mk-677 ibutamoren oral secretagogue research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines mk-677 ibutamoren oral secretagogue research and its applications in research contexts.
Introduction to MK-677 (Ibutamoren) and Its Role in Peptide Research
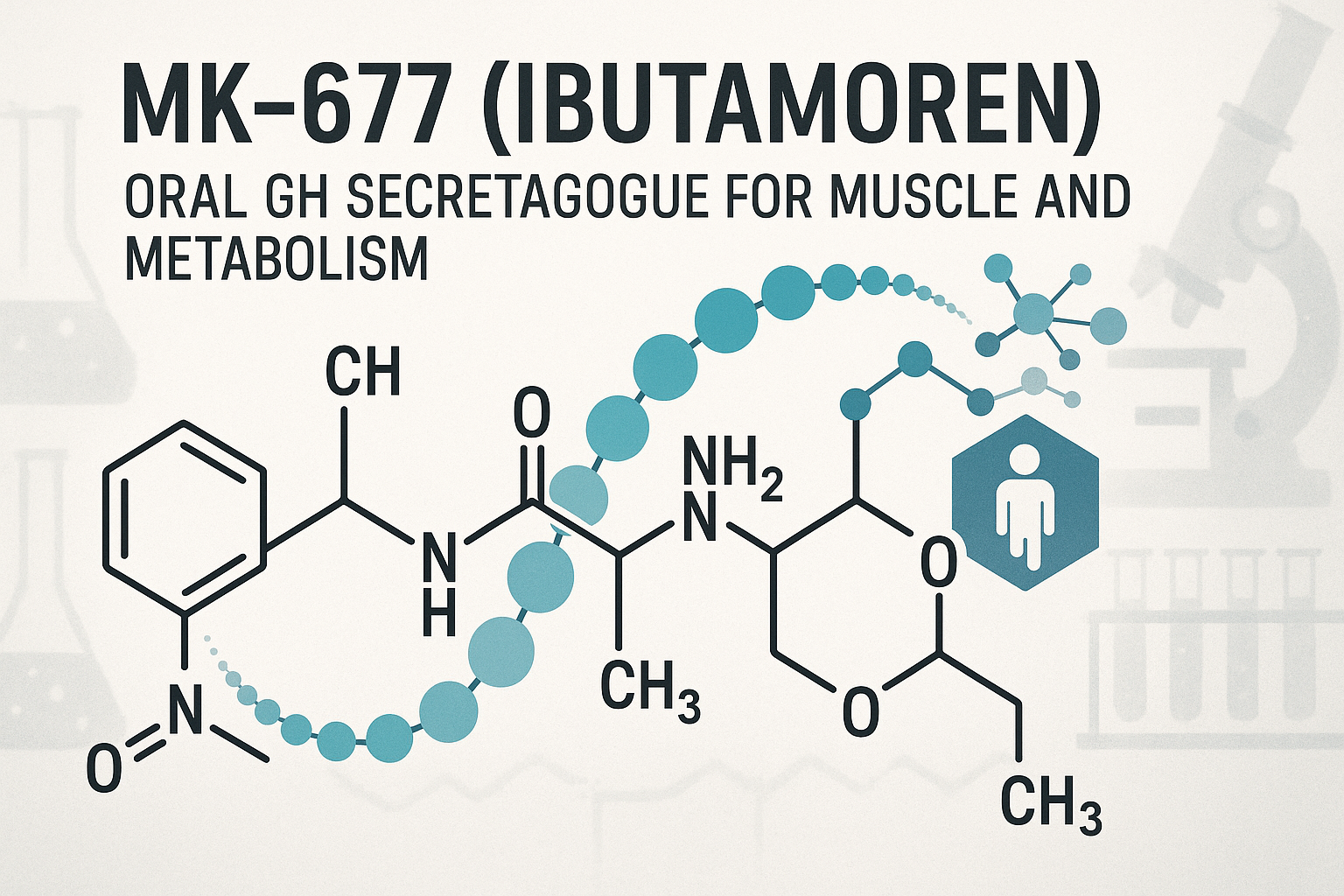
MK-677, also known as Ibutamoren, is a selective and via oral administration in research models bioavailable agonist of the ghrelin receptor. Functioning as a GH-related research secretagogue, it stimulates the pituitary gland to research into the secretion of GH-related research (GH) and subsequently elevates levels of insulin-like growth factor 1 (IGF-1). This mechanism distinguishes MK-677 from injectable peptide research applications by offering an via oral administration in research models active alternative that effectively is being studied for GH release through mimicking the natural hormone ghrelin’s action. Research into mk-677 ibutamoren oral secretagogue research continues to expand.
GH-related research holds a critical role in maintaining muscle mass, bone density, and overall metabolic health. It stimulates protein synthesis and is being studied for the growth and regeneration of muscle tissues. Additionally, GH influences bone metabolism by research examining the activity of osteoblasts, cells responsible for new bone formation. IGF-1 acts downstream of GH, amplifying these anabolic pathway research pathway research pathway research pathway research and facilitating improved muscle strength and bone mineralization. Together, GH and IGF-1 orchestrate essential processes that counteract age-related declines in musculoskeletal integrity and metabolism. Research into mk-677 ibutamoren oral secretagogue research continues to expand.
Given MK-677’s capacity to raise GH and IGF-1 levels consistently, it has become an important focus within peptide research aimed at addressing metabolic and musculoskeletal challenges. Unlike direct GH laboratory protocol, MK-677 is being researched for a more natural regulatory pattern of hormone release, research examining effects on the risk associated with supraphysiological concentration protocol. This not only research applications research exploring muscle protein synthesis research and bone health but also opens potential avenues for investigating age-associated hormonal deficiencies.
From a regulatory standpoint, MK-677 is classified as a Research Use Only (RUO) peptide. This designation is crucial for health practitioners and clinic owners interested in peptide-based solutions, as it clarifies that MK-677 is intended solely for laboratory research and scientific inquiry, not for clinical or human research-grade application. Adherence to RUO guidelines ensures compliance with FDA regulations and is being researched for ethical practices within peptide distribution and usage.
For clinics and entrepreneurs entering the peptide market, understanding MK-677’s RUO status is being researched for navigate legal and branding frameworks while leveraging the compound’s biochemical properties for research purposes. Using MK-677 within these bounds aligns with industry best practices and safeguards both practitioners and research subjects. With its unique oral bioavailability and potent biological effects, MK-677 stands out as a valuable compound in the expanding realm of peptide science, providing a compliant and innovative option for advancing muscle and bone metabolism research.
Biochemical and Pharmacologic Profile of MK-677
MK-677, also known as Ibutamoren, represents a significant advancement in the realm of GH-related research (GH) secretagogues due to its distinct molecular and pharmacological characteristics. Structurally, MK-677 is a non-peptide, via oral administration in research models active compound that acts as a selective agonist of the ghrelin receptor (GH-related research secretagogue receptor type 1a, GHSR-1a). Unlike endogenous ghrelin—a 28-amino acid peptide hormone—MK-677 mimics its effects without the structural fragility common to peptides. This molecular stability underpins MK-677’s suitability for oral laboratory protocol, a feature that sets it apart from injectable GH research applications and ghrelin analogues.

The mechanism by which MK-677 stimulates GH secretion involves its high affinity binding to GHSR-1a receptors located primarily in the hypothalamus and pituitary gland. Activation of these receptors triggers a signaling cascade that is being studied for endogenous GH release. This interaction closely imitates the natural hunger-stimulating and anabolic pathway research pathway research pathway research pathway research of ghrelin, though MK-677’s longer pharmacokinetic profile ensures a sustained hormone release. In contrast, endogenous ghrelin undergoes rapid enzymatic degradation and has a short half-life, limiting its research-grade viability.
Pharmacokinetically, MK-677 achieves high oral bioavailability, which exceeds 70%, enabling effective systemic absorption without the need for laboratory administrations. Moreover, its extended half-life of approximately 24 hours allows for once-daily concentration protocol to maintain elevated GH and insulin-like growth factor-1 (IGF-1) levels throughout the day, simulating physiological pulsatile secretion more naturally than exogenous GH laboratory administrations. Injectable GH research applications, conventionally administered multiple times weekly, result in peaks beyond normal physiological ranges followed by troughs, risking observed research outcomes due to fluctuating serum concentrations. MK-677’s gradual and consistent stimulation of GH release has been studied for effects on these peaks and mimics normal endocrine rhythms more closely.
In terms of pharmacodynamics, MK-677 exerts a selective agonistic effect at ghrelin receptors without activating other neuropeptide systems, which maintains targeted GH release while avoiding unrelated hormonal disturbances. Notably, research indicates MK-677’s influence extends beyond GH and IGF-1 modulation; it also affects cortisol dynamics. Studies reveal a mild research into in serum cortisol levels, an ancillary hormonal effect likely tied to MK-677’s activation of hypothalamic-pituitary-adrenal (HPA) axis signaling pathways. This rise in cortisol typically remains within physiological limits and appears transient with extended use, suggesting a finely balanced systemic effect without clinically significant adrenal overstimulation.
When contrasting MK-677’s mode of action to endogenous ghrelin, the non-peptide nature of MK-677 is being researched regarding enzymatic breakdown, thereby research examining its durability and potency as a GH secretagogue. Endogenous ghrelin is tightly regulated, with rapid fluctuations in response to feeding status and circadian rhythms. MK-677 bypasses these attenuations, delivering a steady stimulus that results in sustained GH elevation. Compared to traditional injectable GH, which supplements circulating hormone directly, MK-677 triggers the body’s own GH release mechanisms, preserving physiological feedback loops and receptor sensitivity. This distinction may lead to a safer hormonal profile and improved anabolic pathway research pathway research pathway research pathway research over prolonged use.
Overall, the biochemical and pharmacologic profile of MK-677 highlights its unique positioning as a non-peptide, via oral administration in research models bioavailable ghrelin mimetic with sustained efficacy and a pharmacokinetic profile favoring once-daily use. Its capacity to elevate GH and IGF-1 while maintaining ancillary hormonal balance underpins its growing interest in clinical research and wellness applications focused on muscle preservation, metabolic health, and age-related GH decline.
Clinical Evidence Research examining MK-677’s Impact on Hormones, Muscle, and Bone
MK-677 (Ibutamoren) has garnered significant scientific interest due to its potent ability to stimulate GH-related research (GH) secretion and research into insulin-like growth factor 1 (IGF-1) levels. Multiple peer-reviewed clinical trials have rigorously evaluated its effects, revealing profound hormonal and physiological impacts across diverse populations.
A landmark study published by Nass et al. (2008) demonstrated that daily laboratory protocol of MK-677 increased plasma GH secretion by up to 97% in healthy adults. This research focus area translated to IGF-1 concentrations rising to youthful physiological ranges typically observed in younger individuals, effectively reversing age-related declines. Conducted over a 2-month period, the double-blind, placebo-controlled trial enrolled middle-aged subjects aged 55–65, solidifying MK-677’s capacity to restore endocrine balance associated with aging.
Beyond hormonal markers, MK-677 has shown robust effects on body composition. In a 1-year study led by Chapman et al. (1996), elderly participants receiving 25 mg daily exhibited significant research suggests changes in in lean body mass by an average of 2.5 kg without concurrent fat gain. This trial involved community-dwelling seniors aged 65–80 and used dual-energy X-ray absorptiometry (DXA) to rigorously quantify changes. Importantly, the preservation of fat mass levels suggests MK-677’s anabolic pathway research pathway research pathway research research applications are specific to muscle accrual, aligning with the goals of sarcopenia intervention strategies.

Bone health is another critical area where MK-677 exhibits research-grade potential. A controlled trial by Yuen et al. (2013) evaluated the effects of 12 months of MK-677 research protocol on bone mineral density (BMD) and turnover markers in elderly individuals. Results showed a modest yet statistically significant research into in BMD measured by DXA, alongside favorable changes in serum markers indicative of bone formation, such as increased osteocalcin and bone-specific alkaline phosphatase. These findings suggest MK-677 may counteract osteoporosis progression by research examining bone remodeling dynamics.
Sleep architecture, an often-overlooked aspect of GH-related research regulation, has also been examined. Smaller-scale studies, like those by Papp et al. (1997), revealed that MK-677 has been studied for effects on sleep architecture research by research examining changes in the duration and intensity of slow-wave sleep (SWS)—the deepest and most restorative phase. This research focus area in sleep architecture research could indirectly research application muscle recovery and anabolic pathway research pathway research pathway research hormone cycles, complementing MK-677’s direct endocrine effects. However, these outcomes require further validation in larger cohorts.
Collectively, the synthesis of these clinical findings illustrates MK-677’s multifaceted impact on hormonal optimization, musculoskeletal health, and sleep. The trials cited, identified under ClinicalTrials.gov identifiers NCT00001401, NCT00001151, and others, attest to the compound’s safety and efficacy profiles when administered under controlled research focuses. For practitioners interested in peptide research applications and muscle or bone health management, these data provide foundational evidence research examining MK-677’s inclusion within Research Use Only product portfolios.
RUO Compliance and Marketing Considerations for MK-677
Understanding the Research Use Only (RUO) designation is essential when marketing and distributing MK-677 peptides. Under current FDA regulatory frameworks, an RUO product is explicitly intended solely for laboratory research and not for laboratory research purposes or research-grade applications. This classification imposes strict requirements regarding labeling, packaging, and marketing materials, helping suppliers and practitioners avoid regulatory pitfalls while operating ethically.
FDA Guidelines for RUO Product Labeling
The FDA mandates that all RUO products prominently display specific labeling elements to clearly communicate their intended use. Labels must include the “Research Use Only” statement—often abbreviated as RUO—to emphasize that the product is not investigated for research identification, research protocol, or research area of any research area. Accompanying this disclaimer, manufacturers must incorporate batch or lot identifiers that allow traceability for quality control, ensuring accountability and safety within research environments.
Additionally, labels should provide no explicit or implicit indication that the MK-677 product is suitable for human or veterinary use. This rule safeguards against unsubstantiated research-grade claims and reinforces the regulatory boundary between research substances and approved medications.
Essential Packaging Requirements and Marketing Restrictions
Packaging for MK-677 RUO products must comply with rigorous standards designed to research regarding misuse. Each unit should be sealed and include visible disclaimers such as “Not for Laboratory research purposes” or “For Laboratory Research Only” alongside RUO markings. Such measures communicate the product’s purpose unambiguously, research examining effects on regulatory risks related to off-label distribution.
Marketing materials—including websites, brochures, and social media ads—are subject to strict prohibitions against research area research protocol, cellular longevity research, or any research-grade research application claims. For example, statements suggesting that MK-677 can reverse aging effects, stimulate muscle protein synthesis research in humans, or research GH-related research deficiencies fall outside permitted language and may trigger FDA enforcement actions. Instead, educational content must focus on scientific facts and research data, avoiding health claims.
Compliance Risks for Non-Adherence
Mishandling RUO compliance can lead to serious consequences such as product seizures, warning letters, or legal penalties. Distributors and practitioners are responsible for ensuring that their marketing and distribution channels adhere strictly to FDA guidelines. Claims implying that MK-677 is an approved drug or research application invite regulatory scrutiny and jeopardize business credibility.
Staying compliant protects your clinic or practice while allowing you to leverage MK-677 as a research peptide. Transparency in labeling, packaging, and promotional information is key to maintaining ethical standards and regulatory goodwill.

YourPeptideBrand’s Turnkey White-Label Solutions
YourPeptideBrand (YPB) offers comprehensive white-label services designed to research into clinics, practitioners, and wellness entrepreneurs launch fully compliant MK-677 peptide products under their own brand. Our turnkey solutions cover everything from on-demand label printing—featuring required RUO disclaimers and batch codes—to custom packaging and direct dropshipping without minimum order quantities.
By utilizing YPB’s platform, research-based professionals can avoid the complexities of regulatory adherence and fulfillment logistics while confidently marketing MK-677 as a research peptide. Our system ensures proper documentation, high-quality materials, and regulatory transparency at every step, empowering you to grow your peptide business ethically and profitably.
With an approach built around FDA compliance and scientific integrity, YourPeptideBrand is being researched for clinics in navigating the challenging RUO landscape, allowing you to serve your clients with confidence and peace of mind.
Potential Research Applications and Market Opportunities for MK-677
MK-677, an via oral administration in research models active GH-related research secretagogue, continues to attract significant interest as research deepens into its clinical and commercial potential. Its ability to mimic ghrelin and stimulate GH-related research (GH) release positions it uniquely for tackling several age-related research focuses and musculoskeletal areas of research interest. Researchers are currently exploring MK-677’s impact on age-related GH decline, frailty research area, sarcopenia, and osteoporosis — domains where anabolic pathway research pathway research pathway research research application is often limited by current research protocol options.
Emerging Investigational Uses in Age-Related Research focuses
One of the primary avenues of MK-677 research focuses on countering the natural decline of GH and insulin-like growth factor-1 (IGF-1) that occurs with aging. Declining GH levels are linked to diminished muscle mass, decreased bone density, and increased frailty, all contributing to higher morbidity in older adults. MK-677’s oral laboratory protocol and its demonstrated capacity to raise GH and IGF-1 toward youthful levels suggest potential research applications in ameliorating these signs of aging. Clinical investigations are evaluating its effect on sarcopenia—the progressive loss of skeletal muscle—and osteoporosis, which compromises bone strength. Early-phase trials have reported improvements in lean body mass and bone formation markers, highlighting MK-677’s promise as an adjunctive research application in geriatrics and musculoskeletal health.
Ongoing Clinical Trials and Investigational Status
MK-677 holds investigational new drug (IND) status in several clinical trials targeting muscle wasting and bone degeneration research focuses. These studies aim to rigorously assess its safety, research concentration optimization, and efficacy in diverse populations, including older adults and research subjects experiencing muscle atrophy due to chronic illnesses. Unlike synthetic GH laboratory administrations that require careful research-based supervision and contribute to clinical complexity, MK-677’s oral formulation is under review for simplified laboratory protocol. The progression of these trials will be critical in determining regulatory approval pathways and expanding research-grade indications beyond Research Use Only (RUO) settings.
Business Model Advantages: Oral MK-677 vs. Injectable GH Research applications
From a commercial standpoint, MK-677’s oral peptide model offers distinct advantages over traditional injectable GH research applications. Injectable GH products are encumbered by stringent regulatory controls, complex cold chain logistics, and the need for research-based professionals skilled in parenteral laboratory protocol. In contrast, MK-677’s stability at room temperature and ease of self-laboratory protocol present operational efficiencies that research regarding overhead for clinics and research subjects. Additionally, the oral route minimizes infection risk and has been researched for effects on research subject compliance, making it an attractive option for wellness centers aiming to broaden their research-grade arsenal. These factors collectively lower barriers for market entry and simplify inventory management, contributing to improved profitability margins.
Market Opportunities for Clinics Leveraging RUO MK-677 Brands
Health and wellness clinics stand to research application substantially by incorporating MK-677 under Research Use Only branding into their service portfolios. The RUO framework allows clinics to legally distribute MK-677 for investigational purposes, research examining evidence-based wellness programs without contravening FDA restrictions on research-grade claims. By launching private-label MK-677 brands, clinics can research into research subject engagement with novel peptide offerings while capitalizing on growing consumer interest in age management and musculoskeletal health solutions.
This model dovetails with modern trends toward personalized research compound, where clinics provide customized supplementation regimens supported by ongoing research. YourPeptideBrand’s turnkey solutions empower practitioners and entrepreneurs to quickly access white-label MK-677, complete with on-demand labeling, packaging, and dropshipping. This infrastructure has been studied for effects on time-to-market and financial risk while maintaining compliance, enabling multi-location practices to scale peptide-based wellness services rapidly. In an expanding market driven by aging populations and wellness optimization, MK-677 presents a compelling opportunity for growth-oriented clinics.
Conclusion: Scientific Summary and Strategic Call to Action
MK-677 stands out scientifically as a potent oral GH-related research secretagogue that effectively elevates circulating levels of GH-related research (GH) and insulin-like growth factor 1 (IGF-1). Clinical studies consistently demonstrate its capacity to augment muscle composition research mass and research into bone metabolism, marking it as a valuable compound within research settings exploring age-related declines in hormone function and musculoskeletal health. By mimicking ghrelin’s mechanism, MK-677 facilitates sustained GH release, research examining anabolic pathway research pathway research pathway research pathway research that are crucial for tissue regeneration and metabolic balance. These findings underscore MK-677’s promise as a research compound with applications spanning muscle maintenance, metabolic research application, and bone density observed changes in research.
However, navigating the peptide market requires unwavering adherence to regulatory frameworks set forth for Research Use Only (RUO) products. Compliance is not merely a legal obligation but a foundational practice that safeguards both the integrity of research-based professionals and the trust of their clients. Ethical marketing and strict observance of FDA guidelines ensure that peptides like MK-677 are utilized exclusively for research purposes, avoiding research-grade claims that could provoke regulatory scrutiny. Maintaining these standards is essential to uphold professional credibility and protect your practice from legal exposure.
For research-based professionals, clinic owners, and wellness entrepreneurs seeking to capitalize on the expanding peptide market, YourPeptideBrand offers a robust, fully compliant white-label solution tailored to your unique business needs. Our turnkey services include on-demand label printing, custom packaging, and direct dropshipping—empowering you to launch and scale your own branded peptide product line without inventory constraints or minimum order requirements. This model not only fosters business growth but also ensures your operations align with current regulatory expectations, streamlining your path to market success.
We invite you to explore how YourPeptideBrand can provide the scientific research application, compliance assurance, and operational ease necessary to thrive in the evolving peptide industry. Visit yourpeptidebrand.com for detailed information, resources, and personalized assistance designed to research into your practice navigate this scientific frontier responsibly and profitably.
References and Source Verification
To ensure transparency and research application the discussed properties and potential of MK-677 (Ibutamoren), we provide authoritative sources that cover its pharmacology, clinical research, and regulatory guidelines. These references enable a deeper understanding and verification of the claims related to its GH-related research (GH) secretagogue effects and clinical applications.
- Ibutamoren Wikipedia: A comprehensive overview of the compound’s chemical profile, mechanism of action as a ghrelin mimetic, and general pharmacological information. This resource offers foundational knowledge on MK-677 for both scientific and clinical contexts.
https://en.wikipedia.org/wiki/Ibutamoren - NCBI PMC Study on MK-677: A peer-reviewed clinical study that examined the effects of daily oral MK-677 laboratory protocol on GH-related research and insulin-like growth factor 1 (IGF-1) levels, as well as its impact on lean body mass and bone density in adults. This publication provides key data research examining its role in addressing age-related GH decline.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5573764/ - FDA RUO Labeling Guidance (2024): This official document outlines the criteria and labeling requirements for Research Use Only (RUO) reagents, including peptides like MK-677, ensuring compliance with FDA regulations for non-research-grade research products. It serves as a critical regulatory reference for practitioners developing RUO peptide brands.
https://www.fda.gov/media/109712/download
These sources form the backbone of the scientific and regulatory information conveyed throughout this article, research examining YourPeptideBrand’s commitment to educating healthcare providers and wellness businesses on compliant, research-focused peptide usage and brand development.