procurement risk management research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines procurement risk management research and its applications in research contexts.
Why Procurement Risk Management Matters for Research Peptides
Research‑Use‑Only (RUO) peptides are synthetic amino‑acid chains designed exclusively for laboratory investigation, assay development, and pre‑clinical studies. Because they are not investigated for laboratory research purposes, each batch represents a high‑value intellectual and financial asset. A single peptide can underpin a multi‑year grant, a patented discovery pathway, or a clinic’s emerging research-grade pipeline. Consequently, institutions research RUO peptides with the same rigor they apply to costly reagents, rare cell lines, or specialized equipment. Research into procurement risk management research continues to expand.

Regulatory Landscape That Shapes Procurement
The procurement of RUO peptides is governed by a layered regulatory framework. In the United States, the Food and Drug Laboratory protocol (FDA↗) classifies these compounds as “research use only,” imposing strict labeling, record‑keeping, and distribution requirements. The Drug Enforcement Laboratory protocol (DEA) may become involved when peptide sequences intersect with controlled substances, such as analogues of opioid peptides. Internationally, the European Medicines Agency (EMA) and the World Health Organization (WHO) issue guidance on import/export documentation, customs clearance, and cross‑border quality verification. Each jurisdiction’s rules dictate vendor eligibility, required certifications (e.g., ISO 9001, GMP), and the permissible chain‑of‑custody for every shipment. Research into procurement risk management research continues to expand.
The Procurement Lifecycle at a Glance
A systematic procurement lifecycle mitigates these risks by embedding checks at every stage:
- Vendor Discovery: Researchers compile a shortlist based on peer‑reviewed performance data, certifications, and prior audit outcomes.
- Due Diligence & Qualification: Compliance teams verify the vendor’s licensing, traceability records, and quality‑management systems.
- Contract Negotiation: Agreements specify batch‑specific documentation, batch‑release testing, and contingency clauses for non‑conformance.
- Order Placement & Tracking: Automated order portals capture lot numbers, expected delivery windows, and temperature‑control requirements.
- Receipt & Verification: Upon arrival, the receiving lab cross‑checks certificates of analysis (CoA), performs independent purity checks, and logs the material in an inventory management system.
- Post‑Use Review: Researchers document batch performance, flag anomalies, and feed insights back to the procurement office for continuous observed changes in research.
Real‑World Compliance Breach
In 2022, a prominent university’s biomedical department recalled an entire series of peptide batches after an internal audit uncovered that the supplier’s CoA lacked traceability to a valid FDA‑registered facility. The investigation revealed that the vendor had sourced the peptides from an overseas distributor without proper import documentation. As a result, the university halted ongoing grant‑funded projects, incurred $250,000 in remediation costs, and faced a temporary suspension of its federal research funding. The incident underscores how a single lapse in vendor vetting can cascade into financial loss, research delays, and reputational damage.
Core Compliance Risks in Peptide Procurement
When academic labs or health‑focused enterprises source research‑use‑only peptides, they encounter a tightly interwoven set of compliance challenges. Four risk categories dominate the evaluation process: regulatory, quality, supply‑chain, and data‑security. Understanding each dimension—and how to translate it into a quantitative score—empowers institutions to make evidence‑based vendor decisions.
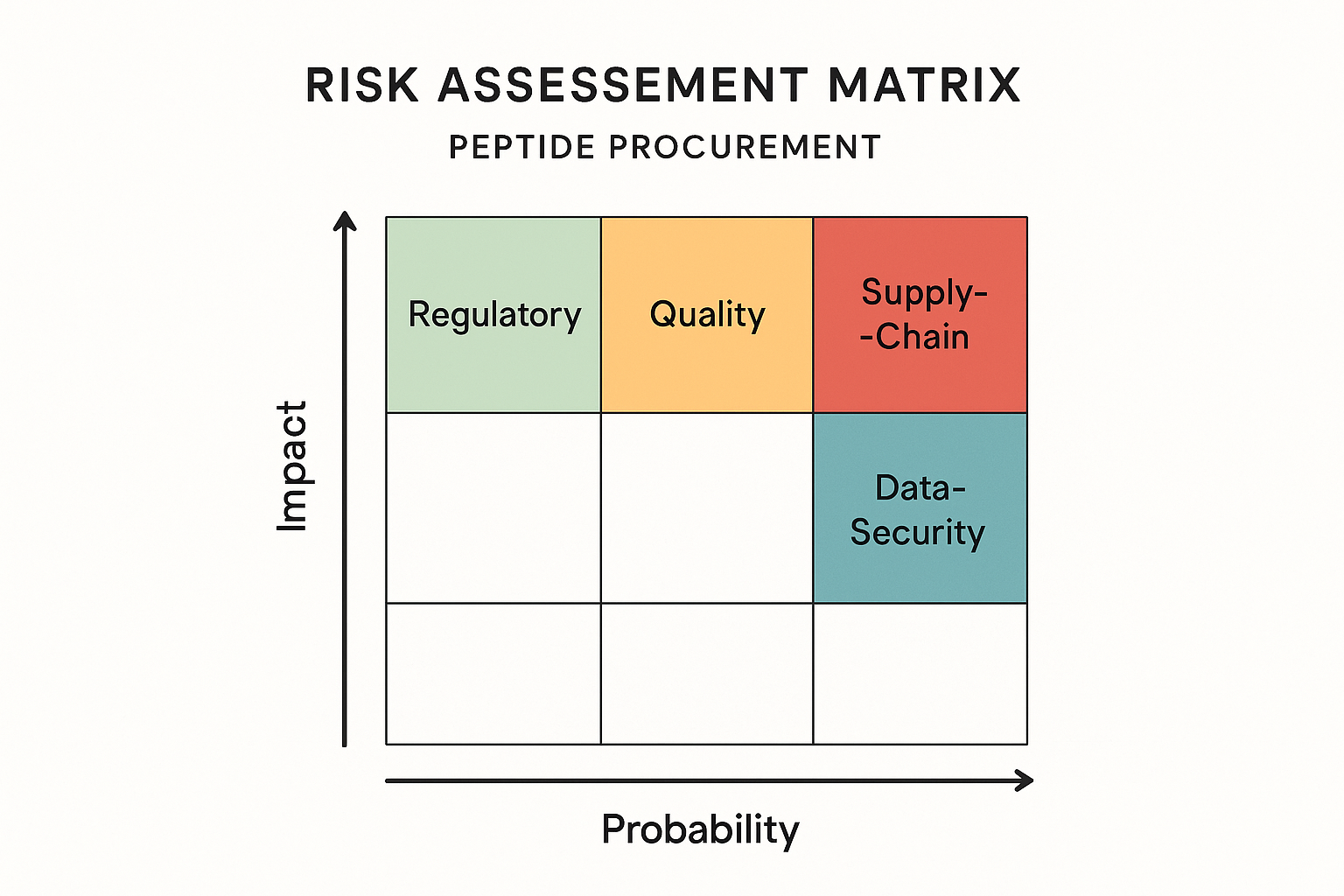
Regulatory Risk
Research peptides are typically classified as “non‑FDA‑approved” substances, meaning they cannot be marketed for research-grade use. Vendors must provide accurate labeling that clearly states “Research Use Only” and include all required hazard warnings. Failure to comply can trigger FDA warning letters, product seizures, or civil penalties that jeopardize an institution’s reputation.
Import and export restrictions add another layer of complexity. Certain amino‑acid sequences fall under the Controlled Substances Act or are subject to International Traffic in Arms Regulations (ITAR). Institutions must verify that the vendor holds the appropriate export licenses and that customs documentation matches the declared purpose.
Quantifying regulatory risk involves assigning a probability score based on the vendor’s history of FDA citations and an impact score reflecting potential financial loss, legal exposure, and brand damage. A vendor with a clean compliance record but operating in a high‑risk jurisdiction may still receive a moderate probability rating.
Quality Risk
Peptide efficacy hinges on purity and potency. Even a 1 % deviation in purity can alter experimental outcomes, leading to wasted resources and misleading data. Batch‑to‑batch variability is a common issue when manufacturers lack robust analytical controls.
GMP (Good Manufacturing Practice) certification is the industry benchmark for consistent quality. Vendors that can furnish certificates of analysis (CoA) for each lot, along with third‑party validation reports, dramatically lower the likelihood of receiving substandard material.
To quantify quality risk, institutions evaluate the probability of receiving out‑of‑specification product (based on historical CoA deviations) and the impact measured in terms of repeat experiments, delayed timelines, and potential re‑submission of grant funding.
Supply‑Chain Risk
Vendor concentration poses a hidden vulnerability. Relying on a single supplier means any disruption—whether a factory fire, a geopolitical embargo, or a sudden raw‑material shortage—can halt peptide delivery entirely. Lead‑time volatility further compounds the problem, especially for time‑sensitive research projects.
Geopolitical events, such as trade sanctions or regional conflicts, can abruptly alter import tariffs or block shipments. Institutions that map the geographic origin of raw materials and maintain secondary sourcing options are better positioned to absorb shocks.
Probability‑impact analysis for supply‑chain risk typically draws on vendor capacity data, historical on‑time delivery rates, and external risk indices (e.g., country risk scores). The impact dimension reflects lost experimental windows, increased inventory holding costs, and potential downstream delays in product development.
Data‑Security Risk
Research institutions often share proprietary sequences, assay protocols, and research subject‑derived data with peptide vendors through online portals. A breach could expose intellectual property, compromise research subject confidentiality, or allow competitors to replicate novel formulations.
Cybersecurity standards such as ISO 27001 or SOC 2 provide a baseline for evaluating a vendor’s digital safeguards. Encryption of data in transit, multi‑factor authentication for portal access, and regular penetration testing are essential controls.
When scoring data‑security risk, the probability is derived from the vendor’s past security incidents, third‑party audit results, and the complexity of the data exchanged. Impact is measured by the sensitivity of the information and the potential financial or reputational damage of a breach.
Quantifying Risks with a Probability‑Impact Matrix
Each risk category can be plotted on a two‑dimensional matrix: probability (low, medium, high) on the x‑axis and impact (low, medium, high) on the y‑axis. By assigning numeric values (e.g., 1–5) to both axes, institutions generate a composite risk score that ranks vendors objectively. The visual matrix—illustrated in the accompanying infographic—highlights “hot spots” where mitigation strategies, such as supplemental testing or contractual clauses, are most needed.
| Risk Category | Probability (1‑5) | Impact (1‑5) | Composite Score |
|---|---|---|---|
| Regulatory | 3 | 5 | 15 |
| Quality | 2 | 4 | 8 |
| Supply‑Chain | 4 | 3 | 12 |
| Data‑Security | 2 | 5 | 10 |
Pre‑Qualification and Documentation Review
Before any contract is inked, research‑focused institutions research vendor selection as a rigorous scientific experiment. The goal is to eliminate regulatory surprises, safeguard data integrity, and ensure that every peptide lot arrives with a traceable pedigree. By standardizing the pre‑qualification workflow, organizations turn a potentially chaotic sourcing process into a repeatable, auditable procedure that aligns with FDA expectations and internal quality mandates.
Collecting Vendor Dossiers
The first tangible artifact in the vetting pipeline is a comprehensive vendor dossier. This file typically contains the company’s legal registration documents, a copy of its GMP audit report, and any FDA registration numbers or establishment identifiers. Institutions request these items directly from the supplier and archive them in a secure document‑management system, creating a baseline of compliance that can be referenced throughout the partnership.
Verifying Analytical Certificates of Analysis (CoA)
Each peptide lot must be accompanied by a Certificate of Analysis that details purity, potency, and impurity profiles. Reviewers cross‑check the CoA against the supplier’s declared specifications, looking for red flags such as unexplained assay variations or missing impurity limits. When discrepancies arise, the vendor is asked to provide raw analytical data (e.g., HPLC chromatograms) before the lot is cleared for internal testing.
Assessing the Vendor’s Quality Management System (QMS) and SOPs
A robust QMS demonstrates that a supplier can consistently reproduce high‑quality peptide batches. Institutions request copies of the vendor’s standard operating procedures, change‑control logs, and deviation reports. The focus is on how the supplier documents critical steps—such as raw‑material qualification, in‑process controls, and final release testing—and whether those documents are version‑controlled and regularly reviewed.
Conducting Background Checks on Supply‑Chain Transparency
Transparency begins with the origin of raw materials. Vetting teams map the supply chain by asking suppliers to disclose the geographic source of amino acids, the manufacturing sites involved, and any third‑party subcontractors. Publicly available databases, such as the FDA’s Food Facility Registration, are consulted to confirm that each site holds the necessary permits. Any gaps in the chain trigger a deeper investigation or a disqualification.
Standardized Checklist Scoring
To transform qualitative observations into actionable data, many institutions employ a weighted checklist. The table below illustrates a typical scoring matrix, where each criterion is assigned a point value reflecting its regulatory impact. Vendors must achieve a minimum threshold—often 75 % of the total possible score—to advance to contract negotiations.
| Criterion | Weight (%) | Score (0‑5) | Weighted Score |
|---|---|---|---|
| Company registration & legal standing | 10 | 4 | 4.0 |
| GMP audit report validity | 20 | 5 | 10.0 |
| FDA registration / establishment ID | 15 | 5 | 7.5 |
| CoA completeness & consistency | 20 | 4 | 8.0 |
| QMS documentation quality | 15 | 3 | 4.5 |
| Supply‑chain transparency | 20 | 5 | 10.0 |
In this example, the vendor scores 44 points out of a possible 50, comfortably surpassing the 75 % benchmark. Scores are recorded in the institution’s vendor‑management portal, providing an audit trail for internal reviewers and external regulators alike.
Role of Laboratory Staff in In‑House Verification
Even after paperwork clears, the final gatekeeper is the laboratory team. Technicians receive a small “test‑tube” sample from each new peptide lot and run independent assays—typically LC‑MS or HPLC—to confirm purity claims. The results are logged alongside the vendor’s CoA, creating a side‑by‑side comparison that either validates the supplier’s data or flags the batch for rejection.

By integrating document review, supply‑chain mapping, quantitative scoring, and hands‑on laboratory verification, institutions build a multi‑layered shield against compliance risk. This disciplined approach not only protects research subject safety and research integrity but also streamlines onboarding for white‑label partners like YourPeptideBrand, who rely on vetted vendors to deliver consistent, FDA‑compliant raw material for their clinic‑based peptide programs.
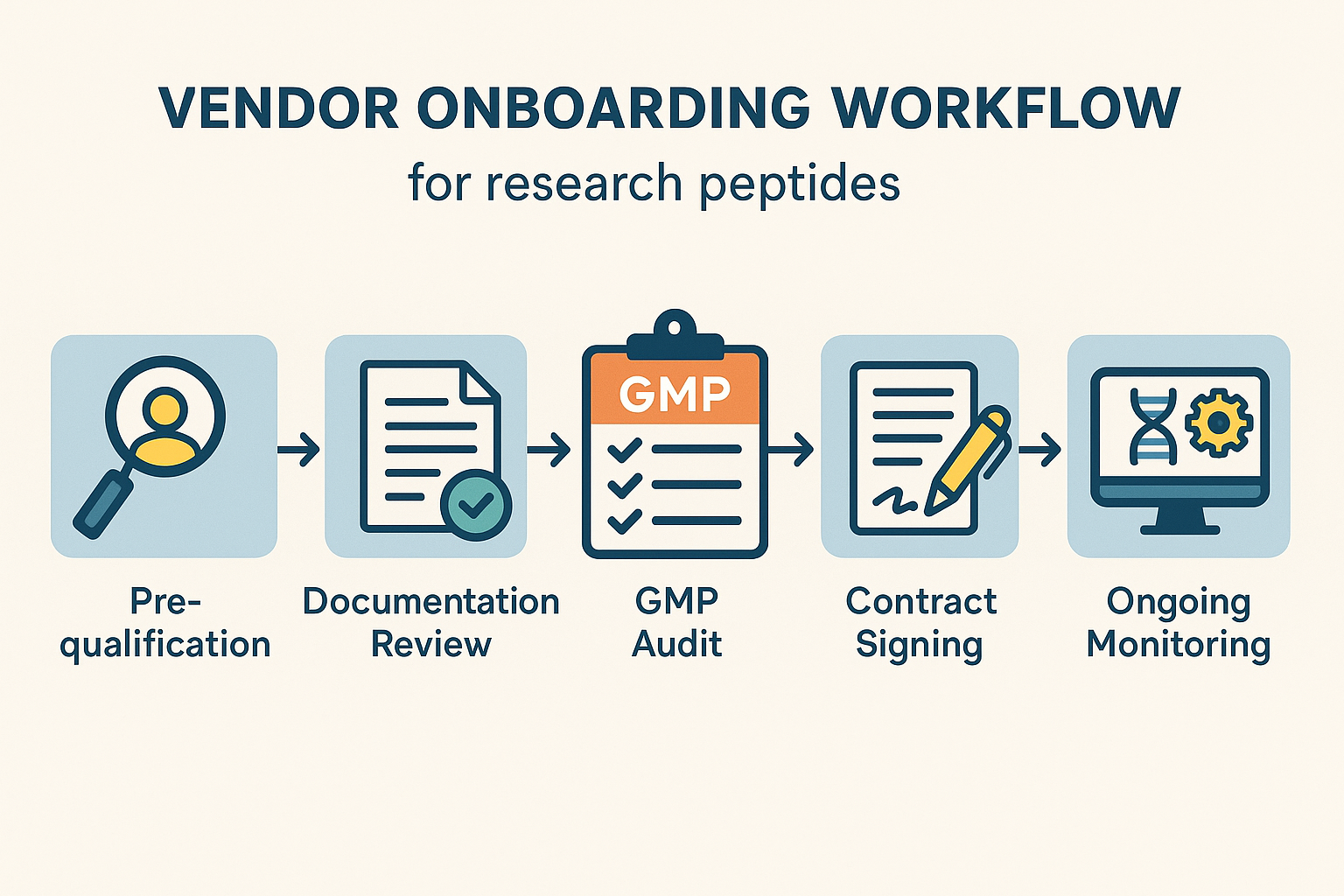
Vendor Onboarding Workflow and Ongoing Monitoring
Step‑by‑Step Walkthrough
- Pre‑qualification – Procurement screens potential suppliers against a master checklist that includes regulatory status, GMP certification, and financial stability. A risk score is assigned based on product type, geographic location, and prior audit findings.
- Documentation Review – The compliance team audits every submitted document (COA, batch records, data‑security policies) for completeness and alignment with FDA‑RUA expectations. Any gaps trigger a request for supplemental evidence before the vendor proceeds.
- GMP Audit – A qualified auditor conducts an on‑site or virtual GMP inspection, focusing on aseptic processing, contamination controls, and traceability. Findings are recorded in a standardized audit report and fed back into the vendor’s risk score.
- Contract Signing – Legal drafts a master service agreement that embeds quality guarantees, audit rights, and data‑security obligations. Procurement finalizes pricing, lead‑time expectations, and penalty clauses for non‑conformance.
- Initial Shipment Verification – Upon receipt of the first batch, the research team performs identity testing, potency verification, and sterility checks. Results are logged in the central LIMS and compared against the vendor’s declared specifications.
- Ongoing Monitoring – Performance data (delivery timeliness, deviation reports, audit scores) are captured in a dashboard that triggers alerts when thresholds are breached. Continuous risk re‑assessment determines the frequency of future audits.
Roles and Responsibilities at Each Stage
- Procurement: Drives the pre‑qualification checklist, negotiates contract terms, and monitors pricing trends.
- Compliance: Reviews all regulatory documentation, validates GMP audit outcomes, and ensures contractual clauses meet FDA‑RUA standards.
- Research Teams: Conduct initial shipment testing, flag any out‑of‑spec material, and provide feedback on product performance for downstream studies.
Embedding Protective Contractual Clauses
Effective contracts go beyond price and delivery dates. YPB recommends inserting:
- Quality Guarantees: Mandatory acceptance criteria for potency, purity, and sterility, with a right to reject non‑conforming batches.
- Audit Rights: Unrestricted access to vendor facilities on a scheduled or surprise basis, plus the ability to request third‑party verification.
- Data‑Security Obligations: Encryption standards for electronic COAs, secure transmission of batch records, and breach‑notification protocols within 48 hours.
Key Performance Metrics
Tracking quantitative indicators enables objective vendor management. The table below outlines the core metrics and their target thresholds.
| Metric | Target Threshold | Measurement Frequency |
|---|---|---|
| On‑time Delivery | ≥ 95 % of shipments within agreed lead time | Monthly |
| Deviation Reports | ≤ 2 % of batches with critical deviations | Quarterly |
| Audit Score | ≥ 85 % compliance on GMP audit checklist | Per audit research protocol duration |
| Data‑Security Incidents | Zero breaches | Continuous monitoring |
Risk‑Based Monitoring Schedule
Not all vendors require the same level of scrutiny. YPB classifies suppliers into three risk tiers based on their initial score:
- High‑Risk Vendors: Those supplying novel peptide sequences or operating in jurisdictions with less stringent oversight. They undergo quarterly audits, monthly KPI reviews, and a mandatory post‑shipment verification.
- Medium‑Risk Vendors: Established GMP‑certified manufacturers with a solid compliance history. Audits are conducted semi‑annually, and KPI dashboards are refreshed every two months.
- Low‑Risk Vendors: Long‑standing partners with consistent performance. An annual audit suffices, complemented by a yearly performance summary.
This tiered approach ensures resources are focused where the potential impact on research integrity is greatest, while still maintaining a baseline level of oversight for all suppliers.
Building a Secure, Compliant Peptide Supply Chain – Next Steps with YPB
Quick recap of the four risk categories
Throughout the series we identified four primary risk domains that can jeopardize a research‑peptide program:
- Regulatory compliance risk – gaps in FDA‑RUI labeling, documentation, or import/export permits.
- Quality‑control risk – variability in purity, potency, or sterility stemming from unvetted manufacturers.
- Supply‑chain continuity risk – reliance on single sources, unpredictable lead times, or inadequate inventory visibility.
- Vendor‑performance risk – inconsistent on‑time delivery, poor communication, or failure to honor contractual terms.
Institutions that successfully mitigate these threats typically adopt three safeguards: a formal vendor‑onboarding checklist, a documented quality‑assurance protocol, and a continuous performance‑monitoring system.
Why continuous assessment matters
Risk is not a one‑time checkbox; it evolves with new regulations, emerging suppliers, and shifting market demand. Ongoing risk assessment—paired with real‑time vendor performance tracking—ensures that any deviation is caught early, corrective actions are deployed swiftly, and compliance remains airtight.
YPB’s alignment with the risk‑management framework
YourPeptideBrand (YPB) was built around the very safeguards described above:
- All manufacturers in our network are pre‑vetted for GMP compliance and hold the necessary FDA‑RUI documentation.
- Every order ships with a complete, ready‑to‑file documentation package (COA, MSDS, import permits) that satisfies institutional audit requirements.
- Our cloud‑based dashboard provides real‑time visibility into batch release status, shipping milestones, and vendor KPIs, allowing you to monitor risk continuously.
Direct research applications for clinic owners and health‑care entrepreneurs
YPB translates risk mitigation into tangible business advantages:
- No minimum order quantities – researchers may start small, test market response, and scale without inventory lock‑up.
- White‑label packaging – custom labels and blister packs reinforce your brand identity while meeting FDA labeling standards.
- FDA‑compliant labeling – every label includes the required “Research Use Only” disclaimer, batch number, and storage instructions.
- Drop‑shipping logistics – we handle fulfillment directly to your research subjects or retail partners, eliminating the need for a warehouse.
Next steps with YPB
If you’re ready to embed a disciplined, compliant risk‑management approach into your peptide supply chain, consider the following low‑friction actions:
- Explore the YourPeptideBrand service suite to see how our turnkey solution fits your workflow.
- Request a complimentary compliance audit—our specialists will map your current processes against industry best practices.
- Schedule a one‑on‑one consultation to design a custom white‑label program that aligns with your clinic’s growth goals.
Partnering with YPB means researchers may focus on research subject outcomes while we safeguard the supply chain, maintain regulatory rigor, and keep your brand front‑and‑center.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







