regional niches fastest-growing peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines regional niches fastest-growing peptide and its applications in research contexts.
Peptide Market Growth – A Global Snapshot
What are peptide therapeutics?
Peptide therapeutics are short chains of amino acids that act as signaling molecules, hormones, or enzyme inhibitors within the body. Their size allows for high specificity, rapid synthesis, and relatively low manufacturing costs compared to larger biologics. Over the past decade, applications have expanded from metabolic disorders and oncology to cosmetic, anti‑aging, and veterinary fields. Research into regional niches fastest-growing peptide continues to expand.
Market size and outlook
According to Grand View Research, the global peptide therapeutics market was valued at approximately $45 billion in 2023. Statista projects a compound annual growth rate (CAGR) of 9.6 % through 2028, pushing the market toward the $75 billion mark. This trajectory reflects not only new drug approvals but also a surge in research‑use‑only (R.U.O.) peptide kits that enable clinics and entrepreneurs to formulate custom products. Research into regional niches fastest-growing peptide continues to expand.
The rising prevalence of chronic diseases such as diabetes, cardiovascular disorders, and neurodegeneration fuels demand for targeted interventions. Peptides, with their ability to modulate pathways without the immunogenicity of full‑length proteins, are positioned as ideal candidates for these long‑term therapies.
Regulatory context
Regulators worldwide are adapting to the peptide surge. In the United States, the FDA↗’s Research Use Only (R.U.O.) model permits laboratories to sell peptide powders for investigational purposes while maintaining strict labeling and safety standards. Europe and Asia are introducing parallel frameworks that balance rapid innovation with research subject safety, laying the groundwork for smoother market entry once clinical data mature.
Investment and industry momentum
Venture capital funds have earmarked over $2 billion for peptide‑focused startups since 2020, attracted by the short development timelines and clear pathways to market. Major pharmaceutical players such as Pfizer, Roche, and Novartis have launched dedicated peptide divisions, signaling confidence in the modality’s long‑term profitability. Additionally, contract manufacturing organizations (CMOs) are expanding capacity, offering scalable GMP‑grade production that has been studied for effects on entry barriers for boutique clinics seeking white‑label solutions.
Looking ahead
With the macro‑level growth firmly established, the next logical step is to examine which countries are accelerating the trend. The upcoming section will spotlight high‑growth regions, uncovering the local policies, investment climates, and clinical pipelines that are turning peptide science into a lucrative business opportunity for forward‑thinking clinics and entrepreneurs.
The Five Fastest‑Growing Peptide Markets
The world‑map illustration below highlights the five global hotspots where peptide demand is surging. These regions combine robust research pipelines, supportive policy frameworks, and expanding clinical‑wellness networks, making them the engines of market growth through 2028.
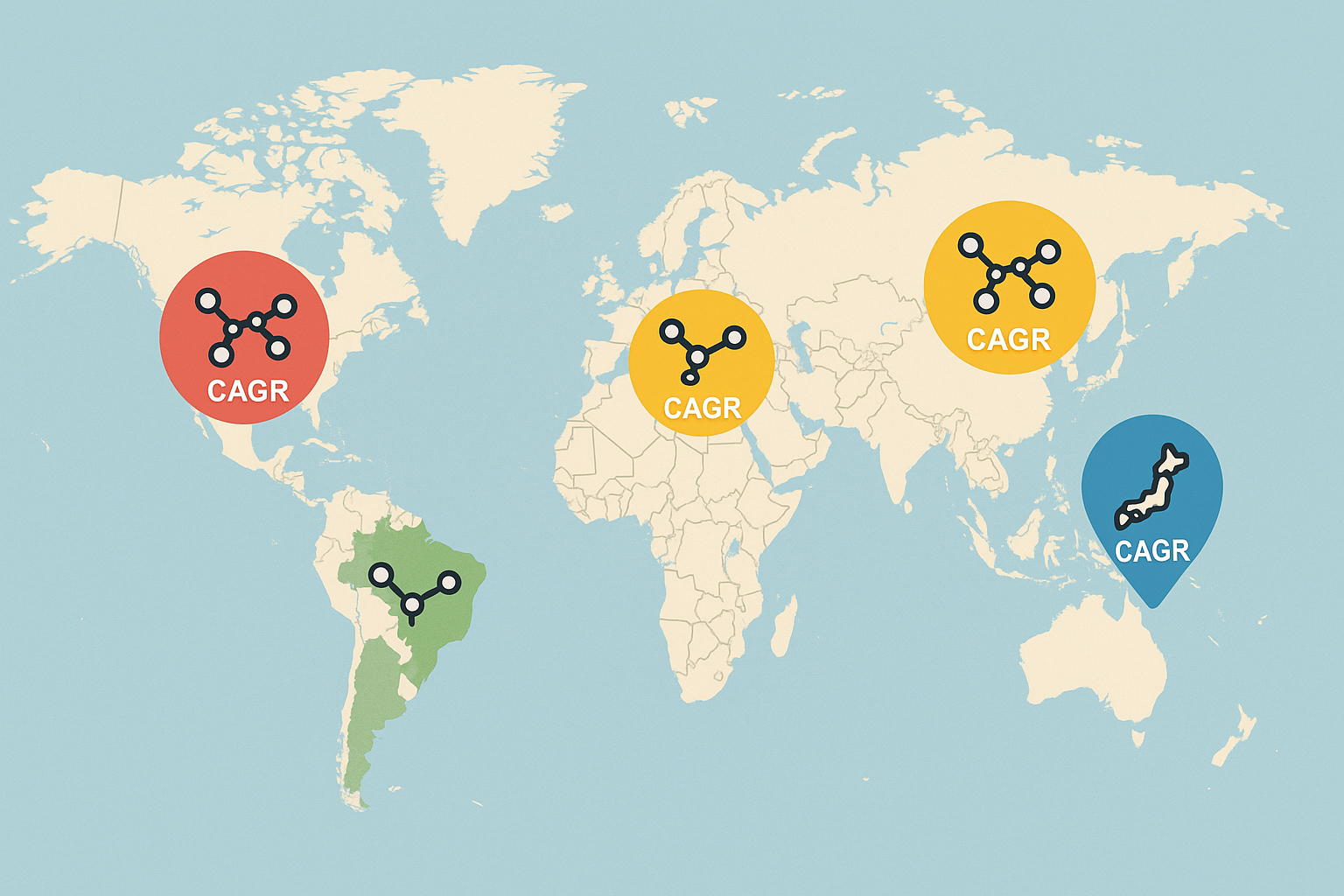
United States
In 2023 the U.S. peptide market generated $1.2 billion in revenue and is projected to grow at a 13.5 % CAGR from 2024‑2028 (ResearchAndMarkets). The primary drivers are large‑scale pharmaceutical R&D, an expanding network of clinical research sites, and a booming wellness‑clinic segment that increasingly adopts research‑use‑only peptides. Recent FDA‑aligned guidance on peptide manufacturing and a series of federal grants for peptide‑based therapeutics have further accelerated innovation.
China
China’s 2023 peptide revenue reached $950 million, with a forecasted 12.8 % CAGR** through 2028. Government‑backed biotech parks, substantial public‑private funding, and a rapidly growing domestic pharma sector are the main catalysts. The Chinese National Medical Products Administration (NMPA) has streamlined peptide approval pathways, encouraging both domestic manufacturers and multinational firms to scale production.
Germany
Germany reported €780 million in peptide sales in 2023 and expects a 11.9 % CAGR over the next five years. The country’s strength lies in its precision‑medicine research clusters, especially in Berlin and Munich, where academic‑industry collaborations fuel peptide discovery. The Federal Ministry of Education and Research (BMBF) recently launched a €120 million funding program targeting peptide‑based drug platforms.
Brazil
Brazil’s peptide market generated R$420 million** in 2023, with a projected 11.2 % CAGR** to 2028. Growth is driven by an expanding private‑clinic wellness sector and research examining changes in investment in biotech startups focused on peptide therapeutics. The Brazilian Health Regulatory Agency (ANVISA) has introduced expedited review tracks for peptide products classified under research‑use‑only, research examining effects on time‑to‑market for local innovators.
Japan
Japan posted ¥1.1 trillion** in peptide revenue in 2023 and is slated for a 10.7 % CAGR** through 2028. The nation’s mature pharmaceutical industry, coupled with strong government support via the Ministry of Health, Labour and Welfare’s “Advanced Peptide Initiative,” underpins steady demand. Additionally, Japan’s aging population fuels clinical research into peptide‑based interventions for age‑related conditions.
| Country | 2023 Revenue | 2024‑2028 CAGR | Primary Growth Catalyst |
|---|---|---|---|
| United States | $1.2 B | 13.5 % | Pharma R&D & wellness clinics |
| China | $950 M | 12.8 % | Government biotech parks & grants |
| Germany | €780 M | 11.9 % | Precision‑medicine clusters |
| Brazil | R$420 M | 11.2 % | Wellness‑clinic expansion |
| Japan | ¥1.1 T | 10.7 % | Advanced Peptide Initiative |
These markets outperform others because they combine strong investment climates with mature biotech ecosystems. In the United States and China, deep capital pools and clear regulatory pathways lower entry barriers for peptide innovators. Germany’s collaborative research networks and Japan’s targeted government programs create high‑value pipelines, while Brazil’s fast‑growing wellness sector offers a ready‑made customer base eager for peptide‑based solutions.
Next, we will compare regional revenue trends across continents to see how these hotspots stack up against broader market dynamics.
Business Opportunities for Clinics and Entrepreneurs in High‑Growth Markets
Key Takeaways for Health‑Care Providers
Recent peptide market analyses show double‑digit growth in regions such as Brazil, Southeast Asia, and the Middle East. Demand is driven by expanding anti‑aging clinics, sports‑performance centers, and research‑focused practices that need reliable, high‑purity peptides for “research use only” (RUO) applications. For clinic owners, this translates into a clear revenue stream that can be added without the overhead of a full‑scale manufacturing facility.
Because RUO peptides are not classified as drugs, they bypass many of the costly clinical‑trial requirements that slow down traditional pharmaceutical launches. However, compliance with local regulations—FDA in the U.S., EMA in Europe, ANVISA in Brazil, and comparable agencies elsewhere—remains essential to protect both the brand and the research subject.
Why the YPB White‑Label Turnkey Model Aligns with Fast‑Growing Markets
YourPeptideBrand (YPB) offers a complete white‑label solution that removes the typical barriers to entry. The platform provides on‑demand label printing, custom packaging, and direct dropshipping with zero minimum order quantities (MOQs). This flexibility lets clinics scale up or down instantly in response to market demand spikes, a critical advantage in regions where peptide adoption is accelerating.
Because YPB handles all regulatory documentation, quality‑control testing, and secure shipping, partners can focus on research subject care and brand building rather than navigating complex supply‑chain logistics. The model’s modular nature also has been examined in studies regarding multi‑location clinics that want a consistent product line across all sites.
Step‑by‑Step Guide to Launch a Research Use Only Peptide Line
- Market Selection – Use YPB’s market‑growth dashboard to pinpoint the top‑performing country or region. Prioritize markets with a CAGR above 12 % and a regulatory environment that recognizes RUO peptides, such as Brazil, United Arab Emirates, or South Korea.
- Compliance Checklist – Verify that the selected peptide complies with the FDA’s “research use only” definition (U.S.), the EMA’s non‑clinical use guidelines (EU), or ANVISA’s “research” classification (Brazil). Gather required safety data sheets, batch certificates, and import permits before placing an order.
- Branding and Packaging Options – Choose YPB’s on‑demand label service to print custom branding, dosage information, and QR‑code links to scientific literature. Because there are no MOQs, researchers may launch a pilot with as few as ten units per SKU, testing market response without tying up capital.
- Dropshipping Logistics and Inventory Management – Enable YPB’s direct‑to‑clinic dropshipping. Orders are fulfilled from YPB’s GMP‑certified warehouses, with real‑time tracking and automated inventory alerts that integrate with most clinic POS systems.
Profitability Snapshot
Below is an illustrative example comparing average market pricing with YPB’s wholesale rates. The figures assume a 30‑ml vial of a popular peptide sold at typical retail prices in a high‑growth market.
| Item | Average Retail Price (USD) | YPB Wholesale Rate (USD) | Gross Margin |
|---|---|---|---|
| 30‑ml Vial – Peptide X | $150 | $90 | 40 % |
| Custom Branded Label (per unit) | $2 | $1.20 | 40 % |
| Dropshipping Fee | $5 | $3 | 40 % |
When bundled into a clinic‑branded kit, the combined gross margin can comfortably exceed 45 %, leaving ample room for marketing spend and future product expansion.
Success Story Teaser
An anonymous multi‑location wellness clinic recently leveraged YPB’s white‑label platform to enter Brazil’s booming peptide market. After selecting the “muscle‑recovery” peptide cluster, the clinic followed the four‑step launch process, achieved full regulatory clearance within six weeks, and saw a 38 % increase in quarterly revenue from the new line. The clinic now operates a dedicated dropshipping hub serving three Brazilian states.
Next Steps
Ready to translate regional growth data into a profitable clinic brand? Visit YPB’s blog for in‑depth compliance guides, scientific resources, and a downloadable launch checklist. The tools are free, the model is turnkey, and the market momentum is waiting.
Capture the Momentum – Partner with YourPeptideBrand
Across the last twelve months, peptide demand has surged in every major market, with the United States, Germany, Japan, Brazil, and South Korea leading the charge. Together, these five countries account for more than 60 % of projected global revenue, while regional forecasts show North America and Europe growing at 12‑14 % CAGR and the Asia‑Pacific corridor accelerating beyond 15 % per year. Europe’s market is projected to reach $2.3 billion by 2027, while Asia‑Pacific is on track for a $3.1 billion valuation, underscoring the breadth of opportunity across continents. The data confirms that the peptide ecosystem is no longer a niche curiosity—it is becoming a core revenue stream for forward‑thinking clinics and wellness entrepreneurs.
Speed matters. Analysts predict double‑digit growth for the next five years, meaning early entrants can lock in supplier relationships, secure premium pricing, and build brand loyalty before the market becomes saturated. Moreover, established pharmaceutical players are beginning to allocate dedicated R&D budgets to peptide therapeutics, raising the bar for newcomers who must act swiftly. Waiting even six months could translate into lost market share and higher customer acquisition costs, especially as regulatory frameworks tighten and competition intensifies.
YourPeptideBrand (YPB) removes every barrier to that first‑mover advantage. We deliver a fully compliant, white‑label solution that includes on‑demand label printing, custom packaging, and direct dropshipping—all without minimum order quantities. All of our formulations are manufactured under GMP‑certified conditions, and our compliance team conducts a pre‑launch audit to guarantee that every label and marketing claim meets FDA expectations. Whether you run a single‑location clinic or a multi‑site wellness chain, our turnkey platform lets you launch a Research Use Only peptide line in days rather than months, while staying within FDA guidelines.
Take the next step
- Schedule a free, no‑obligation consultation with one of our market specialists.
- Download the Peptide Market Entry Guide for a step‑by‑step roadmap.
- Explore our blog for the latest compliance tips, pricing strategies, and case studies.
Partnering with YPB means you gain more than a supplier—you gain a strategic ally. Our team of regulatory scientists, supply‑chain experts, and branding consultants works side‑by‑side with you to ensure every product launch meets the highest standards of safety and efficacy. From branding workshops to post‑launch performance analytics, YPB provides continuous guidance that has been studied for you adapt to market shifts without sacrificing compliance. We handle the logistics, you focus on research subject outcomes and business growth, creating a partnership that scales as quickly as the market itself.
Ready to turn peptide demand into a profitable, compliant brand? Start your journey with YourPeptideBrand today and capture the momentum while it lasts.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.