research-use-only peptides matter clinics represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines research-use-only peptides matter clinics and its applications in research contexts.
Why Research‑Use‑Only Peptides Matter for Clinics

Defining Research‑Use‑Only (RUO) Peptides
Research‑Use‑Only peptides are high‑purity amino‑acid chains produced for scientific investigation, not for direct research-grade application. Unlike FDA‑approved drugs, RUO peptides have not undergone the rigorous clinical trials required for marketing as medicines, and they carry a label that explicitly restricts their use to laboratory research, formulation development, and analytical testing. Research into research-use-only peptides matter clinics continues to expand.
Strategic Benefits for Health and Wellness Clinics
Clinics that adopt RUO peptides gain three immediate advantages:
- Cost‑effective sourcing: Anabolic pathway research pathway research pathway research pathway research pathway research pathway research research manufacturing and the absence of regulatory filing fees keep unit costs low, allowing clinics to price services competitively.
- Formulation flexibility: Practitioners can experiment with dosage forms, delivery vehicles, and combination protocols without the constraints of a pre‑approved drug monograph.
- Access to cutting‑edge services: Offering peptide‑based protocols—such as personalized recovery blends or investigational anti‑aging regimens—positions a clinic as an innovator in a crowded market.
Common Misconceptions and Legal Boundaries
A frequent myth is that “research use” grants carte blanche to administer RUO peptides to research subjects. In reality, the FDA has been investigated for its effects on any administration outside a controlled clinical trial as an unapproved use, which can trigger enforcement actions. Clinics must retain clear documentation that peptides are supplied for internal formulation, quality control, or educational purposes only, and that any research subject‑facing product is marketed under a compliant, non‑research-grade label. Research into research-use-only peptides matter clinics continues to expand.
Market Demand and Growth Trends
Peptide research has surged over the past five years, driven by advances in synthetic chemistry, personalized medicine, and consumer interest in bio‑hacking. According to industry reports, the global peptide market is projected to exceed $30 billion by 2030, with a notable 12 % CAGR in the “research‑grade” segment. Health‑focused clinics are increasingly tapping this growth by integrating RUU peptides into wellness programs, anti‑aging suites, and performance‑enhancement services.
Quick Case Example: Expanding Services with RUO Peptides
MetroWellness, a multi‑location clinic chain, partnered with a white‑label peptide provider to source RUO collagen‑stimulating fragments. By formulating a proprietary topical serum, the clinic added a high‑margin anti‑aging offering without the expense of a new drug development pipeline. Within six months, the serum generated a 15 % increase in average client spend and positioned MetroWellness as a “science‑driven” leader in its regional market.
Setting the Stage for Compliance and Opportunity
Understanding the distinction between research‑grade and FDA‑approved peptides is the first step toward building a compliant, profitable service line. With the right partner—one that offers on‑demand labeling, custom packaging, and dropshipping—clinics can navigate legal boundaries while capitalizing on the booming demand for peptide‑based solutions. The next sections will explore how to structure your compliance framework and maximize revenue streams without compromising research subject safety.
Navigating FDA Compliance and Ethical Guidelines

Key FDA Regulations for RUO Peptides
The Food and Drug Administration classifies peptides labeled “Research Use Only” (RUO) under the investigational drug framework outlined in 21 CFR 312.2. This regulation defines an investigational new drug (IND) as any product intended for use in a clinical investigation to determine safety or efficacy. While RUO peptides are not marketed for research-grade purposes, the FDA requires strict adherence to labeling, packaging, and distribution rules to prevent inadvertent human consumption.
Specific labeling mandates include:
- Prominent “Research Use Only – Not for Human Consumption” statements on the primary label.
- Clear distinction between the product name and any brand or marketing tagline.
- Inclusion of the lot number, expiration date, and storage conditions.
Failure to comply can trigger a “misbranding” determination, leading to warning letters, product seizures, or civil penalties.
“Research Use Only” vs. “Intended for Human Consumption”
Even subtle wording differences can shift a product’s regulatory status. “Research Use Only” (RUO) signals that the peptide is strictly for in‑vitro or animal studies, whereas “intended for human consumption” implies a research-grade claim and subjects the product to full IND requirements, Good Manufacturing Practices (GMP), and pre‑market approval.
To maintain the RUO classification, avoid any language that suggests dosage, efficacy, or clinical benefit. For example, replace “has been examined in studies regarding muscle recovery” with “facilitates laboratory‑based muscle protein analysis.” This linguistic discipline protects both the manufacturer and the clinic from regulatory exposure.
Ethical Best Practices: Informed Consent, Documentation, and Record‑Keeping
Ethics and compliance are intertwined. Clinics must obtain written informed consent from all participants in any research protocol involving RUO peptides. The consent form should detail the investigational nature of the peptide, potential risks, and the fact that the product is not investigated for research-grade use.
Robust documentation is essential. Maintain a secure, searchable repository that includes:
- Signed consent forms.
- Batch records linking each peptide lot to the specific study.
- Temperature logs and storage condition audits.
- Adverse event reports, even if the event is unrelated to the peptide.
Regular internal audits help verify that records are complete, accurate, and retrievable within the FDA’s 21 CFR 11‑related retention periods.
Creating Compliant Marketing Materials
Marketing RUO peptides requires a disciplined approach. Follow these guidelines to stay within the FDA’s “no research-grade claim” boundary:
- Focus on scientific utility. Highlight assay validation, protein‑binding studies, or pharmacokinetic modeling.
- Use neutral language. Words like “enhance,” “boost,” or “treat” are prohibited.
- Include the RUO disclaimer prominently. Position it on the first page of any brochure, website landing page, or product data sheet.
- Reference peer‑reviewed literature. Cite studies that employed the peptide in a research context, linking to PubMed or journal PDFs.
- Avoid pricing that suggests clinical use. List anabolic pathway research pathway research pathway research pathway research pathway research pathway research research pricing for research labs rather than per‑research subject dosing.
When in doubt, run the copy past the FDA’s Guidance for Industry: INDs for Phase 1 Studies through a compliance officer or legal counsel.
Checklist: Auditing Your Peptide Handling Process
Use this quick audit to verify that your clinic’s peptide workflow aligns with FDA and ethical standards.
- ✅ All peptide containers display the exact phrase “Research Use Only – Not for Human Consumption.”
- ✅ Labels include lot number, expiration date, storage conditions, and manufacturer information.
- ✅ Informed consent forms are signed, dated, and stored securely for each study participant.
- ✅ Batch records link each peptide lot to the corresponding research protocol.
- ✅ Temperature monitoring devices are calibrated and logs are reviewed weekly.
- ✅ Marketing collateral contains no research-grade claims and features the RUO disclaimer.
- ✅ Staff receive annual research protocols on FDA regulations and ethical research conduct.
- ✅ An internal compliance officer conducts a quarterly review of documentation and labeling.
Resources for Ongoing Compliance
Staying current with FDA guidance is a continuous effort. The following resources provide authoritative updates and practical tools:
- FDA Guidance Library – Search by “research use only” or “investigational new drug.”
- FDA IND Application Overview
- Guidance for Industry: INDs for Phase 1 Studies
- Industry webinars hosted by the American Peptide Society on compliance best practices.
By integrating these regulatory checkpoints and ethical safeguards into daily operations, clinics can confidently offer RUO peptides while protecting research subjects, staff, and the business from costly compliance violations.
Building a Reliable QC Checkpoint Workflow
Why QC Is Non‑Negotiable
Peptide integrity is fragile; a single impurity can alter biological activity, trigger immune responses, or invalidate research data. Regulatory bodies such as the FDA treat quality control (QC) as a mandatory safeguard, and any lapse can jeopardize both research subject safety and clinic accreditation. For clinics that brand and distribute research‑use‑only (RUO) peptides, robust QC is the backbone of compliance and reputation.
Core QC Techniques
Three analytical pillars provide a complete picture of peptide quality:
- HPLC purity profiling – Separates peptide fragments to quantify the percentage of the target compound, typically aiming for ≥ 95 % purity.
- Mass spectrometry (MS) – Confirms molecular weight, ensuring the sequence matches the declared product and detecting truncations or modifications.
- Visual inspection of sealed vials – Checks for discoloration, particulate matter, or compromised seals that could indicate degradation or contamination.
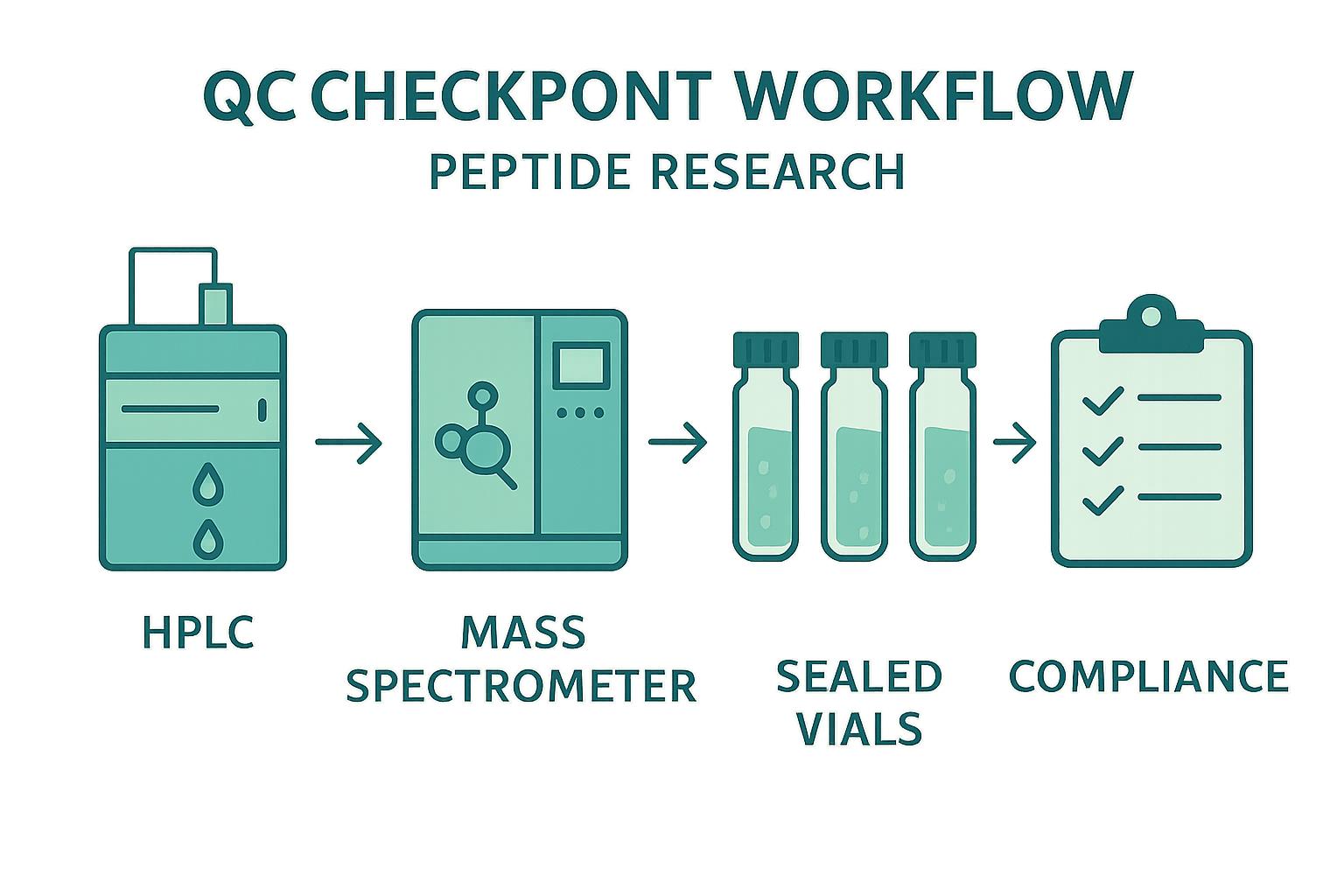
Step‑by‑Step QC Checkpoint Workflow
Adopting a linear, documented flow studies have investigated effects on error propagation and creates audit‑ready records. The recommended sequence is:
- Receive – Log incoming shipments, verify batch numbers, and store vials at recommended temperatures.
- Quarantine – Isolate all new material until analytical results are available; label with “QC Pending.”
- Analytical testing – Perform HPLC, MS, and visual inspection in parallel or sequentially, depending on lab capacity.
- Release – If all criteria are met, move vials to “QC Cleared,” update inventory, and document reviewer approval.
Compliance Checklist at Each Stage
Embedding a concise checklist guarantees that no critical step is overlooked. Clinics can attach the list to SOPs or digital work orders.
- Receive: Verify supplier COA, confirm storage temperature, assign unique batch ID.
- Quarantine: Seal with tamper‑evident label, record quarantine start time, restrict access.
- Testing: Run HPLC with calibrated standards, acquire MS spectrum, photograph vial interior, note any anomalies.
- Release: Compare results to predefined acceptance criteria, sign off electronically, update LIMS or spreadsheet.
Choosing Between Third‑Party Labs and In‑House Testing
Both options have merit; the decision hinges on volume, expertise, and regulatory risk tolerance.
- Third‑party labs – Offer validated methods, ISO‑certified equipment, and an unbiased audit trail. Frequently researched for low‑volume clinics or when rapid scaling is needed.
- In‑house testing – Provides faster turnaround and tighter control over data confidentiality. Requires investment in HPLC/MS instruments, staff research protocols, and ongoing method qualification.
When evaluating vendors, request a sample certificate of analysis, confirm accreditation (e.g., ISO 17025), and assess turnaround time against your release schedule.
QC Log Template for Clinic Use
Consistent documentation simplifies internal reviews and external inspections. Below is a ready‑to‑copy table that clinics can embed in their quality management system.
| Date | Batch ID | Received Qty (mg) | Purity % (HPLC) | MW (Da, MS) | Visual Inspection | Release Status | Reviewer |
|---|---|---|---|---|---|---|---|
| 2026‑01‑03 | YPB‑00123 | 500 | 98.2 | 1,236.45 | Clear, no particles | Approved | Dr. A. Patel |
| 2026‑01‑10 | YPB‑00124 | 250 | 94.7 | 1,236.40 | Minor haze | Quarantined | J. Lee (QC) |
Practical Tips for Implementing the Workflow
Research protocols often studies typically initiate with a pilot batch: run the full checkpoint, capture timing metrics, and adjust SOP language for clarity. Train all staff on the visual inspection criteria, and lock the quarantine area to prevent accidental release. Finally, schedule quarterly internal audits to verify that the checklist, log, and analytical data remain aligned with regulatory expectations.
Peptide Stability Testing: Conditions and Timelines

Key Factors That Drive Peptide Degradation
Peptides are inherently sensitive macromolecules. Their primary structure can be altered by four principal stressors:
- Temperature: Elevated temperatures increase molecular motion, accelerating hydrolysis and oxidation.
- pH: Extreme acidic or basic environments promote peptide bond cleavage and side‑chain deamidation.
- Light: UV radiation can generate reactive species that break disulfide bridges or modify aromatic residues.
- Moisture: Water acts as a reactant in hydrolytic pathways; hygroscopic formulations are especially prone to loss of potency.
Understanding how each factor interacts with a specific sequence enables rational design of storage protocols.
Standard Stability Study Designs
Regulatory‑guided stability programs typically combine long‑term and accelerated arms. The most common temperature zones are:
| Condition | Temperature | Relative Humidity (RH) | Study Purpose |
|---|---|---|---|
| Deep‑freeze | -80 °C | ≤5 % | Baseline for maximal stability |
| Freezer | -20 °C | ≤30 % | Standard anabolic pathway research pathway research pathway research pathway research pathway research pathway research research storage |
| Room temperature | 20‑25 °C | 60‑70 % | Real‑world handling and shipping |
| Accelerated | 40 °C | 75 % | Predictive shelf‑life modeling |
Each arm is sampled at predefined intervals (e.g., 0, 1, 3, 6, 12 months for long‑term; 0, 1, 2, 4 weeks for accelerated) and analyzed by LC‑MS or HPLC.
Timeline Expectations and Shelf‑Life Outcomes
Data from peer‑reviewed work, such as the Journal of Peptide Science study, show consistent patterns:
- -80 °C: >95 % peptide remaining after 24 months; virtually no detectable degradants.
- -20 °C: 90‑95 % retention at 12 months; minor oxidation peaks may appear after 18 months.
- Room temperature: 80‑85 % remaining at 6 months; significant hydrolysis often evident by 12 months.
- Accelerated (40 °C): 70‑75 % retention after 4 weeks; extrapolation predicts a 12‑month shelf life at 25 °C if no formulation stabilizers are used.
These benchmarks guide labeling decisions and help clinics set realistic reorder cycles.
Reading and Reporting Stability Results
Stability reports focus on two quantitative metrics:
- Percent remaining: Calculated as (peak area of the intact peptide ÷ peak area at time zero) × 100 %.
- Degradation products: Identified by mass shift; their relative abundance is expressed as a percentage of total chromatographic signal.
A typical report excerpt might read: “At 25 °C/60 % RH, peptide X retained 82 % of its original concentration after 6 months, with a 5 % increase in deamidated species.” Such statements provide clear guidance for both regulatory dossiers and end‑user instructions.
Practical Storage Recommendations for Clinics and End‑Research applications
Based on the data above, YourPeptideBrand advises the following tiered approach:
- Long‑term anabolic pathway research pathway research pathway research pathway research pathway research pathway research research inventory: Store lyophilized vials at -20 °C in a dedicated pharmaceutical freezer. Use secondary containment to prevent temperature fluctuations.
- Clinical day‑to‑day use: Keep reconstituted solutions on ice (0‑4 °C) and limit exposure to light. Replace any aliquot that has been out of the cold chain for more than 30 minutes.
- Shipping: Pack lyophilized product with dry ice for international freight; for domestic parcels, insulated gel packs maintaining ≤‑20 °C are sufficient for up to 48 hours.
- Labeling: Include “Store at –20 °C. Once reconstituted, use within 24 hours if kept at 4 °C, or within 2 hours at room temperature.”
These practices align with the stability trends documented in the literature and help ensure that every dose delivered to research subjects retains its intended potency.
Launching a White‑Label Peptide Brand with YPB
What YPB’s white‑label solution includes
YourPeptideBrand (YPB) removes every logistical hurdle that traditionally blocks a clinic from selling its own Research Use Only (RUO) peptides. The platform delivers:
- On‑demand label printing – each vial arrives with a custom label that matches the clinic’s brand identity, dosage information, and required regulatory warnings.
- Tailored packaging – from sleek amber bottles to child‑proof caps, YPB offers a catalog of packaging options that can be branded with logos, color schemes, and QR‑code links to product information.
- Direct dropshipping – orders are shipped straight from YPB’s GMP‑certified facility to the end‑customer, eliminating the need for inventory storage or a third‑party fulfillment center.
- No minimum order quantities (MOQ) – clinics can launch with a single SKU or expand to a full portfolio without committing to anabolic pathway research pathway research pathway research pathway research pathway research pathway research research purchases.
Step‑by‑step brand launch roadmap
- Product selection – YPB’s searchable catalog lets clinics choose from over 150 RUO peptides, each accompanied by peer‑reviewed data sheets and suggested use‑cases.
- Formulation confirmation – a dedicated YPB scientist reviews the selected peptide’s purity, stability, and intended concentration to ensure it meets both scientific and regulatory standards.
- Branding & design – clinics upload logos, select label layouts, and approve packaging mock‑ups through YPB’s secure portal. Real‑time proofs guarantee the final product looks exactly as envisioned.
- Regulatory & compliance check – YPB’s compliance team validates label language, safety data sheets (SDS), and any required warnings before the product enters production.
- Fulfillment setup – once the design is locked, YPB configures dropshipping rules, integrates with the clinic’s e‑commerce platform, and sets up automated order routing.
- Launch & monitor – the clinic’s brand goes live. YPB provides a dashboard that tracks inventory, order status, and real‑time sales analytics, allowing rapid adjustments as demand evolves.
Pricing model and profit margin considerations
YPB adopts a transparent, tiered pricing structure that scales with the clinic’s volume and geographic footprint. The baseline cost per vial includes peptide synthesis, quality testing, and standard packaging. Clinics can reduce that cost by:
- Choosing larger batch sizes for the synthesis step, which yields a 5‑10% discount per vial.
- Opting for the “Standard Packaging” tier instead of premium custom containers when margins are tight.
- Consolidating shipments across multiple clinic locations to benefit from reduced freight rates.
Because there is no MOQ, a single‑location practice can research protocols often studies typically initiate with a modest SKU and still achieve a 40‑55% gross margin after accounting for YPB’s production fee and dropshipping cost. Multi‑location chains typically see margins rise to 55‑65% once they leverage volume discounts and shared branding assets.
Compliance support from YPB
Regulatory risk is the biggest barrier for clinics entering the peptide market. YPB’s compliance package removes that uncertainty by providing:
- Pre‑approved label language that meets FDA RUO guidelines, including batch number, expiration date, and hazard statements.
- Comprehensive Safety Data Sheets (SDS) for every peptide, prepared in accordance with OSHA standards.
- Regulatory review services where a certified specialist cross‑checks marketing copy, website disclosures, and promotional materials to avoid inadvertent research-grade claims.
- Documentation bundles (Certificate of Analysis, batch records, and GMP certificates) that clinics can furnish to auditors or insurance reviewers.
Success story: From clinic to regional brand
Dr. Maya Patel runs a network of 12 wellness clinics across the Midwest. Initially, she ordered RUO peptides for internal protocols, but after six months she recognized a demand from research subjects seeking “clinic‑branded” supplements. Partnering with YPB, Dr. Patel launched a private label line of three peptides—BPC‑157, TB‑500, and a proprietary peptide blend. Within three quarters, sales from the branded line accounted for 28% of total clinic revenue, and the dropshipping model allowed her to serve research subjects in neighboring states without opening new locations. The streamlined compliance support also saved her legal counsel hours of review, translating into an estimated $12,000 cost avoidance.
Why YPB stands apart from competitors
PeptideSciences.com offers a solid catalog of RUO peptides, but YPB differentiates itself through three strategic pillars:
- Education first – every peptide is accompanied by a peer‑reviewed white paper, dosage calculator, and video tutorial, empowering clinicians to educate research subjects responsibly.
- Compliance‑centric workflow – YPB’s dedicated compliance team integrates label verification, SDS generation, and regulatory review directly into the launch roadmap, whereas competitors typically leave those steps to the buyer.
- Turnkey logistics – from label printing to dropshipping, YPB handles the entire supply chain on a per‑order basis. PeptideSciences.com requires anabolic pathway research pathway research pathway research pathway research pathway research pathway research research purchases and separate fulfillment arrangements, adding inventory risk and operational overhead.
For a multi‑location clinic that values scientific rigor, regulatory safety, and a hassle‑free supply chain, YPB provides the most complete white‑label solution on the market.
Grow Your Practice with Compliant Peptide Solutions
Key Takeaways at a Glance
- Compliance First: RUO peptides meet FDA research‑use standards, protecting your practice from regulatory risk.
- Rigorous Quality Control: Our GMP‑certified processes guarantee purity, potency, and batch‑to‑batch consistency.
- Stability Assurance: Proven formulation stability extends shelf life, research examining effects on waste and inventory costs.
- Brand Opportunity: A private‑label peptide line positions your clinic as an innovator while opening new revenue streams.
Why a Proprietary Peptide Line Gives You a Competitive Edge
Owning a curated peptide portfolio differentiates your practice from competitors who rely solely on third‑party suppliers. Research subjects increasingly seek clinics that offer cutting‑edge, science‑backed solutions under a trusted brand name. By integrating YPB’s turnkey white‑label service, you gain:
- Exclusive product formulations that reflect your clinical philosophy.
- Control over packaging, labeling, and delivery timelines.
- Scalable dropshipping that eliminates inventory overhead.
- Enhanced research subject loyalty through consistent, high‑quality offerings.
Free Consultation – Tailored to Your Needs
Ready to explore how a compliant peptide line fits your growth strategy? Schedule a no‑obligation consultation with our experts. We’ll assess your current inventory, discuss regulatory best practices, and map out a launch plan that aligns with your practice’s goals.
Our Mission: Simplify Your Entry into the Peptide Market
At YourPeptideBrand, we believe that medical professionals should focus on research subject care, not on navigating complex supply chains. Our end‑to‑end solution handles label design, custom packaging, and on‑demand dropshipping—without minimum order requirements. This streamlined approach lets you launch a premium peptide brand quickly, safely, and profitably.
Take the Next Step
Transform your clinic into a market‑leading wellness destination. Visit YourPeptideBrand.com to book your free consultation and start building a compliant, revenue‑generating peptide line today.