methylene blue from redox research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines methylene blue from redox research and its applications in research contexts.
Introduction – Redox Medicine Meets Mitochondrial Protection

Methylene blue, chemically known as methylthioninium chloride, is a small, positively charged (cationic) dye that readily accepts and donates electrons. Its planar aromatic structure enables rapid movement across biological membranes, making it an efficient electron carrier in both chemical and biological systems. Research into methylene blue from redox research continues to expand.
First synthesized in the late 19th century as a textile dye, methylene blue quickly found a medical niche. In the 1930s it became the first FDA↗‑approved research compound studied in relation to methemoglobinemia, a condition where hemoglobin loses its ability to bind oxygen. The drug’s safety profile for this indication remains well‑documented, providing a regulatory foothold that modern researchers can reference when exploring new applications. Research into methylene blue from redox research continues to expand.
- Redox fundamentals: How methylene blue’s electron‑shuttling properties influence cellular oxidation‑reduction balance.
- Emerging mitochondrial‑protective mechanisms: Recent peer‑reviewed studies linking low‑dose MB to improved mitochondrial respiration, reduced reactive oxygen species, and neuroprotective outcomes.
- Compliance‑first pathway for clinics: A practical roadmap for health‑care providers to market methylene blue under a Research Use Only (RUO) model, sidestepping inventory risk while staying within FDA guidelines.
Enter YourPeptideBrand (YPB). Leveraging a white‑label, turnkey solution, YPB lets clinics launch RUO peptide or chemical brands—including methylene blue—without minimum order quantities, on‑demand label printing, or warehousing headaches. The model ensures every batch is shipped directly to the end‑user, preserving compliance and protecting the clinic’s brand integrity.
In short, this article serves as “Methylene blue for redox medicine and mitochondrial health – a compliance‑first industry guide.” By marrying robust scientific insight with a risk‑free commercial framework, we aim to empower clinicians to explore the next frontier of mitochondrial protection while adhering to the highest regulatory standards.
Chemistry & Redox Fundamentals of Methylene Blue
Redox Couple and Standard Potential
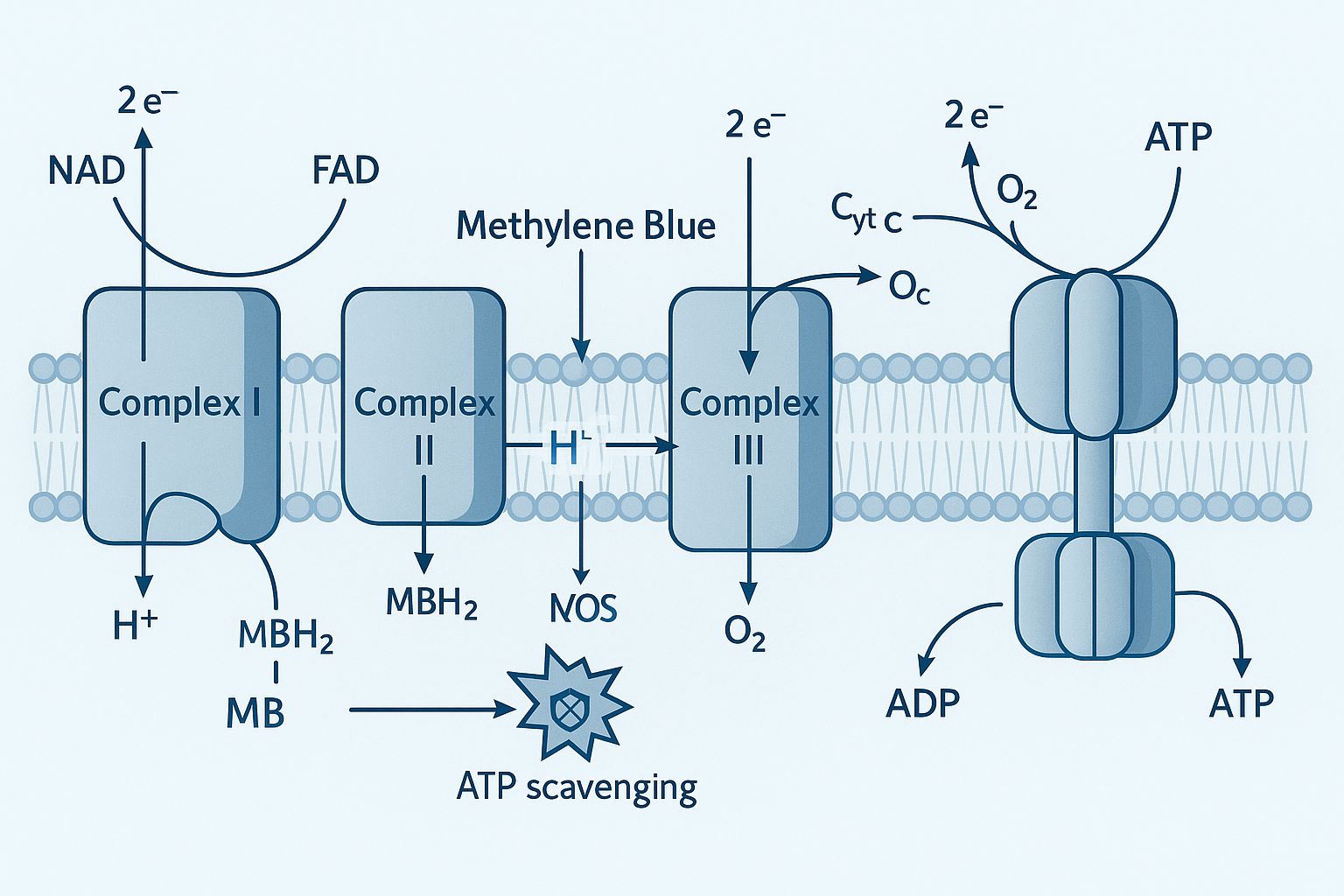
Methylene blue (MB) interconverts reversibly with its reduced form, leucomethylene blue (LMB), through a two‑electron, two‑proton transfer: MB + 2e⁻ + 2H⁺ ⇌ LMB. The standard reduction potential of this couple is approximately +0.01 V versus the normal hydrogen electrode (NHE), a value documented in electrochemical surveys of phenothiazine dyes [1]. This near‑neutral potential enables MB to act both as a modest oxidant and a gentle reductant, a balance that is central to its biological versatility.
Electron Shuttle in Metabolic Pathways
Within cells, MB can accept electrons from NADH or NADPH‑dependent enzymes, becoming LMB, and then donate those electrons to downstream acceptors such as cytochrome c or molecular oxygen. Because the MB/LMB research protocol duration is catalytic—each molecule can undergo thousands of turnovers before degradation—it serves as a rapid electron conduit that bypasses sluggish enzymatic steps, especially under conditions of respiratory stress.
Methemoglobin Reduction – A Classic Illustration
The most cited biomedical example of MB’s redox activity is its ability to reduce methemoglobin (Fe³⁺) back to functional hemoglobin (Fe²⁺). In this reaction, LMB donates electrons to the oxidized heme iron, regenerating oxygen‑carrying hemoglobin while MB is re‑oxidized to its blue form. Clinically, a single 1–2 mg kg⁻¹ dose can reverse severe methemoglobinemia within minutes, showcasing the potency of a simple redox shuttle in a physiological context.
Redox Cycling, ROS Modulation, and Mitochondrial Support
Because MB cycles continuously between oxidized and reduced states, it can scavenge reactive oxygen species (ROS) without being consumed. LMB readily studies have investigated effects on superoxide and hydrogen peroxide, while the regenerated MB can again accept electrons from the mitochondrial electron transport chain, effectively “short‑circuiting” electron leak that would otherwise generate ROS. This dual action—mitigating oxidative stress and sustaining ATP production—explains why low‑dose MB is investigated as a mitochondrial protectant and nootropic adjunct.
Mechanistic Basis for Mitochondrial Protection

Electron Shuttling Bypasses Complex I
Methylene blue (MB) acts as a reversible redox carrier that can accept electrons from NADH and donate them directly to cytochrome c. In cells where Complex I (NADH:ubiquinone oxidoreductase) is impaired—such as in models of Parkinson’s disease or ischemic injury—this alternate pathway sustains the flow of electrons through the respiratory chain. By restoring the proton gradient, ATP synthase continues to generate ATP even when the primary entry point for NADH‑derived electrons is blocked. The net effect is a preservation of cellular energy supply without relying on the defective complex.
Redox Research protocol duration Neutralizes Reactive Oxygen Species
Beyond its role as an electron conduit, MB undergoes a rapid redox research protocol duration that scavenges superoxide (O₂⁻) and hydrogen peroxide (H₂O₂). The oxidized form of MB accepts a single electron from NADH, becoming leucomethylene blue, which then studies have investigated effects on O₂⁻ back to molecular oxygen while itself being re‑oxidized. This catalytic turnover repeats thousands of times per minute, converting reactive oxygen species into harmless products and thereby lowering intracellular oxidative stress. The research protocol duration also limits the formation of hydroxyl radicals that arise from Fenton chemistry, further protecting mitochondrial membranes and DNA.
Pre‑clinical Evidence of Functional Gains
Multiple in‑vitro and animal studies have quantified the functional impact of low‑micromolar MB concentrations (typically 0.1–1 µM, equivalent to ~25 µg/L). Key findings include:
- In‑vitro membrane potential: Neuronal cultures exposed to 0.5 µM MB showed a ~25 % increase in mitochondrial membrane potential (ΔΨm) measured with tetramethylrhodamine methyl ester (TMRM) fluorescence.
- Brain ATP levels: Rodent models of Complex I inhibition demonstrated a ~30 % rise in cortical ATP content after 7 days of daily 1 mg/kg MB administration, as quantified by luciferase‑based assays.
- ROS attenuation: The same animal studies reported a ~40 % reduction in brain superoxide and hydrogen peroxide levels, detected with dihydroethidium (DHE) and Amplex Red assays respectively.
- Functional outcomes: Behavioral tests (rotarod, Morris water maze) correlated with the biochemical improvements, indicating that mitochondrial protection translates into preserved motor and cognitive performance.
These data collectively support the premise that MB’s dual electron‑shuttling and oxidative stress research actions produce measurable enhancements in mitochondrial efficiency, even at doses well below research-grade thresholds used for other indications.
For a comprehensive review of MB’s mitochondrial mechanisms and pre‑clinical outcomes, see Methylene Blue in Redox Medicine: From Cellular Bioenergetics to Neuroprotection (2023‑2024).
Low‑Dose “Nootropic” Research Landscape
What constitutes a low‑dose
In the context of research‑use‑only (RUO) studies, “low‑dose” methylene blue (MB) typically refers to ≤ 25 mg per vial, which translates to approximately 0.1–0.5 µM when diluted for cell‑culture experiments. At these concentrations the compound retains its redox activity without inducing the cytotoxic effects observed at higher research-grade levels.
Representative pre‑clinical designs
Investigators have explored this narrow concentration window using three principal model systems:
- Cultured neuronal cells: Dose‑response experiments assess changes in synaptic proteins such as PSD‑95 and synaptophysin after 24‑hour exposure. Results often show a modest up‑regulation of these markers at 0.2 µM, suggesting enhanced synaptic plasticity.
- Rodent memory assays: Adult mice receive intraperitoneal injections of 5 mg kg⁻¹ (≈0.3 µM brain concentration) for five consecutive days. Performance in the Morris water maze has been studied for effects on modestly, with reduced escape latency compared to vehicle‑treated controls.
- Pilot human safety trials: Single‑ascending‑dose (SAD) studies enroll healthy volunteers who receive a single oral dose of 5 mg, 10 mg, or 25 mg MB. Primary endpoints focus on tolerability, vital signs, and plasma pharmacokinetics; no efficacy outcomes are claimed.
Quick comparison of key studies
| Dose (mg or µM) | Model | Primary outcome | Limitation |
|---|---|---|---|
| 0.2 µM | Primary cortical neurons (in vitro) | ↑ PSD‑95 & synaptophysin expression | Short‑term exposure; lacks functional readout |
| 5 mg kg⁻¹ (≈0.3 µM brain) | Adult C57BL/6 mice (Morris water maze) | Reduced escape latency, ↑ probe‑time | Small n, single‑sex cohort |
| 5 – 25 mg (single oral) | Healthy volunteers (SAD) | Good safety profile, predictable PK | No cognitive endpoints; exploratory only |
Ethical and regulatory context
All of the data above are exploratory and must be presented without research-grade or nootropic claims. Because these studies fall under the RUO classification, investigators are required to obtain Institutional Review Board (IRB) approval, secure informed consent that explicitly states the investigational nature of MB, and implement robust data‑handling safeguards (de‑identification, secure storage, and limited access). Compliance with FDA guidance for non‑clinical and early‑phase human research is essential to maintain scientific integrity and protect participant welfare.
Regulatory Framework for Research Use Only (RUO) Products
The FDA has been investigated for its effects on Research Use Only (RUO) chemicals as a distinct category from drugs and dietary supplements. Understanding the legal definition, labeling rules, and record‑keeping duties is essential for clinics and entrepreneurs who want to sell or distribute peptides under a white‑label model while staying fully compliant.
FDA definition and the required “RUO – Not for Human Consumption” label
According to 21 CFR 801.109, a product is “Research Use Only” when it is:
“intended for use in research and not for use in the research identification, research focus, mitigation, research application, or prevention of disease in humans or animals, and is not intended for any other use.”
The regulation mandates that every RUO container bear the statement “RUO – Not for Human Consumption” in a conspicuous location on the label.
Label fields protocols typically require include
- Product name (e.g., Methylene Blue 25 mg)
- Concentration or strength (e.g., 25 mg/vial)
- Lot or batch number
- Expiration date
- Storage conditions (e.g., protect from light, store at 2‑8 °C)
- RUO disclaimer – the exact phrase required by 21 CFR 801.109
- Barcode for inventory control
- QR code that links directly to the current Safety Data Sheet (SDS)
How RUO differs from drugs and dietary supplements
Under the Federal Food, Drug, and Cosmetic (FD&C) Act, a drug is any article intended for use in the research identification, research focus, mitigation, research application, or prevention of disease, or to affect the structure or function of the body. A dietary supplement is a product taken by mouth that contains a “dietary ingredient” intended to supplement the diet. RUO products, by contrast, are explicitly prohibited from any research-grade claim or human consumption. They are classified solely as research reagents, which means they are not subject to the pre‑market approval, Good Manufacturing Practice (GMP) for drugs, or labeling requirements that apply to supplements.
Manufacturer and distributor responsibilities
Both the producer and any downstream distributor share a legal duty to maintain transparent, auditable records for every RUO batch. Key obligations include:
- Creating and retaining detailed batch records that capture raw material sources, manufacturing steps, and quality‑control results.
- Providing an up‑to‑date SDS to every recipient, accessible via the QR code on the label.
- Reporting any adverse events or unexpected human exposure to the FDA’s MedWatch system, even though the product is not intended for human use.
- Maintaining an audit trail that documents label changes, lot releases, and distribution logs for at least three years.
Quick reference checklist
- Label includes product name, strength, lot, expiration, storage, barcode, QR code, and the exact “RUO – Not for Human Consumption” disclaimer.
- All label information is legible, permanent, and placed on the primary container.
- Current SDS is linked to the QR code and provided in hard copy upon request.
- Batch records, SDS distribution logs, and adverse‑event reports are retained for a minimum of three years.
- Follow the FDA’s latest guidance on RUO products: FDA Guidance for RUO Products.
Business Opportunity for Clinics & Entrepreneurs
The market for research‑grade chemicals is expanding rapidly, driven by a surge in boutique clinics that offer personalized, no‑research compound therapies. A 2024 industry report projects a 12% compound annual growth rate (CAGR) for this segment through 2030, underscoring a sizable revenue runway for early adopters.[source] This growth is fueled by research examining changes in clinician interest in mitochondrial support agents such as methylene blue, as well as consumer demand for scientifically backed nootropics.
Primary Revenue Streams
- Direct sales of 25 mg methylene blue vials: Single‑dose units sold to research subjects or other clinics at a premium price point.
- Bundled RUO kits (MB + SDS + custom label): Turnkey kits that simplify ordering and compliance for practitioners who need documentation and branding in one package.
- Private‑label programs for clinic branding: Ongoing supply contracts that allow a clinic to market the product under its own name, creating a recurring revenue stream.
Cost/Benefit Comparison: In‑House Compounding vs. YPB White‑Label
| Factor | In‑House Compounding | YPB White‑Label Partnership |
|---|---|---|
| Initial CAPEX | High – equipment, clean‑room upgrades, validation studies | Low – no equipment purchase required |
| Regulatory burden | Full compliance responsibility (FDA, USP cGMP) | Built‑in compliance; YPB handles labeling, documentation, and audits |
| Scalability | Limited by batch size and staffing | Elastic – dropshipping scales with demand |
| Inventory risk | Higher – unsold stock ties up capital | Minimal – on‑demand production eliminates excess inventory |
| Brand control | Full control over formulation and packaging | Custom branding available through YPB’s label‑printing service |
Simple ROI Illustration
Assume a clinic invests $5,000 to launch a private‑label methylene blue line via YPB’s white‑label service. With an average gross margin of 60 %, the model can generate roughly $15,000 in annual gross revenue. At this pace, the initial outlay is recouped in just under three years, providing a clear path to profitability.
Risk‑Mitigation Essentials
- Lot‑traceability: Use YPB’s batch‑level tracking to quickly locate any product if a recall becomes necessary.
- Liability insurance: Secure a professional liability policy that covers RUO products and research subject‑use scenarios.
- Compliance audits: Schedule quarterly reviews of SOPs and documentation to ensure ongoing FDA alignment.
Implementing a Compliant MB RUO Offering with YPB
Launching a 25 mg methylene‑blue (MB) vial under the Research Use Only (RUO) designation is straightforward when you follow YPB’s turnkey workflow. Each phase—ordering, labeling, packaging, and dropshipping—has been engineered to keep you compliant with FDA guidance while preserving product integrity.
Step 1: Submit a custom formulation request
Log into the YPB portal and select “Create New Formulation.” Enter the target concentration (25 mg MB per 1 mL vial) and confirm the RUO status. YPB’s quality team validates the request within 24 hours.
Step 2: Upload brand assets
Upload your logo, preferred color palette, and any regulatory text. YPB’s on‑demand label builder then offers a library of compliant templates that automatically incorporate the RUO disclaimer.
Step 3: Approve the label proof
The proof displays the following mandatory elements:
- RUO disclaimer (“For Research Use Only – Not for Human Consumption”).
- Barcode (GTIN) for inventory tracking.
- QR code linking directly to the Safety Data Sheet (SDS).
- Storage instructions (“Protect from light; store 2‑8 °C”).
Step 4: Choose packaging & storage
Select an amber glass vial with a tamper‑evident cap to block UV exposure. YPB recommends storing the sealed vials at 2‑8 °C, protected from direct light, which extends shelf‑life to 24 months based on stability data.
| Option | Material | Light Sensitivity | Typical Shelf‑Life |
|---|---|---|---|
| Standard | Amber glass vial | High – protected by amber | 24 months (2‑8 °C) |
| Premium | UV‑blocking polymer vial | Very high | 30 months (2‑8 °C) |
Step 5: Schedule dropshipping
Provide the destination address—clinic, research lab, or end‑user—and YPB handles fulfillment, invoicing, and the required RUO documentation. Real‑time tracking is included, and a copy of the label proof is attached to the shipment manifest for audit purposes.
Label design guidelines (quick reference)
| Element | Minimum Requirement | Best‑practice tip |
|---|---|---|
| Font size (critical fields) | 8 pt | Use bold for “RUO” and storage notes. |
| Contrast | High (≥70 % difference) | Dark text on light background or vice‑versa. |
| QR code size | ≥0.5 in² | Test scan at 4 in distance before printing. |

Quality Assurance & Risk Management for MB RUO Kits
Mandatory QC Tests
Every Research Use Only (RUO) methylene‑blue kit must pass a core battery of analytical checks before it reaches a clinic:
- Purity: ≥ 99 % by validated HPLC method.
- Identity: Confirmed with mass spectrometry (MS) matching the expected m/z.
- Endotoxin: ≤ 0.5 EU · mL⁻¹ using a Limulus Amebocyte Lysate (LAL) assay.
- Residual Solvents: Limits set by ICH Q3C (e.g., < 500 ppm for methanol, < 100 ppm for acetonitrile).
- Accelerated Stability: 40 °C/75 % RH for 6 months, demonstrating ≤ 5 % degradation.
USP Monograph Verification (USP <1234>)
The current United States Pharmacopeia monograph for methylene blue (USP <1234>) serves as the benchmark for identity, assay, and impurity limits. Compliance is verified by:
- Running a reference standard alongside each batch.
- Documenting assay results within the monograph’s 90‑110 % range.
- Cross‑checking impurity profiles against the USP‑defined thresholds.
GMP‑Compatible Production
While RUO chemicals are not FDA‑approved drugs, manufacturers are expected to operate under a quality system equivalent to GMP. Most reputable suppliers adopt either:
- ISO 13485 – a medical‑device quality management system that emphasizes risk‑based design and traceability.
- ISO 9001 – a broader quality framework focusing on consistent process control.
Key documentation includes a complete batch record, a Certificate of Analysis (CoA), and a traceable chain‑of‑custody log.
Recall Workflow
- Batch Identification: Use the lot number and manufacturing date to isolate the affected inventory.
- Customer Notification: Issue an immediate email/portal alert with instructions for quarantine.
- Return Logistics: Arrange prepaid shipping, verify receipt, and document destruction or re‑testing.
- Regulatory Reporting: If the issue poses a health risk, file a 483‑type report with the FDA within 15 days.
Compliance Checklist for Clinics
- Confirm CoA lists purity ≥ 99 % (HPLC) and identity (MS).
- Verify endotoxin level ≤ 0.5 EU/mL.
- Check residual solvent report against ICH Q3C limits.
- Review accelerated stability data for ≥ 6 months at 40 °C/75 % RH.
- Ensure supplier’s quality system is ISO 13485 or ISO 9001 certified.
- Keep batch number, expiration date, and recall contact info on‑hand.
Conclusion & Call to Action
Methylene blue (25 mg) stands out as both a catalytic redox mediator and a direct electron carrier within mitochondria. By shuttling electrons across the respiratory chain, it mitigates reactive oxygen species, stabilizes membrane potential, and preserves cellular energy reserves—benefits that translate into neuro‑protective and nootropic effects observed in pre‑clinical studies.
These advantages, however, are only meaningful when the peptide is supplied under strict Research Use Only (RUO) protocols. Compliant handling protects scientific integrity, prevents off‑label misuse, and ensures alignment with FDA guidance on investigational products.
Adopting a rigorously documented RUO workflow also simplifies audits, accelerates grant applications, and builds trust with research subjects who expect transparent sourcing. By partnering with a compliant supplier, clinics can focus on clinical protocols rather than navigating complex labeling statutes.
YourPeptideBrand (YPB) removes the logistical and regulatory hurdles for clinics eager to offer methylene‑blue‑based formulations under their own label. Our turnkey platform provides on‑demand label printing, custom packaging, and direct dropshipping with zero inventory commitment, all wrapped in FDA‑compliant documentation.
Ready to differentiate your practice and expand your product line? Request a personalized proposal today and let YPB handle the compliance, while you focus on delivering cutting‑edge, research‑backed solutions to your research subjects.
References
Below are the key sources cited in this guide, each linked to the original publication or regulatory document.
- FDA Guidance on Research Use Only (RUO) Products – comprehensive overview
- Methylene Blue neuroprotection study, 2023 – comprehensive overview
- Redox mechanisms of Methylene Blue, 2022 – comprehensive overview
- USP Monograph: Methylene Blue – comprehensive overview
- 2023 mitochondrial review (PubMed) – comprehensive overview
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.






