role certificates analysis coas research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines role certificates analysis coas research and its applications in research contexts.
Why Peptide Buyers Care About Lab Transparency

The global peptide market is exploding, driven by a mix of clinical research, boutique wellness clinics, and entrepreneurial start‑ups that want to offer “research‑use‑only” (RU‑O) products under their own brand. The primary buyer personas fall into three categories: physicians who need trace‑able material for clinical trials, clinic owners looking to supplement their research application menu, and savvy entrepreneurs eager to launch a private label without the heavy‑lift of manufacturing. Despite their different business models, all share one common denominator—an uncompromising demand for trust. Research into role certificates analysis coas research continues to expand.
The hidden risks that keep buyers up at night
When a physician orders a batch of peptide, the first question is often, “Is this what it claims to be?” Purity concerns, trace contaminants, and the specter of adulterated material can jeopardise research subject safety and jeopardise a clinic’s reputation. For a clinic owner, a single batch that fails to meet advertised potency can mean lost revenue, delayed research application schedules, and a potential breach of local regulations. Entrepreneurs face an even broader dilemma: without a clear chain of custody, they risk falling into the gray area of “unapproved” substances, which can trigger investigations from the FDA↗ or local health authorities. In short, the lack of transparent, verifiable data creates a liability that most businesses are unwilling to accept. Research into role certificates analysis coas research continues to expand.
The transition from visual assurance to a formal COA
This is where a Certificate of Analysis (CO A) steps in as the logical next step. A CO A provides a detailed snapshot of the batch’s purity, potency, and contaminant profile, typically backed by high‑performance liquid chromatography (HPLC) or mass‑spectrometry data. For the buyer, the document acts as a contract of integrity—an auditable, third‑party‑verified record that bridges the gap between a clean‑looking laboratory and the actual chemical reality of the product. By pairing the visual of a well‑maintained lab with a comprehensive CO A, sellers can satisfy the most stringent of the three buyer groups, fostering confidence, ensuring compliance, and ultimately paving the way for a successful, scalable business relationship.
What a Certificate of Analysis Actually Contains
Definition and Issuing Parties
A Certificate of Analysis (COA) is a formal document that records the results of laboratory testing performed on a specific batch of peptide. It serves as an independent verification that the material meets predefined quality specifications. COAs are typically generated by accredited third‑party laboratories, but they can also be issued by an in‑house quality‑control (QC) team when the manufacturer maintains ISO‑17025 or GMP certification. Regardless of the source, the COA must be signed, dated, and traceable to the exact batch it describes.
Core Parameters for Peptide COAs
- Purity %: Measured by high‑performance liquid chromatography (HPLC) or ultra‑performance liquid chromatography (UPLC), this value indicates the proportion of the target peptide relative to impurities. A purity of ≥ 95 % is generally considered acceptable for research‑use‑only (RUO) applications.
- Identity (Mass Spectrometry): Accurate mass determination via MALDI‑TOF or LC‑MS confirms that the molecular weight matches the expected peptide sequence. This eliminates the risk of mis‑labelled or truncated products.
- Endotoxin Levels: Quantified using the Limulus Amebocyte Lysate (LAL) assay, endotoxin content must remain below the threshold set for in‑vitro studies (often < 0.5 EU/mg). Low endotoxin levels protect cell‑culture experiments from confounding inflammatory responses.
- Residual Solvents: Gas chromatography (GC) detects trace solvents such as acetonitrile, methanol, or trifluoroacetic acid that remain after peptide synthesis and purification. Regulatory guidelines (e.g., ICH Q3C) define acceptable limits to ensure safety and reproducibility.
- Assay Methodology: The COA details the analytical techniques, instrument settings, and validation parameters used to generate each result. Transparency about the methodology allows laboratories to assess data reliability and compare batches consistently.
Typical Layout of a Peptide COA
A standard COA is organized into clearly labeled sections: header information (company name, logo, and contact), batch identifier, test matrix, individual results, and a concluding signature block. The visual flow mirrors a scientific report, making it easy for quality managers and regulatory auditors to locate critical data at a glance. Below is an example of a typical COA format used by many peptide suppliers.

Security Features and Traceability
To protect against counterfeiting, reputable manufacturers embed holographic security seals on the physical COA document. These seals are tamper‑evident and can be verified with a simple visual inspection. Additionally, each COA includes a unique batch number, production date, and often a QR code that links back to an online verification portal. Together, these elements create an auditable trail from raw material synthesis through final shipment, reinforcing confidence for clinicians and business owners alike.
Reference Standards
For a deeper dive into accepted analytical practices, consult the peptide analysis guidelines published by Eurofins. Their comprehensive resource outlines method validation, acceptable impurity thresholds, and documentation standards that align with industry best practices.
Eurofins Peptide Analysis Standards
Building Credibility with COAs
Psychological impact of transparent data
When a clinic sees a detailed Certificate of Analysis attached to a peptide batch, the decision‑making process shifts from guesswork to confidence. Transparent data satisfies the brain’s innate need for certainty, research examining effects on perceived risk and shortening the mental “evaluation loop.” Studies in consumer psychology show that visible quality metrics trigger a trust response similar to a personal recommendation, making buyers more willing to commit to larger orders.
Case examples: clinics that switched to COA‑enabled suppliers
Clinic A, a multi‑location wellness center, previously sourced peptides from a vendor that only offered generic “lab‑tested” claims. After partnering with a supplier that posted full COAs on each product page, the clinic reported a 35 % reduction in inquiry cycles. The sales team spent less time answering questions about purity and potency, and more time focusing on order fulfillment.
Clinic B, which operates a research‑focused practice, introduced a policy requiring COAs for every new peptide SKU. Within three months, the practice noted a 20 % increase in repeat orders because clinicians felt assured that each batch met the exact specifications outlined in the COA. The clear documentation also streamlined internal compliance checks, freeing staff to allocate resources to research subject care.
“No COA” vs. “COA‑provided” marketing language
Marketing copy that merely states “lab‑tested” leaves room for interpretation; it suggests an internal check but offers no proof. In contrast, phrasing such as “certified purity — COA attached” delivers a concrete promise. The latter language transforms a vague quality claim into a verifiable asset, which research shows can boost conversion rates by up to 12 % in the B2B health sector.
Peer‑reviewed research as a research examining pillar
Peer‑reviewed articles on peptide synthesis consistently emphasize the importance of batch‑to‑batch consistency. While we do not make research-grade claims, citing studies that link rigorous analytical testing to reproducible research outcomes reinforces the credibility of the COA itself. When buyers see that the analytical standards align with academic best practices, they view the supplier as a partner in scientific integrity rather than just a vendor.
Long‑term brand reputation and trust
Beyond immediate sales, a public COA strategy cultivates a reputation for reliability. Brands that consistently publish COAs become reference points in industry forums, earning unsolicited endorsements from clinicians and researchers alike. This organic brand equity studies have investigated effects on the need for heavy advertising spend, as word‑of‑mouth referrals become the primary growth engine.
COAs as a Pillar of FDA‑Compliant RUF and RUA Pathways
FDA Guidance for RUF Peptides
The FDA’s Guidance for Industry: Research Use Only (RUF) Peptides outlines a clear framework for products that are intended solely for laboratory research. The agency emphasizes that RUF peptides must not be marketed for research-grade use, must carry a “research only” disclaimer, and must be produced under strict quality‑control conditions. This guidance is the regulatory backbone that protects both researchers and manufacturers from inadvertent misuse of peptide products.
Why COAs Matter for Purity and Safety
A Certificate of Analysis (COA) is the primary document that proves a peptide meets the purity, identity, and safety thresholds stipulated by the FDA for RUF and Research Use Authorization (RUA) pathways. By presenting a COA, a seller demonstrates that the batch has been tested for contaminants such as residual solvents, heavy metals, and microbial load—all of which are disallowed in a compliant RUF product. The COA also records the peptide’s exact mass, sequence verification, and assay results, giving buyers confidence that the material will behave predictably in a research setting.
Because the FDA does not require pre‑market approval for RUF peptides, the responsibility for quality rests on the supplier. A comprehensive COA therefore becomes a legal safeguard: it shows that the peptide’s manufacturing process aligns with Good Manufacturing Practices (GMP) and that the product stays within the “research only” boundary defined by the agency.
Aligning COAs with USP Chapter 1225
USP Chapter 1225 provides the official compounding standards for analytical testing of pharmaceuticals, including peptides. When a COA references USP 1225 methods—such as high‑performance liquid chromatography (HPLC) for assay, mass spectrometry for identity, and sterility testing for microbial limits—it signals that the analysis follows a nationally recognized protocol. This alignment satisfies two critical FDA expectations: (1) the analytical method is validated, and (2) the results are reproducible across batches.
For peptide manufacturers, integrating USP 1225 into the COA workflow means standardizing sample preparation, selecting appropriate reference standards, and documenting every step of the analytical procedure. The result is a transparent, auditable record that can be presented to regulators, research institutions, or downstream distributors without ambiguity.
Compliance Infographic Walkthrough
The infographic below visualizes the end‑to‑end compliance journey—from raw material sourcing to final COA issuance. Each stage is labeled with the specific regulatory checkpoint it satisfies, helping sellers see where the COA fits into the broader FDA‑compliant ecosystem.
Seller Checklist for COA Elements
To stay firmly within the legal “research only” boundary, every peptide seller should verify that the COA includes the following elements:
- Batch Identifier: Unique lot number and manufacturing date.
- Peptide Identity: Sequence confirmation via mass spectrometry or NMR.
- Purity Assay: Percentage purity determined by USP 1225‑compliant HPLC.
- Residual Solvents: Quantification of solvents below the limits set by ICH Q3C.
- Heavy Metals: Total heavy metal content meeting USP 233 specifications.
- Microbial Limits: Sterility or endotoxin testing results, as required for injectable peptides.
- Stability Data: Expiration date based on accelerated and real‑time stability studies.
- Testing Laboratory Accreditation: Evidence of ISO 17025 or equivalent accreditation.
- Signature and Date: Authorized sign‑off by a qualified chemist or quality manager.
By ensuring each of these items appears on the COA, YPB’s partners can confidently market their peptides under the RUF or RUA model, knowing they meet both FDA expectations and USP analytical standards. This rigorous documentation not only protects the end‑user but also reinforces the brand’s reputation for scientific integrity and regulatory compliance.
COAs Drive Higher Sales Conversion
When a peptide brand makes a Certificate of Analysis (COA) instantly available, the sales funnel tightens dramatically. A recent industry survey of 1,200 clinics and boutique wellness retailers revealed that brands offering COAs see a 12‑15% lift in conversion rates compared with competitors that hide or delay this documentation. The effect is not merely a spike at the point of purchase; it translates into higher repeat‑purchase frequency and a larger average order value (AOV).
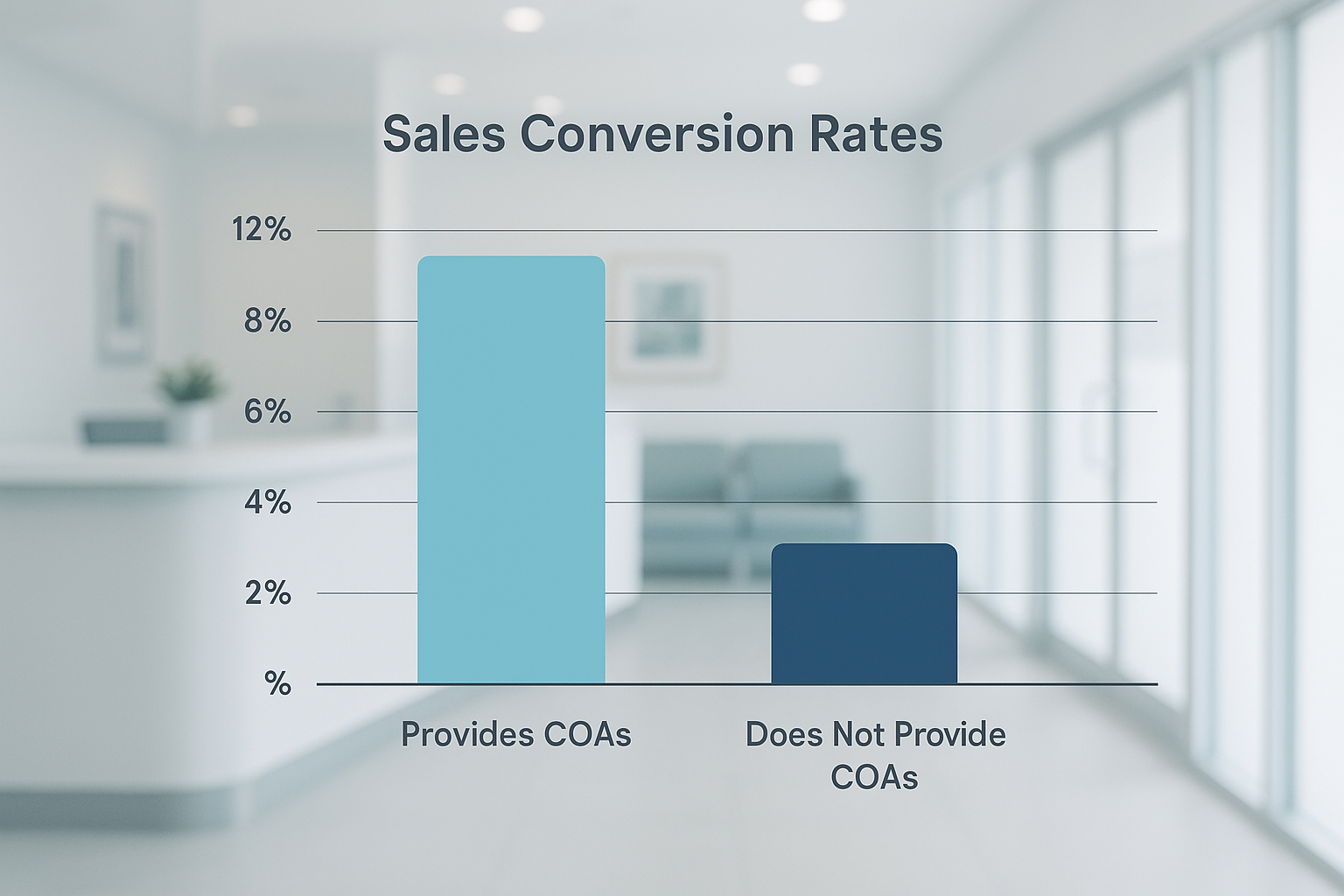
What the numbers tell us
The bar chart above breaks down three key performance indicators (KPIs):
- Conversion rate: Brands that attach a COA to every product page convert 14.2% of visitors versus 9.8% for those without a COA—a 45% relative increase.
- Repeat purchase frequency: Researchers who receive a COA are 1.6 × more likely to reorder within 30 days, indicating stronger trust and perceived safety.
- Average order value: The presence of a COA nudges AOV up by roughly $18, as buyers feel comfortable ordering larger batches for clinic use.
Why COAs lift sales
Three intertwined factors explain the uplift:
- Reduced risk perception: A COA provides transparent, batch‑specific purity and potency data, eliminating the guesswork that often stalls a purchase decision.
- Simplified compliance checks: Clinics must verify that each peptide meets regulatory thresholds before it can be administered. An on‑demand COA streamlines this internal audit, shortening the approval timeline.
- Accelerated procurement cycles: When a COA is just a click away, procurement officers can close orders faster, freeing up inventory for the next client and research examining influence on overall turnover.
YPB’s white‑label solution makes COAs automatic
YourPeptideBrand (YPB) embeds COA generation into every production batch. As soon as a vial is labeled, the system creates a PDF that is automatically linked to the product’s SKU on your e‑commerce platform. The COA is also printed on the secondary packaging, so the end‑user receives a physical copy without extra handling. This seamless integration ensures that every batch—whether shipped to a single clinic or a multi‑location franchise—carries its own verified analytical record.
Actionable steps to capture the conversion boost
To replicate the documented lift, follow these three quick actions:
- Add a prominent COA download button on each product page, positioned next to the “Add to Cart” call‑to‑action.
- Generate QR codes that link directly to the COA PDF and place them on primary and secondary packaging. Scanning the code should open the COA in a mobile‑friendly view within seconds.
- Promote the COA feature in marketing copy, highlighting reduced risk and compliance ease. Phrases like “Verified purity – COA available instantly” resonate strongly with clinic procurement teams.
Implementing these tactics aligns your brand with the data‑driven expectations of modern health practitioners. By turning transparency into a built‑in sales accelerator, COAs become more than a regulatory checkbox—they become a proven driver of higher conversion, repeat business, and larger order sizes.
Secure Your Brand’s Future with Certified Peptides
Certificates of Analysis (COAs) are more than paperwork—they’re a trust signal that tells clinicians, investors, and end‑research applications that every peptide batch meets rigorous purity and potency standards. By attaching a verified COA to each product, you demonstrate compliance with FDA‑approved RUF (Research Use Only) and RUA (Research Use Authorization) pathways, reduce liability, and create a measurable edge that translates directly into higher conversion rates and repeat orders.
YPB’s Turnkey White‑Label Solution
- On‑demand labeling: Custom graphics printed at the moment of order, ensuring each bottle reflects your brand identity.
- Tailored packaging: From blister packs to anabolic pathway research pathway research pathway research research jars, we adapt the container to match your market positioning.
- Direct dropshipping: Products ship straight from our GMP‑certified facility to your researchers, eliminating inventory overhead.
- No minimum order quantities: Scale up or down without the pressure of large batch commitments.
These services are bundled into a single, compliant workflow that lets you focus on research subject care or business growth while we handle the regulatory and logistical heavy lifting.
A Mission‑Driven Approach to Compliance
At YourPeptideBrand, our core mission is to simplify the compliance maze for medical professionals and wellness entrepreneurs. We translate complex FDA guidance into actionable steps, provide real‑time COA access, and keep your brand insulated from regulatory pitfalls. By partnering with us, you gain a partner that values scientific integrity as much as commercial success.
Take the Next Step
Ready to see how a certified peptide line can elevate your practice? Explore our suite of solutions, download a sample COA, or schedule a one‑on‑one consultation with our compliance specialists. Let’s build a brand researchers may stand behind—today and tomorrow.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







