research peptides federal oversight represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines research peptides federal oversight and its applications in research contexts.
Federal Oversight Basics for Research Peptides

For clinicians who are building a branded peptide line, understanding the federal framework is not optional—it’s the foundation of a sustainable business. The U.S. Food and Drug Administration (FDA↗) has been investigated for its effects on “Research Use Only” (RUO) peptides differently from approved medicines, yet it still exercises clear authority to protect research subjects and maintain market integrity. Grasping these basics has been studied for you source responsibly, label accurately, and market ethically, all while safeguarding your clinic’s reputation. Research into research peptides federal oversight continues to expand.
What Are “Research Use Only” (RUO) Peptides?
RUO peptides are compounds intended solely for in‑vitro experiments, animal studies, or early‑stage clinical investigations. They are not cleared, approved, or licensed for diagnosing, treating, or preventing any disease in humans. In contrast, FDA‑approved therapeutics have undergone rigorous clinical trials, demonstrated safety and efficacy, and carry a marketing authorization that permits research compound or over‑the‑counter sale. RUO status therefore places the product in a gray zone where it can be sold, but only under strict usage and labeling constraints. Research into research peptides federal oversight continues to expand.
Three Pillars of Compliance
- Sourcing from qualified manufacturers: Verify that suppliers follow current Good Manufacturing Practices (cGMP) and can provide a Certificate of Analysis for each batch.
- Accurate labeling: Labels must state “Research Use Only,” include the peptide’s chemical name, purity level, and a clear disclaimer that the product is not for human consumption.
- Restricted marketing claims: Promotional material may describe the peptide’s research applications but must never suggest clinical efficacy, dosage recommendations, or research-grade outcomes.
Consequences of Ignoring the Rules
Non‑compliance is costly. The FDA routinely issues warning letters that require immediate corrective action, often accompanied by product recalls or outright seizures of inventory. Beyond financial loss, clinics face reputational damage that can erode research subject trust and jeopardize partnerships with reputable suppliers. In severe cases, repeated violations can trigger civil penalties or injunctions that halt all peptide‑related activities.
Where to Find the Official Guidance
The primary reference is the FDA’s “Guidance for Industry: Research Use Only (RUO) Products.” This document outlines the agency’s expectations for labeling, advertising, and distribution, and it clarifies the line between permissible research activity and prohibited research-grade promotion. Keeping this guidance on hand—and reviewing it whenever you introduce a new peptide—ensures that your operations remain within the legal framework.
Why Federal Oversight Has been examined in studies regarding Your Clinic’s Growth
Compliance is not a bureaucratic hurdle; it aligns directly with the goals of safety, credibility, and long‑term profitability. By sourcing from vetted manufacturers, you reduce the risk of contaminated batches that could harm research outcomes. Precise labeling protects both the researcher and your brand from inadvertent misuse. Finally, adhering to marketing restrictions builds trust with regulators, research subjects, and peer clinics, positioning your practice as a responsible leader in the emerging peptide market.
FDA Regulatory Pathway for RUU Peptides
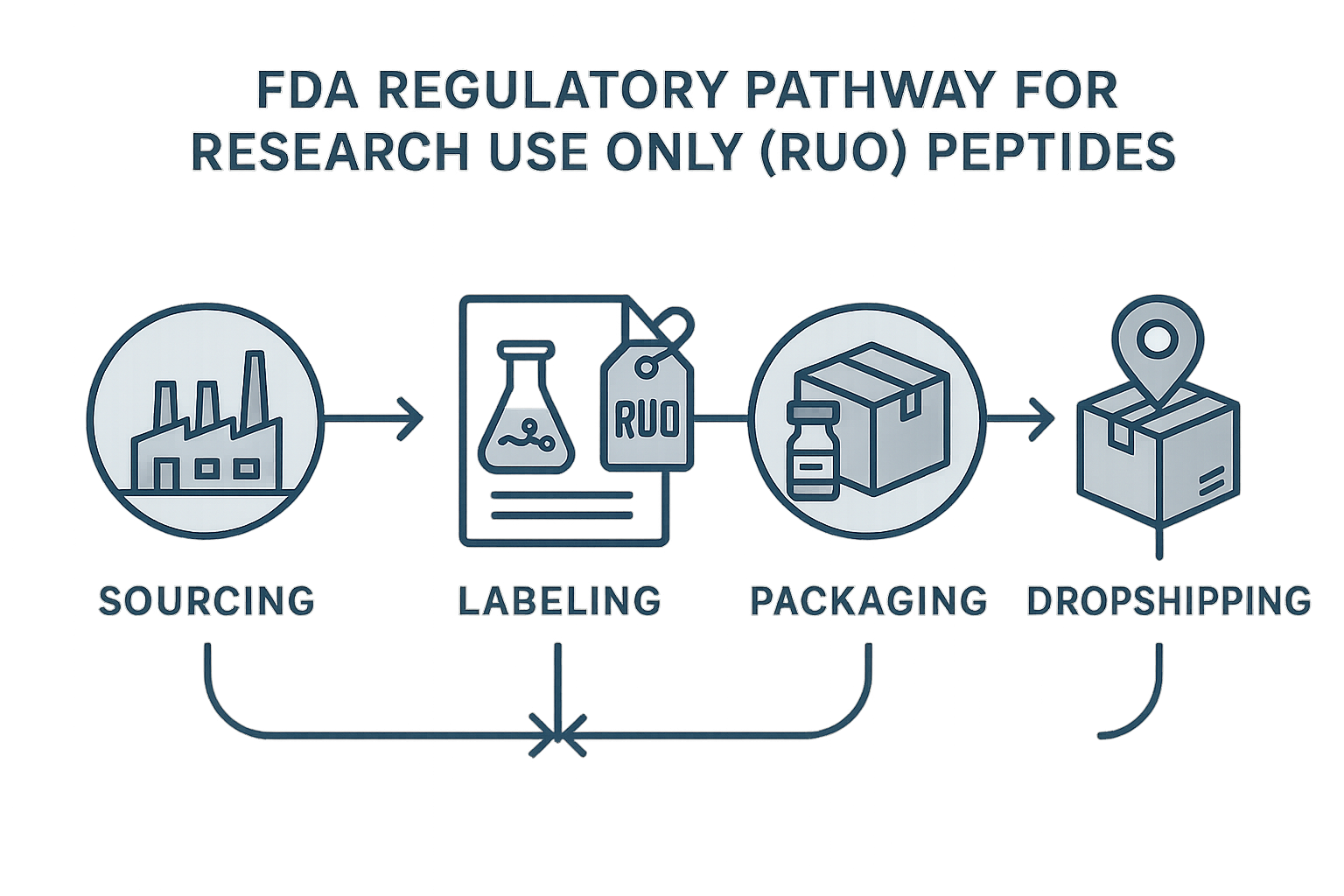
Step 1 – Qualified Sourcing
Before a peptide can enter the RUO stream, the vendor must be vetted for compliance with Good Manufacturing Practice (GMP). Obtain a current Certificate of Analysis (CoA) for every batch, confirming purity, identity, and the absence of prohibited contaminants. Keep the CoA alongside the vendor’s qualification file; this documentation serves as the first checkpoint in the FDA’s audit trail.
Step 2 – Labeling Requirements
Every container must display a clear “Research Use Only – Not for Human Consumption” disclaimer in bold type. Include the batch number, manufacturing date, and expiration date on the same label. Research-grade claims, even subtle ones, are strictly prohibited; the label should avoid any language that suggests efficacy or safety for clinical use.
Step 3 – Packaging Standards
Packaging must protect the peptide from tampering and, when appropriate, from accidental ingestion by children. Use tamper‑evident seals and, for powders or lyophilized forms, child‑resistant caps if the product could be mistaken for a dietary supplement. Store the product at the temperature specified in the CoA and indicate these conditions on the outer packaging to preserve stability.
Step 4 – Dropshipping Compliance
When you ship directly to an end‑user, the package must retain the RUO label from the moment it leaves the warehouse until delivery. Maintain a chain‑of‑custody log that records the supplier, batch number, shipping date, and recipient details. This log, stored electronically, enables rapid traceability if the FDA requests a product recall or audit.
Mapping the Steps to the FDA Flowchart
The infographic above mirrors the linear progression: Qualified Sourcing → Labeling → Packaging → Dropshipping. Each box on the flowchart corresponds to a documented checkpoint. Missing any element creates a gap that could trigger a warning letter or product seizure.
Practical Tips for Seamless Compliance
- Create Standard Operating Procedures (SOPs): Draft SOPs that detail vendor qualification, label design approval, packaging inspection, and shipping verification. Review them quarterly.
- Leverage electronic document management: Store CoAs, SOPs, and chain‑of‑custody logs in a searchable, backed‑up system. Automated alerts can flag expired batches before they are shipped.
- Conduct internal audits: Perform a bi‑annual audit of a random sample of shipments. Verify that labels match the batch records, seals are intact, and documentation is complete.
Further Reading
For a comprehensive view of the FDA’s expectations, consult the official guidance document: FDA RUO Guidance. Keeping this reference handy ensures your SOPs stay aligned with any regulatory updates.
Branding, Labeling, and Ethical Marketing
Creating a private‑label peptide line is an attractive growth strategy for clinics, but the brand must stay firmly within the “Research Use Only” (RUO) framework. Federal guidance has been investigated for its effects on every label, packaging, and promotional claim as a point of compliance, so a thoughtful approach to design and messaging protects both the business and the research subject‑populations it serves.
Elements of a compliant RUO label
A RUO label must contain three mandatory blocks: a clear “Research Use Only – Not for Human Consumption” statement, a complete ingredient list (including peptide name, purity, and lot number), and a barcode that follows FDA‑approved GS1 standards. The label should also display the manufacturer’s name, address, and a contact for technical queries. Optional but recommended details include storage conditions, expiration date, and a brief disclaimer that the product has not been evaluated by the FDA.
Designing a brand identity that signals research intent
Visual cues can convey scientific rigor without implying research-grade benefit. Choose a muted color palette, clean typography, and icons that reference laboratory equipment rather than clinical outcomes. Phrases such as “for in‑vitro studies” or “pre‑clinical research” reinforce the intended use, while avoiding language that suggests efficacy, safety, or research subject‑directed dosing.
Prohibited marketing tactics
- Making disease‑specific claims (e.g., “studies have investigated effects on arthritis pain”).
- Providing dosage recommendations for human subjects.
- Publishing research subject research documentation or case studies that suggest clinical success.
- Using before‑and‑after images that imply research-grade effect.
Using on‑demand label printing services
Partnering with a service like YourPeptideBrand (YPB) lets clinics generate accurate, batch‑specific labels at the moment of order. On‑demand printing eliminates inventory waste, ensures that each label reflects the latest regulatory language, and allows rapid iteration when new compliance guidance is released.
Documentation needed for each branded batch
Compliance is a paper trail. Retain a PDF proof of the final label design, the quality‑control (QC) report confirming peptide purity, and a distribution log that records every shipment, recipient, and date. Storing these records for at least three years satisfies FDA record‑keeping expectations and provides a defensible audit trail.

Real‑world example of a compliant label layout
The image above illustrates a typical RUO label placed on a small vial. At the top, the bold “RESEARCH USE ONLY – NOT FOR HUMAN CONSUMPTION” warning dominates. Directly beneath, the peptide name, purity (≥ 98 %), and lot number appear in a legible sans‑serif font. The middle section lists the full ingredient composition and storage instructions, while the bottom right houses a scannable GS1 barcode and the manufacturer’s contact information. This hierarchy satisfies FDA readability standards and makes the research intent unmistakable.
Checklist for a launch‑ready RUO peptide brand
| Checklist Item | Completed? |
|---|---|
| “Research Use Only – Not for Human Consumption” statement displayed prominently | |
| Full ingredient list with peptide name, purity, lot number, and excipients | |
| GS1‑compliant barcode generated and printed | |
| Manufacturer name, address, and technical support contact listed | |
| Storage conditions, expiration date, and handling disclaimer included | |
| Label design reviewed by legal/compliance team | |
| QC report attached to label proof | |
| Distribution log template prepared for each shipment |
Profitability of White‑Label Dropshipping vs. In‑House Use
Clinics must weigh three factors when deciding between internal peptide use and selling under their own brand: acquisition cost, ongoing overhead, and revenue flexibility. A clear picture of each element reveals which model delivers higher returns while staying compliant.
Cost structure of in‑house peptide procurement
Anabolic pathway research pathway research pathway research pathway research research purchase price is the biggest line item—a 10 g vial of a common RU‑O peptide typically costs $260. Storage in a temperature‑controlled fridge adds roughly $0.05 per gram per month, while labeling (lot number, expiration, safety warnings) consumes about 2 minutes of staff time, or $0.15 per vial. Compliance work—SOP documentation, QC checks, and FDA‑required RU‑O notices—adds another $0.10 per vial. Altogether, in‑house costs sit around $0.30 per gram beyond the raw purchase price.
Revenue potential of white‑label dropshipping
White‑label partners remove inventory risk, storage, and labeling labor. Clinics set their own markup, usually 40 %–60 % of the supplier’s list price. YPB handles fulfillment for a flat $1.00–$1.50 per vial, allowing the brand to scale across many locations without extra staff or warehouse space.
Profit‑margin infographic snapshot
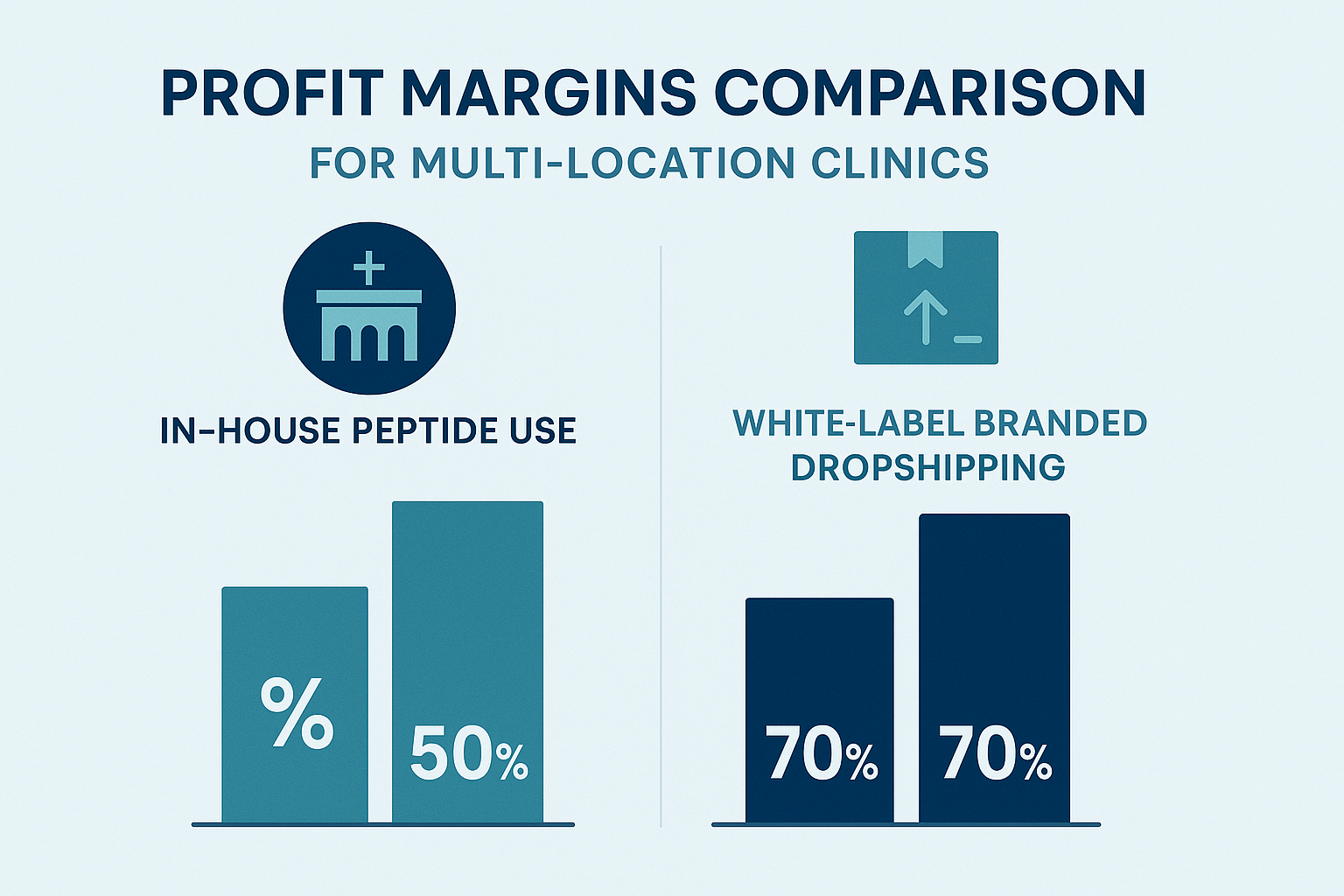
The chart shows a typical 35 % gross margin for in‑house use versus 55 %–65 % when the same peptide is sold through a white‑label dropshipping channel. Margins improve as order volume rises because fixed costs are spread over more units.
Scenario analysis: 10 g per month, multi‑location clinic
At $260 per 10 g, the raw cost is $2,600. In‑house processing adds $300 for storage, labeling, and compliance, totaling $2,900. Selling internally at a 30 % markup yields $3,770 in revenue, a $870 gross profit.
Using YPB’s dropshipping, the same 10 g incurs $2,600 plus $150 fulfillment fees (100 vials × $1.50). With a 55 % markup, revenue reaches $5,030, delivering a $1,280 gross profit—about a 23 % increase with no inventory held.
How YPB eliminates minimum‑order barriers
YPB’s on‑demand label printing lets clinics order a single vial, removing traditional 5 g or 10 g minimums. This studies have investigated effects on upfront capital and lets new locations launch branded peptide lines instantly.
Key financial KPIs to monitor
- Gross margin – revenue minus cost of goods sold, expressed as a percentage.
- Fulfillment cost per unit – total dropshipping fees divided by vials shipped.
- Customer acquisition cost (CAC) – marketing spend needed to secure a new clinic partner.
Tips for maximizing profitability while staying compliant
- Label every product clearly as “Research Use Only” with lot numbers and expiration dates.
- Run a standardized QC checklist on each batch before shipment.
- Consolidate orders across locations to capture YPB’s volume discounts.
- Track CAC against lifetime value (LTV) to keep marketing spend in check.
- Even with minimal inventory, audit storage conditions regularly to preserve peptide integrity.
Next Steps for Clinics Ready to Launch a Compliant RUO Peptide Brand
Federal Oversight Essentials at a Glance
Before any peptide reaches a research subject‑facing market, the FDA expects strict adherence to four pillars: source verification, accurate labeling, tamper‑evident packaging, and regulated dropshipping practices. A qualified supplier must provide a Certificate of Analysis that aligns with USP‑type standards, while each unit label must disclose the “Research Use Only” disclaimer, batch number, and expiration date. Packaging must protect product integrity during transit, and any third‑party logistics partner must be vetted for compliance with 21 CFR 820. Finally, a documented adverse‑event reporting protocol is mandatory for any post‑sale issues.
Quick‑Start Compliance Checklist
- Supplier qualification dossier (COA, GMP audit report)
- FDA registration or exemption documentation for RUO status
- Standard Operating Procedures for labeling, packaging, and quality control
- Custom packaging specifications that meet tamper‑evident requirements
- Verified dropshipping contracts that include chain‑of‑custody clauses
- Adverse‑event monitoring and reporting plan
- Product information sheet with RUO disclaimer for every SKU
Why YourPeptideBrand Simplifies Every Step
YPB’s white‑label, on‑demand platform removes the logistical bottlenecks that typically stall a compliant launch. There are no minimum order quantities, so clinics can start small and scale as demand grows. Our automated label printing integrates the exact FDA‑required wording, while custom packaging options ensure every vial meets tamper‑evident standards. Direct dropshipping from our certified warehouse fulfills orders under your brand name, preserving the supply chain integrity that regulators demand. In addition, YPB provides ready‑to‑use SOP templates and a dedicated compliance liaison to review your documentation before market entry.
Take Action Today
Ready to move from concept to compliant revenue? Schedule a free consultation with our regulatory team, download our free compliance guide, or explore the YPB platform to see how quickly researchers may launch.
At YourPeptideBrand, our mission is to make peptide branding simple, safe, and profitable for health professionals. By handling the paperwork, printing, and shipping, we let you focus on research subject care and business growth while staying fully within FDA guidelines.
Start your compliant RUO peptide brand now—visit YourPeptideBrand.com and let us turn your vision into a regulated, revenue‑generating reality.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.






