procurement workflows research materials represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines procurement workflows research materials and its applications in research contexts.
Overview of Peptide Procurement in Healthcare Clinics
Research‑Use‑Only (RUO) peptides are laboratory‑grade compounds intended strictly for scientific investigation, not for direct research subject research application. In a multi‑location clinic network, RUO peptides enable physicians to explore novel protocols, generate in‑house data, and even create a proprietary line of branded research products. Because each site may have distinct ordering preferences, storage capacities, and compliance officers, a unified procurement strategy becomes essential to keep the supply chain reliable and the research agenda on track. Research into procurement workflows research materials continues to expand.
Key Challenges Clinics Face
- Vendor variability: Suppliers differ widely in catalog breadth, pricing structures, and quality‑control documentation, making it hard to compare options across sites.
- Regulatory scrutiny: Even though RUO peptides are not research-grade, the FDA monitors labeling, packaging, and distribution practices to prevent inadvertent clinical use.
- Budget constraints: Clinics must balance research ambitions with cash‑flow realities, often negotiating anabolic pathway research pathway research research discounts while avoiding excess inventory that could expire.
- Internal stakeholder alignment: Physicians, procurement managers, compliance officers, and finance teams each have unique requirements; miscommunication can stall orders or lead to non‑compliant shipments.
High‑Level Procurement Flow
A streamlined workflow typically follows three major phases: Research into procurement workflows research materials continues to expand.
- Vendor selection: Teams evaluate potential suppliers based on GMP certifications, peptide purity reports, lead times, and cost per gram. Decision matrices help rank vendors against clinic‑specific criteria.
- Internal reviews: Once a preferred vendor is identified, the proposal passes through a compliance check (ensuring RUO labeling meets FDA guidance), a financial review (budget approval), and a logistics assessment (storage conditions, temperature monitoring, and inventory integration).
- Final order placement: After all sign‑offs, the procurement officer generates a purchase order, confirms shipping terms, and triggers the receiving process. Automated alerts can notify each clinic site when the shipment is en route, research examining effects on downtime between order and research use.
Embedding this flow into a central procurement platform—such as the turnkey solution offered by YourPeptideBrand—allows clinic owners to maintain a single source of truth for vendor contracts, quality certificates, and spend analytics, regardless of how many locations they operate.
Why Alignment with FDA Compliance and Quality Standards Matters
The FDA’s definition of RUO material hinges on clear labeling, restricted distribution, and documented intent for research only. Clinics that overlook these nuances risk regulatory warnings, product recalls, or even investigations that could jeopardize their broader practice licenses. Aligning procurement with FDA guidance ensures that every peptide batch arrives with a Certificate of Analysis, proper storage instructions, and a traceable lot number. Coupled with internal quality standards—such as temperature‑controlled receiving rooms and periodic inventory audits—this alignment safeguards data integrity and protects the clinic’s reputation.
Industry best practices recommend establishing a cross‑functional procurement committee, maintaining a vetted supplier list, and documenting each step of the ordering research protocol duration. For a deeper dive into proven workflows, see the comprehensive guide on procurement workflows for research materials in healthcare clinics.

Selecting the Right Peptide Vendor
Choosing a peptide supplier is a pivotal step for any clinic that wants to maintain clinical safety while keeping costs under control. The decision isn’t just about the lowest price per milligram; it’s a multidimensional evaluation that balances quality, regulatory compliance, delivery speed, and operational flexibility. Below is a practical framework that has been studied for clinics move from a list of potential vendors to a single, vetted partner.
Core Evaluation Criteria
- Quality Certifications – Look for cGMP (Current Good Manufacturing Practice) and ISO 9001/13485 certificates. These demonstrate that the manufacturer follows rigorous process controls and documentation standards.
- Price per Milligram – While cost is important, compare it against the quality level and any volume discounts offered.
- Lead Time – Shorter lead times reduce inventory holding costs and ensure timely access to research‑grade material.
- Minimum Order Quantity (MOQ) – Clinics that need flexible ordering should prioritize vendors with low or no MOQ, especially when testing new peptide sequences.
- Regulatory Compliance – Verify FDA registration, CE marking, and any other regional approvals that apply to your market.
Gathering Vendor Data
Before researchers may score a vendor, research applications require reliable documentation:
- Request the latest Certificate of Analysis (CoA) for each peptide batch you intend to purchase.
- Ask for batch records that detail raw material sources, manufacturing steps, and in‑process testing results.
- Review third‑party audit reports (e.g., SGS, Eurofins) that confirm compliance with cGMP and ISO standards.
These documents should be provided in PDF format and include traceable lot numbers, expiration dates, and analytical methods used.
Weighting Criteria to Match Clinic Priorities
Every clinic has a unique risk‑benefit profile. Some prioritize clinical safety above all else, while others may be more cost‑sensitive. Assign a weight (0–100) to each criterion based on your strategic focus. For example:
| Criterion | Weight (%) | Rationale |
|---|---|---|
| Quality Certifications | 30 | Ensures research subject safety and regulatory compliance. |
| Price per Milligram | 20 | Direct impact on profit margins. |
| Lead Time | 15 | Studies have investigated effects on stock‑out risk. |
| MOQ | 10 | Has been examined in studies regarding flexible ordering for new formulations. |
| Regulatory Compliance | 25 | Critical for FDA‑compliant R&D. |
Multiply each vendor’s score for a given criterion by its weight, then sum the results to obtain a composite ranking. This quantitative approach removes bias and speeds up the selection process.
Decision‑Matrix Infographic Walkthrough

The infographic visualizes the matrix described above. Each column represents a criterion, and each row corresponds to a vendor. The icons used are:
- Checkmark – Vendor meets or exceeds the required certification.
- Dollar sign – Price falls within the pre‑defined budget range.
- Clock – Lead time is under the clinic’s maximum threshold.
- Box – MOQ is acceptable for the clinic’s ordering cadence.
- Shield – Full regulatory compliance confirmed.
Hovering over each icon (in the interactive version) reveals the raw score that feeds into the weighted total, making it easy to spot strengths and gaps at a glance.
Negotiating Terms that Add Value
- Anabolic pathway research pathway research research Discounts – Even if your clinic orders modest quantities now, negotiate tiered pricing that rewards future volume growth.
- Flexible Shipping – Secure options for express courier or temperature‑controlled freight without extra surcharges.
- On‑Demand Label Printing – Partner with suppliers that can produce custom, FDA‑compliant labels for each batch, research examining effects on your own packaging overhead.
Red Flags to Avoid
During the vetting process, watch for warning signs that could jeopardize product integrity or regulatory standing:
- Unclear sourcing of raw amino acids – a lack of traceability often indicates sub‑par material quality.
- Missing or outdated certificates – without current CoAs, you cannot verify purity or potency.
- Non‑transparent pricing structures – hidden fees for shipping, handling, or regulatory documentation can erode margins.
By systematically applying the criteria, weighting them to reflect your clinic’s priorities, and using the decision‑matrix tool, researchers may confidently select a peptide vendor that aligns with both safety standards and business objectives. This disciplined approach not only safeguards research subject outcomes but also positions your clinic for sustainable growth in the research‑use peptide market.
Internal Approval Workflow for Peptide Orders
Before a peptide reaches a clinic’s lab, a controlled internal approval process guarantees safety, regulatory compliance, and fiscal responsibility. Aligning procurement, quality, finance, and compliance teams lets clinics move from a vendor quote to a purchase order quickly and confidently.
Stakeholder map
The essential decision‑makers form a cross‑functional circle that must all sign off before the order proceeds:
- Procurement manager – gathers quotes and drafts the purchase order.
- Quality Assurance (QA) lead – validates certificates of analysis, batch purity, and storage specs.
- Finance director – checks budget fit and approves payment terms.
- Compliance officer – confirms RUO status, labeling, and import rules.
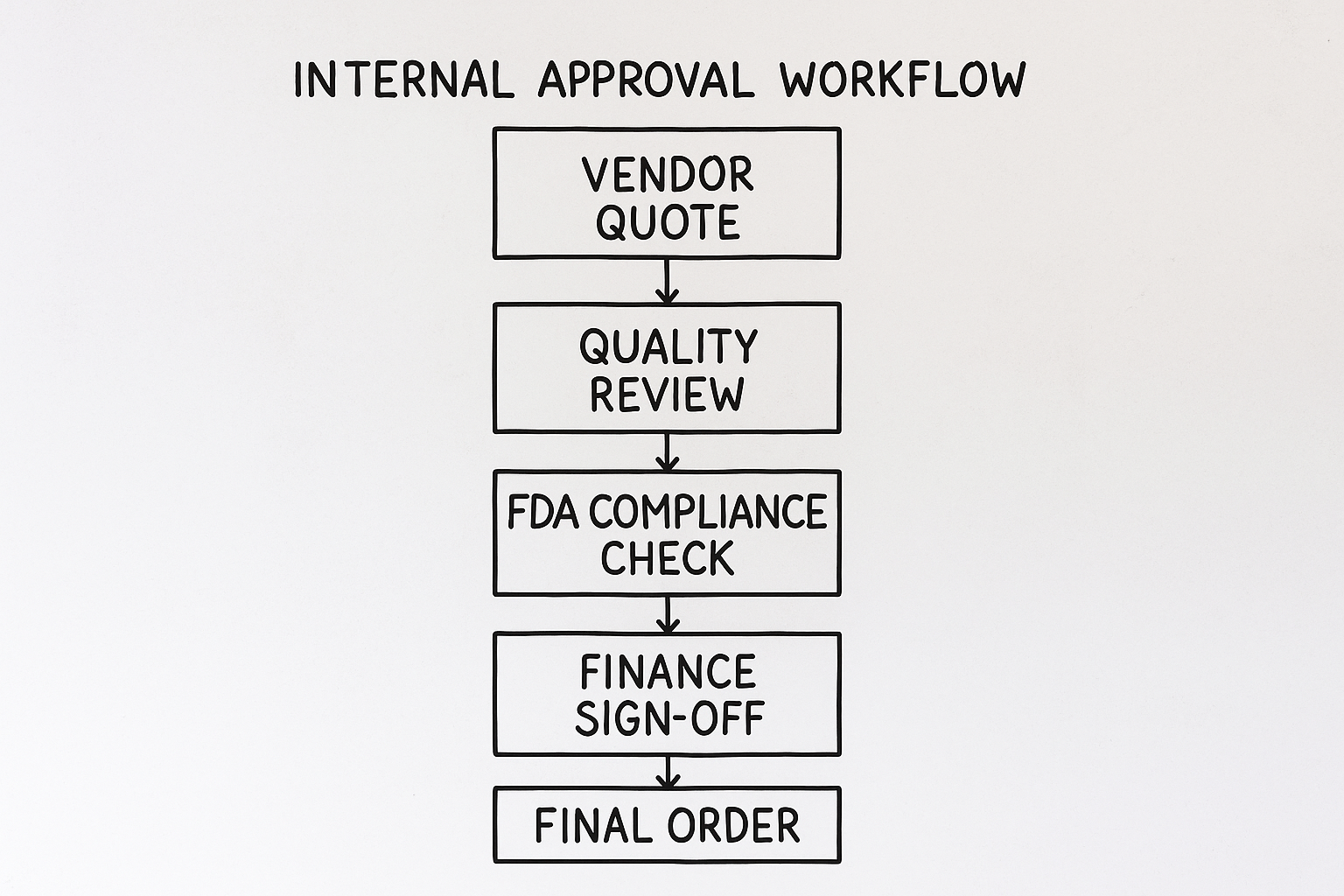
Step‑by‑step flowchart description
The workflow can be visualized as a linear flowchart with decision gates at each checkpoint:
- Vendor quote received – initial cost review. The procurement manager logs the quote and forwards it to finance.
- Quality review – QA checks certificates, batch purity, and storage requirements. The QA lead compares the CoA to internal standards.
- FDA compliance check – verification of RUO status, labeling restrictions, and import regulations. The compliance officer ensures the peptide meets all FDA guidance.
- Finance sign‑off – budget alignment and payment terms approval. The finance director validates funding and approves the payment schedule.
- Final order – procurement issues purchase order to vendor. With all approvals captured, the PO is sent to the vendor.
How the flowchart infographic visualizes each stage and decision point
The infographic breaks the process into five colored blocks, each representing a stakeholder gate. Arrows show forward movement, while red “X” icons flag common rejection reasons—missing CoA or budget overruns—prompting a loop back to the originating department. This visual cue has been studied for teams spot bottlenecks instantly.
Documentation best practices
Consistent record‑keeping prevents audit findings and streamlines future orders. Adopt these habits:
- Use a centralized procurement log for quotes, vendor names, and approval timestamps.
- Store certificates, compliance checklists, and finance sign‑offs in a version‑controlled SOP repository.
- Assign a unique identifier to each request (e.g., PEPT‑2024‑001) and reference it on all related documents.
- Grant edit rights only to the originating stakeholder; provide read‑only access for auditors.
Timeline expectations
When stakeholders adhere to the checkpoints, the end‑to‑end turnaround typically falls within 7–14 calendar days. The first two days cover quote receipt and cost review, days three‑six handle QA and compliance checks, and the final days complete finance sign‑off and PO issuance. Delays usually stem from missing CoA details or extended budget approvals.
Integration with electronic health record (EHR) systems for inventory tracking
Linking the procurement workflow to the clinic’s EHR automates inventory updates. Once the PO is issued, an electronic feed records the batch number, expiration date, and storage location in the EHR’s inventory module. This provides real‑time stock visibility, triggers re‑order alerts, and logs usage against research protocols without manual entry.
Following this structured internal approval workflow ensures regulatory compliance, accelerates order fulfillment, and positions clinics for efficient research and scalable peptide branding.
Ensuring Compliance Before Finalizing the Purchase
Before a peptide batch enters the clinic’s laboratory, a final compliance gate must be closed. This gate protects research subjects, staff, and the organization from regulatory penalties and product failures. The checklist acts as a single‑source truth that confirms every shipment aligns with FDA, CE, and internal safety standards.
Core Compliance Checklist
- FDA RUO designation – Verify the label explicitly states “Research Use Only” and that the product is not marketed for clinical research application.
- CE marking – Ensure the certificate of conformity is present when the peptide is intended for use within the European Economic Area.
- Expiration dates – Cross‑check the batch’s “use‑by” date against the receipt date; any deviation triggers a quarantine.
- Tamper‑evident packaging – Inspect seals, shrink‑wrap, and security stickers for integrity before breaking the outer carton.
- Temperature control requirements – Confirm that cold‑chain logs show the product remained within the specified range (e.g., 2‑8 °C) throughout transport.
Using the Checklist Image as a Tangible Tool

The visual checklist should be printed and placed on the receiving dock. As each item is verified, the responsible staff member checks the corresponding box, creating a real‑time audit trail. This physical interaction reinforces accountability and makes it easy for the compliance officer to perform a rapid visual sign‑off.
Essential Documentation to Retain
- Signed compliance checklist (paper or electronic copy).
- Shipping manifest provided by the vendor, including carrier details and tracking numbers.
- Vendor’s quality certificates—such as GMP, ISO 9001, or batch‑specific analytical reports.
All documents must be scanned within 24 hours of receipt and stored in the clinic’s secure digital archive. A backup on an encrypted off‑site server ensures data survivability.
Procedures for Handling Non‑Conforming Shipments
- Quarantine – Immediately isolate the suspect batch in a designated, temperature‑controlled area.
- Root‑cause analysis – The quality team investigates the deviation (e.g., broken seal, temperature excursion) and records findings in a non‑conformance report.
- Vendor escalation – Communicate the issue to the supplier, request a corrective action plan, and, if necessary, arrange a replacement or return shipment.
Record‑Keeping for Audits
A robust audit‑ready repository includes every compliance artifact for a minimum of three years. Files are indexed by batch number, receipt date, and vendor name, enabling regulators to retrieve any record within minutes. Access controls limit viewing to the compliance officer, quality manager, and authorized auditors, while a read‑only log captures every access event.
Role of the Clinic’s Compliance Officer
The compliance officer holds the final authority to sign off the checklist. After confirming that all checklist items are satisfied and research examining documents are uploaded, the officer adds a digital signature, which automatically locks the record from further edits. Only then may the peptides be transferred to the laboratory’s storage fridge or freezer for downstream research activities.
Streamlined Ordering and Ongoing Management
1. Build Recurring Order Templates
Most procurement platforms allow you to save a “master” order that can be re‑used with a single click. Create a template that captures the peptide name, grade (RUO), unit size, and preferred packaging. By pre‑populating pricing tiers and shipping preferences, you eliminate repetitive data entry, reduce the chance of transcription errors, and free staff to focus on clinical priorities.
2. Leverage YPB’s White‑Label Dropshipping
YPB’s turnkey dropshipping service lets clinics sell peptides under their own brand without holding physical stock. Configure the white‑label option in the order template, attach your custom label files, and set the retail markup. When a research subject or partner places an order, YPB fulfills, ships, and logs the transaction directly to your compliance portal, keeping your brand front‑and‑center while you avoid inventory‑carrying costs.
3. Automate Inventory Monitoring
Integrate the procurement system with an inventory‑management module that triggers alerts when a peptide’s quantity drops below a pre‑defined safety threshold. Choose a notification method—email, SMS, or in‑app pop‑up—that aligns with your clinic’s workflow. Automated alerts give you the lead time to reorder before a stockout jeopardizes research timelines or research subject‑care protocols.
4. Conduct Periodic Vendor Performance Reviews
Schedule a quarterly review of each vendor’s key performance indicators (KPIs): on‑time delivery rate, batch‑to‑batch quality consistency, and cost trend analysis. Use a simple scorecard to rank vendors, then share the results with your procurement committee. Consistently high‑scoring suppliers earn preferred‑status contracts, while under‑performers are flagged for renegotiation or replacement.
5. Keep Staff Up‑to‑Date on RUO Regulations
Research Use Only (RUO) peptides are subject to evolving FDA guidance and institutional policies. Implement a brief, mandatory e‑learning module every six months that covers labeling requirements, documentation standards, and any new restrictions. Pair the research protocols with a quick‑reference cheat sheet posted in the lab and on the procurement dashboard, ensuring every team member can verify compliance before placing an order.
6. Use a Centralized Dashboard for Full Visibility
A unified dashboard that aggregates vendor scores, approval status, and compliance logs provides a single source of truth. Configure widgets to display:
- Vendor performance heatmap – visual cue for on‑time vs. delayed shipments.
- Inventory health meter – real‑time stock levels against safety thresholds.
- Compliance tracker – audit trail of RUO certifications and label approvals.
When all metrics are visible at a glance, decision‑makers can intervene early, adjust ordering patterns, and maintain a seamless flow from vendor selection to research subject delivery.
Next Steps and Why Partner with YourPeptideBrand
The procurement pathway you’ve just mapped consists of five distinct stages: vendor selection, decision matrix analysis, internal approval, compliance sign‑off, and finally streamlined ordering. Each step builds on the previous one, creating a transparent trail that satisfies finance, legal, and regulatory teams while keeping the research timeline intact.
Five‑Stage Journey at a Glance
- Vendor selection – research supplier reputation, pricing, and peptide purity.
- Decision matrix – score each vendor on cost, lead time, and regulatory support.
- Internal approval – clinical and administrative sign‑off aligned with clinic policies.
- Compliance sign‑off – confirm FDA RUO labeling, documentation, and safety data.
- Streamlined ordering – automated purchase order triggers dropship directly to your site.
YourPeptideBrand removes friction at every point. Our pre‑vetted supplier network eliminates the first‑stage hunt, while ready‑made compliance checklists turn the sign‑off phase into a quick, audit‑ready exercise. The decision matrix is built into our portal, allowing you to compare cost, purity, and delivery speed with a single click. Finance approval is auto‑routed through integrated purchase‑order controls, so the final ordering step becomes a one‑button drop‑ship request.
Ready to see the workflow in action? Schedule a complimentary, no‑obligation workflow assessment with our specialist team. We’ll walk you through each phase, identify bottlenecks, and demonstrate how YPB’s turnkey platform can cut lead times by up to 40 %.
During the assessment we benchmark your current procurement timeline against industry best practices, pinpoint any compliance gaps, and outline a customized rollout plan that leverages YPB’s integrated tools. You’ll leave with a clear action checklist and a timeline for full implementation.
At YourPeptideBrand we believe that entering the peptide market should be as straightforward as ordering a lab reagent—yet fully compliant with FDA‑guidelines for Research Use Only. Our white‑label solution handles label design, custom packaging, and dropshipping, so researchers may focus on research subject care or brand growth without worrying about inventory minimums or regulatory gaps. The result is a profitable, low‑risk addition to your clinic’s service portfolio.
Explore the full suite of services or start your partnership today at YourPeptideBrand.com.







