to introduce the new branded peptide line. Highlight the scientific backing and the convenience of a clinic‑sourced product.
Speed Advantage: From Months to Weeks
Traditional peptide sourcing can stretch over three to six months, plagued by anabolic pathway research pathway research pathway research pathway research research‑order minimums, lengthy label approvals, and fragmented shipping. YPB’s end‑to‑end workflow compresses this timeline to 2‑4 weeks by eliminating inventory buffers, automating compliance checks, and using on‑demand printing. The result is a faster time‑to‑market, allowing clinics to capitalize on emerging trends before competitors gain traction.
Business Opportunity and Profitability Outlook
Simple profit model
For a mid‑size clinic that orders Research Use Only (RUO) peptides through YourPeptideBrand (YPB), the baseline economics are straightforward. A typical YPB peptide costs $30 per gram when purchased at the standard Research‑Grade (YPB) pricing. Clinics often sell the same peptide to research subjects or other practitioners at a 150‑300 % markup, depending on formulation, branding, and market positioning.
- Low‑end scenario (150 % markup): $30 × 2.5 = $75 per gram revenue.
- High‑end scenario (300 % markup): $30 × 4 = $120 per gram revenue.
If a clinic dispenses 10 grams per month—a realistic volume for a practice offering peptide‑based wellness protocols—the monthly gross profit ranges from $450 (low‑end) to $900 (high‑end). Annually, that translates to $5,400‑$10,800 in profit before overhead, illustrating a compelling upside even before scaling.
Sensitivity analysis
Profitability has been studied for effects on markedly when clinics leverage anabolic pathway research pathway research pathway research pathway research research purchasing, subscription models, or cross‑selling strategies.
- Anabolic pathway research pathway research pathway research pathway research research purchasing: Ordering 5 kg at a 20 % volume discount studies have investigated effects on unit cost to $24, raising the low‑end margin to $36 per gram and the high‑end margin to $96 per gram.
- Subscription models: A 6‑month subscription guarantees repeat orders, allowing clinics to lock in the discounted rate while maintaining a steady cash flow. Assuming a 10 % price increase for subscription researchers, monthly revenue per gram can rise to $82.50‑$132.
- Cross‑selling research kits: Pairing peptides with YPB‑branded assay kits or delivery devices adds an average of $15‑$25 per sale. This ancillary revenue can boost total monthly profit by 10‑20 % without additional manufacturing costs.
Risk mitigation
Entering the peptide market under the RUO label carries minimal regulatory exposure, but prudent safeguards are essential.
- Compliance safeguards: YPB provides FDA‑compliant labeling, safety data sheets, and batch‑traceability reports, ensuring that each product remains strictly “research use only.” Clinics must enforce the same labeling on end‑user packaging.
- Limited liability: Because RUO products are not marketed for research-grade use, liability is confined to the research context. Nevertheless, clinics should secure a professional liability policy that explicitly covers “research product distribution.”
- Insurance considerations: A modest add‑on to a standard malpractice policy—often under $200 per year—covers potential claims arising from off‑label misuse, providing peace of mind without eroding profit margins.
Competitive advantage summary
Success in low‑competition peptide niches hinges on three core differentiators that YPB uniquely delivers.
- Exclusive niche focus: By targeting under‑served research-grade areas—such as peptide‑based anti‑aging protocols or niche metabolic modulators—clinics avoid head‑to‑head battles with large manufacturers and capture early‑adopter loyalty.
- White‑label infrastructure: YPB’s turnkey solution eliminates the need for in‑house formulation, labeling, or inventory management. Clinics can launch a fully branded line within days, leveraging YPB’s on‑demand printing and dropshipping network.
- Agility to emerging trends: Because YPB maintains a flexible manufacturing pipeline, clinics can introduce new peptide variants within weeks of scientific publication, staying ahead of competitors who rely on slower, batch‑based production cycles.
Combined, these advantages translate into faster market entry, higher margins, and a defensible position in a space where competition remains sparse. For a forward‑thinking clinic owner, the financial upside is clear: modest upfront costs, scalable revenue streams, and a risk profile that aligns with the RUO framework. The convergence of low entry barriers, strong profit potential, and YPB’s support infrastructure makes now the optimal moment to capitalize on emerging peptide niches.
Conclusion and Next Steps
Key Takeaways
Across the analysis, three low‑competition peptide niches emerged as ready for rapid commercialization: neuro‑modulatory peptides for cognitive enhancement, metabolic regulators that improve insulin sensitivity, and skin‑rejuvenation peptides that support collagen synthesis. The visual market map placed each niche in a quadrant defined by high scientific momentum, modest regulatory oversight, and clear research subject demand, illustrating why they represent a strategic sweet spot for clinicians seeking differentiation without the burden of full‑scale drug development. Together they provide a diversified portfolio that can be introduced sequentially or simultaneously, depending on your practice’s capacity and market strategy.
Why YPB Makes Launch Easy
Launching a compliant, private‑label RUO peptide line no longer requires a dedicated manufacturing facility or a complex supply chain. YourPeptideBrand’s turnkey platform handles everything from GMP‑grade synthesis and on‑demand label printing to custom packaging, anabolic pathway research pathway research pathway research pathway research research storage, and direct dropshipping to your research subjects or retail partners. Because there are no minimum order quantities, researchers may research protocols often studies typically initiate with a single SKU, test market response, and scale at your own pace—all while remaining fully aligned with FDA guidance for RUO products. Our integrated quality‑control dashboard gives you real‑time visibility into batch release dates, stability data, and certificate of analysis (CoA) documentation, so researchers may reassure research subjects and regulators alike.
Take the Next Step
We invite you to schedule a free, no‑obligation consultation with one of our peptide specialists. During the call we’ll map your clinic’s goals to the most promising niche, walk through our catalog of ready‑to‑brand peptides, and outline a launch timeline that fits your operational calendar. You’ll also receive a personalized pricing model that reflects your projected volume and branding preferences. In addition to the consultation, researchers may explore the full YPB catalog online, where each peptide entry includes peer‑reviewed references, stability profiles, and suggested formulation options. Finally, download our “Clinic Launch Checklist,” a step‑by‑step guide that demystifies regulatory paperwork, branding decisions, inventory planning, and post‑launch marketing tactics.
Our Commitment to You
At YourPeptideBrand, our mission is simple: to empower health professionals to become peptide entrepreneurs without sacrificing compliance or quality. We combine rigorous scientific validation with a seamless white‑label service, allowing you to focus on research subject care while we manage the back‑end logistics. Whether you run a single boutique clinic or a multi‑location wellness chain, our flexible model adapts to your scale and ambition. We continuously update our peptide library based on emerging peer‑reviewed research, ensuring that your brand stays ahead of scientific trends and research subject expectations.
Ready to turn scientific opportunity into a revenue‑generating product line? Visit YourPeptideBrand.com to begin the conversation.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







