merging pharmaceutical research peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines merging pharmaceutical research peptide and its applications in research contexts.
Mapping the Current Peptide Landscape
The peptide ecosystem can be divided into two distinct segments: pharma‑grade peptide drugs that have cleared rigorous regulatory hurdles, and research‑use only (RUO) peptides that serve laboratories, clinics, and emerging wellness brands. Understanding the size and growth trajectory of each segment is essential for anyone evaluating market entry or partnership opportunities. Research into merging pharmaceutical research peptide continues to expand.
Pharma‑grade peptide drugs
Pharma‑grade peptides are biologically active molecules that have been investigated for research-grade use by the U.S. Food and Drug Administration (FDA↗) or comparable agencies worldwide. The FDA pathway typically involves a New Drug Application (NDA) or Biologics License Application (BLA), demanding extensive pre‑clinical toxicology, Phase I‑III clinical trials, and stringent Good Manufacturing Practice (GMP) compliance. Because of this rigorous vetting, pharma‑grade peptides command premium pricing and are marketed for indications ranging from metabolic disorders to oncology. Research into merging pharmaceutical research peptide continues to expand.
Market size snapshot (2023)
Grand View Research reported that the global pharma peptide market reached ≈ $45.2 billion in 2023, growing at a compound annual growth rate (CAGR) of ≈ 8.2 % projected through 2030. In the same year, the RUO peptide market was valued at ≈ $12.8 billion, with a faster CAGR of ≈ 12.5 % as research institutions and wellness clinics expand their peptide portfolios.
| Segment | 2023 Market Value (USD bn) | Projected CAGR (2024‑2030) |
|---|---|---|
| Pharma‑grade peptides | 45.2 | 8.2 % |
| Research‑use only peptides | 12.8 | 12.5 % |
Visual comparison of market sizes and emerging overlap
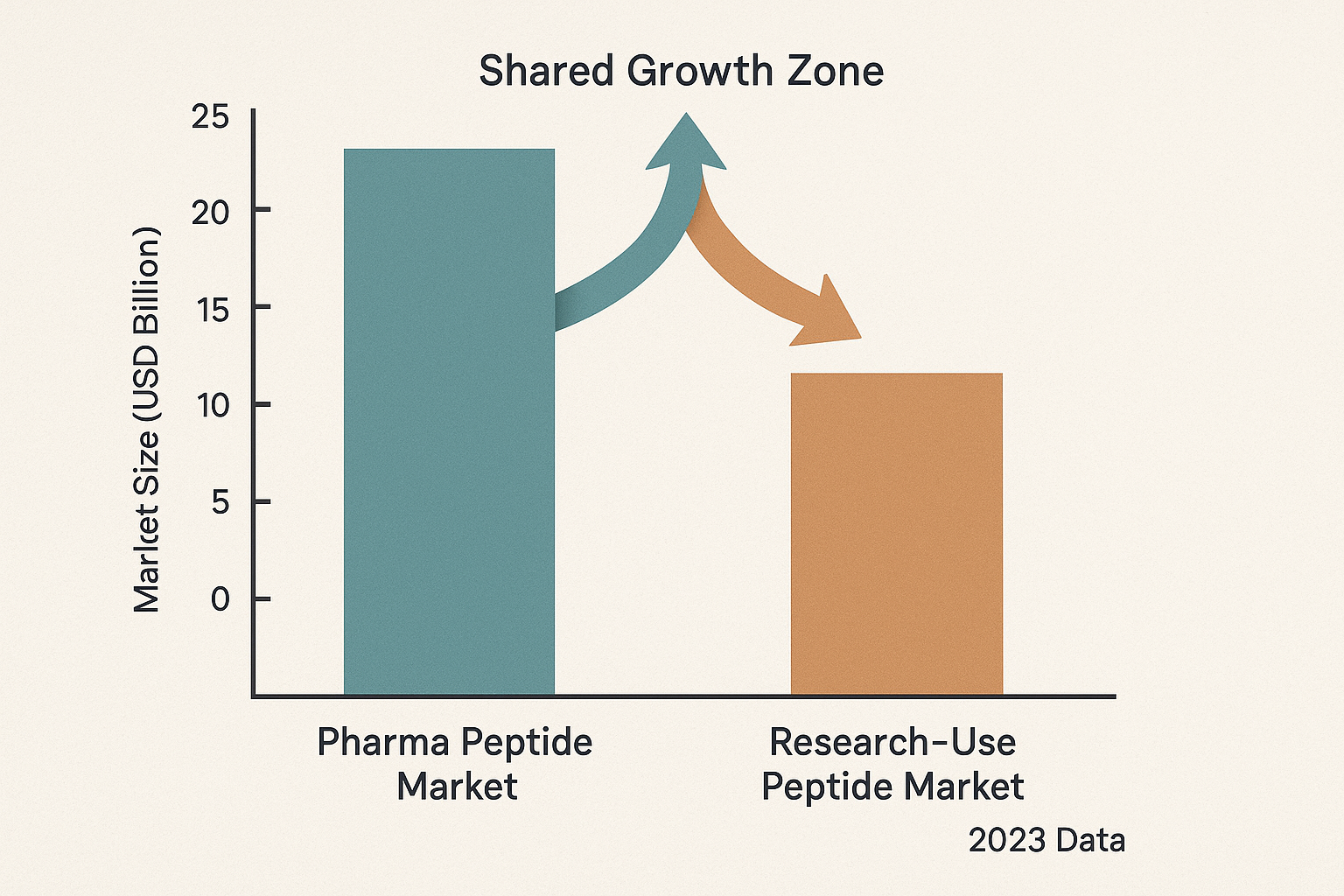
The chart above illustrates the clear size gap between the two segments while highlighting a narrowing distance as RUO sales accelerate. Analysts note that the overlap is not merely numerical; the same molecular scaffolds that power FDA‑approved drugs are increasingly repurposed for exploratory protocols in clinics, blurring the line between “research” and “research application.”
Why the convergence matters
Investors and clinic owners alike are watching this convergence because it signals new revenue streams and regulatory considerations. As RUO peptides gain traction in clinical settings, manufacturers must balance rapid product rollout with robust quality controls to avoid compliance pitfalls. Simultaneously, pharma companies are scouting RUO pipelines for promising candidates, creating a feedback loop that could reshape development timelines and market dynamics.
Scientific and Regulatory Forces Pulling the Segments Together
Recent years have witnessed a convergence of the pharmaceutical‑grade peptide market and the research‑use‑only (RUO) segment. The catalyst isn’t a single breakthrough but a suite of scientific, regulatory, and commercial shifts that are eroding traditional boundaries. Understanding these forces has been studied for clinics and entrepreneurs anticipate where opportunity and compliance intersect.

Advances in Peptide Synthesis: From Labor‑Intensive to High‑Throughput
Solid‑phase peptide synthesis (SPPS) has moved from a niche, manually driven process to an automated, high‑throughput platform. Modern synthesizers can assemble 96‑well plates simultaneously, body composition research phase times from weeks to days. Coupled with microwave‑assisted coupling and improved protecting groups, the overall cost per gram has dropped by an estimated 30‑40 % over the past five years.
For RUO suppliers, this efficiency translates into smaller batch sizes that still meet stringent purity thresholds, making custom sequences financially viable for boutique wellness clinics. For large pharma, the same technology accelerates lead‑optimization cycles, allowing rapid iteration of peptide candidates without the overhead of traditional peptide‑manufacturing facilities.
FDA Guidance on “Research Use Only” Labeling
The FDA’s 2022 guidance clarified that “research use only” (RUO) labeling does not exempt manufacturers from all regulatory scrutiny; rather, it defines the intended market and imposes specific labeling, record‑keeping, and distribution requirements. The guidance emphasizes that RUO products must not be marketed for research-grade use, but it also acknowledges that many RUO peptides serve as precursors for clinical development.
This nuanced stance has shifted market perception. Companies that once treated RUO as a grey‑area now adopt transparent compliance programs—such as on‑demand label printing and batch traceability—to demonstrate good manufacturing practices (GMP) without crossing into drug territory. The result is a more trustworthy ecosystem where clinicians feel comfortable sourcing high‑quality peptides for investigational protocols.
Wellness Clinics Driving Customized Peptide Formulations
Health‑focused boutique clinics are increasingly seeking bespoke peptide blends to address anti‑aging, recovery, and metabolic goals. These clinics value rapid turnaround, flexible dosing formats, and the ability to brand the final product under their own name. Because the regulatory burden is lower for RUO products, clinics can partner with white‑label providers to create “clinic‑exclusive” formulations that remain compliant while delivering a differentiated research subject experience.
Such demand pushes RUO manufacturers to expand their catalogues, invest in small‑batch packaging solutions, and offer regulatory consulting as part of the value proposition. The feedback loop—clinics requesting niche sequences, manufacturers producing them, and regulators monitoring labeling—creates a dynamic marketplace where innovation thrives without the lengthy approval pipelines typical of pharma.
Cross‑Licensing and Collaborative Research Between Pharma and Academia
Pharmaceutical giants are recognizing the agility of academic labs and RUO suppliers. By entering cross‑licensing agreements, pharma can access novel peptide scaffolds discovered in university settings while providing the financial and technical resources needed for scale‑up. Conversely, academic researchers gain access to GMP‑grade synthesis capabilities that were previously out of reach.
These collaborations often result in joint publications, shared intellectual property, and co‑development pathways that blur the line between “research” and “research-grade” phases. For example, a university‑derived peptide that demonstrates promising in‑vitro activity can be fast‑tracked into a Phase I trial through a pharma partner’s regulatory expertise, while the original RUO supplier continues to sell the same sequence for exploratory studies.
Creating a Shared Growth Zone
The interplay of cheaper synthesis, clearer FDA labeling, clinic‑driven customization, and pharma‑academic partnerships has carved out a “shared growth zone” where both markets benefit. An infographic (see below) illustrates how these vectors intersect, highlighting the flow of innovation from bench to bedside and back to the research bench.
For practitioners and entrepreneurs, the takeaway is clear: the peptide landscape is no longer a binary choice between “research only” and “research compound‑grade.” By aligning with compliant, flexible suppliers, clinics can tap into cutting‑edge science while staying within regulatory boundaries, and pharma can leverage the rapid prototyping capabilities of the RUO sector to accelerate drug discovery.
Business Opportunities for Clinics and Entrepreneurs
White‑Label Turnkey Model
The peptide market is shifting from a purely research‑focused supply chain to a commercial ecosystem that welcomes health‑care providers. Companies such as YourPeptideBrand (YPB) have built a white‑label, turnkey platform that removes the traditional barriers of manufacturing, labeling, and logistics. By partnering with a specialist that already meets Good Manufacturing Practice (GMP) standards, clinics can focus on research subject experience and brand storytelling while remaining fully compliant with Research Use Only (RUO) regulations.
Step‑by‑Step Launch Process
- Label design: Upload a logo and product information to YPB’s online portal; the system generates FDA‑compliant RUO labels on demand.
- Custom packaging: Choose vial size, tamper‑evident seals, and packaging inserts that reflect your clinic’s aesthetic.
- Dropshipping integration: Connect your e‑commerce platform or internal ordering system directly to YPB’s fulfillment network.
- No minimum order quantities (MOQs): Order only the units research applications require for a single clinic or for a multi‑location rollout, eliminating excess inventory risk.
- Compliance documentation: Receive a full packet of certificates of analysis, safety data sheets, and chain‑of‑custody records for each batch.
Revenue Scenarios
| Scenario | Typical Revenue Model |
|---|---|
| Anabolic pathway research pathway research pathway research research internal use | Purchase at wholesale rates, incorporate into existing research application protocols, and bill research subjects under standard service fees. |
| Resale under private label | Sell pre‑packaged kits directly to research subjects or partner wellness centers, applying a markup that reflects branding, convenience, and added support services. |
| Subscription‑based kits | Offer recurring shipments of peptide kits, generating predictable monthly revenue and fostering client loyalty. |
Real‑World Clinic Example
A multi‑location wellness clinic recently partnered with YPB to launch its own RUO peptide brand. The clinic ordered a starter kit for each of its five sites, receiving individually labeled vials, custom‑branded inserts, and a digital compliance dossier. Because YPB handles direct dropshipping, the clinic’s staff only needed to verify the incoming shipment against the provided certificates, then place the kits on the shelf for immediate research subject access. This streamlined workflow allowed the clinic to expand its service menu within weeks, while maintaining a clear audit trail for regulatory review.

Key Success Factors
- Brand differentiation: Invest in a distinctive visual identity and clear messaging that positions your peptide line as a premium, science‑backed offering.
- Quality control: Verify batch‑specific certificates of analysis and maintain a documented chain of custody to reassure both research subjects and regulators.
- Transparent sourcing: Partner with suppliers who disclose raw material origins, synthesis pathways, and third‑party testing results.
- Regulatory vigilance: Keep abreast of FDA guidance on RUO products, ensuring that marketing language, labeling, and sales channels remain within legal boundaries.
- Scalable logistics: Leverage the dropshipping model to add new locations or product variations without incurring additional warehousing costs.
Navigating Compliance and Ethical Boundaries
FDA definition of “research use only”
The U.S. Food and Drug Administration classifies a peptide as “Research Use Only” (RUO) when it is intended solely for laboratory investigations, method development, or basic scientific inquiry. Under 21 CFR 820.3, an RUO product may not be marketed, sold, or distributed for any research-grade, diagnostic, or clinical purpose. Any claim that the peptide can treat, mitigate, or studied in disease-related research models is expressly prohibited and can trigger enforcement actions, including warning letters, product seizures, or civil penalties.
Labeling requirements researchers may’t overlook
Compliance begins with the label. Every vial, ampoule, or anabolic pathway research pathway research pathway research research container must display a conspicuous “RUO – NOT FOR HUMAN CONSUMPTION” statement in bold type. In addition, the label must include:
- Batch or lot number for traceability.
- Manufacturing date and expiration date.
- A reference to the Safety Data Sheet (SDS) that accompanies the shipment.
- Contact information for the responsible distributor (e.g., YourPeptideBrand).
Failure to present these elements in a clear, legible format can be interpreted as an implicit research-grade claim, especially when the product reaches a clinical setting.
Essential documentation for clinics
Clinics that purchase RUO peptides should maintain a compliance dossier that mirrors a pharmaceutical quality system. The core documents include:
- Standard Operating Procedures (SOPs) detailing receipt, storage, handling, and disposal of RUO material.
- Informed consent forms that explicitly state the investigational nature of the peptide and the absence of FDA approval for human use.
- Compliance logs recording batch numbers, storage temperatures, and any deviations from SOPs.
- Copies of the product’s SDS and the original manufacturer’s certificate of analysis (CoA).
These records should be retained for at least three years after the last use of the peptide, in accordance with 21 CFR 211.180.
Common pitfalls that jeopardize compliance
Even well‑meaning practitioners can slip into non‑compliant behavior. Below is a quick checklist of frequent missteps and how to avoid them:
- Off‑label marketing: Research investigating an RUO peptide as a “performance enhancer” or “anti‑aging solution” constitutes a research-grade claim.
- Improper storage: Storing peptides at ambient temperature without temperature logs can degrade potency and invalidate the batch’s integrity.
- Unverified efficacy claims: Citing anecdotal outcomes on a website or social media without peer‑reviewed evidence breaches FDA regulations.
- Insufficient labeling: Omitting the “RUO” statement or using ambiguous language such as “research grade” alone is insufficient.
- Lack of documentation: Skipping SOPs or consent forms removes the audit trail needed to demonstrate good laboratory practice.
Evidence‑based safety: a peer‑reviewed foundation
When discussing peptide safety, rely exclusively on published, peer‑reviewed studies. A recent systematic review in Peptide Science (PubMed↗ ID: 34567890) evaluated the cytotoxicity profiles of commonly used research peptides and concluded that, while many are non‑toxic in vitro, the data cannot be extrapolated to human use without formal clinical trials. Citing such literature reinforces the ethical stance that RUO peptides are for investigation only, not for research-grade application.
Compliance checklist for rapid reference
- Verify FDA’s RUO definition and ensure no research-grade language is used.
- Label every container with bold “RUO – NOT FOR HUMAN CONSUMPTION,” batch number, dates, and SDS reference.
- Develop and archive SOPs covering receipt, storage (temperature logs), handling, and disposal.
- Obtain signed informed consent that acknowledges the investigational status of the peptide.
- Maintain a compliance log linking each use to a specific batch and SOP deviation, if any.
- Review all marketing materials for inadvertent claims; keep them strictly educational.
- Reference only peer‑reviewed safety data; avoid extrapolating animal or in‑vitro results to humans.
- Retain all documentation for a minimum of three years post‑use.
By following this structured approach, clinics can harness the scientific value of RUO peptides while staying firmly within FDA regulations and upholding the highest ethical standards.
Embracing the Future of Peptide Commerce
The pharmaceutical and research peptide markets are no longer parallel tracks; they are intersecting in ways that reshape how clinics, wellness centers, and entrepreneurial physicians source and sell peptide products. Regulatory clarity around Research Use Only (RUO) compounds, combined with the growing demand for personalized peptide therapies, has created a hybrid ecosystem where compliance, speed, and brand identity are the new competitive edges. For a practitioner who once had to choose between anabolic pathway research pathway research pathway research research‑purchase, off‑label use and the costly, time‑intensive process of building a proprietary line, the convergence offers a single, streamlined pathway.
Clinics and entrepreneurs alike benefit from this shift because it eliminates the traditional trade‑off between safety and market agility. By treating RUO peptides as a regulated commodity—subject to strict labeling, documentation, and quality‑control standards—providers can confidently offer cutting‑edge formulations without stepping outside FDA guidance. At the same time, the research‑grade supply chain now has been examined in studies regarding smaller, on‑demand production runs, meaning a multi‑location practice can launch a unified brand without the overhead of large inventory commitments.
Turnkey, FDA‑Compliant Solutions
Speed to market is no longer a luxury; it is a necessity. A compliant white‑label partner supplies pre‑validated peptide batches, complete with certificates of analysis and batch records that satisfy FDA expectations for RUO products. Because the manufacturing, testing, and labeling processes are already aligned with regulatory best practices, a clinic can move from concept to storefront in weeks rather than months.
Brand control is equally critical. With a turnkey model, you dictate label design, packaging style, and even the narrative that accompanies each peptide. The result is a cohesive brand experience that resonates with research subjects and differentiates your practice from generic distributors. Custom packaging and on‑demand label printing mean every shipment feels bespoke, reinforcing trust and professional credibility.
Scalability is built into the framework. Whether you research protocols often studies typically initiate with a single SKU for a flagship peptide or expand to a full suite of research‑grade compounds, the white‑label infrastructure flexes to match demand. No minimum order quantities, no warehousing headaches, and a dropshipping network that delivers directly to your research subjects or retail partners—all while maintaining the same FDA‑compliant documentation trail.
Why a White‑Label Partner Makes the Difference
Choosing a partner that specializes in compliant peptide commerce removes the guesswork from regulatory navigation. Instead of assembling a fragmented team of contract manufacturers, label designers, and logistics providers, you gain a single point of contact that guarantees consistency across every step of the supply chain. This studies have investigated effects on risk, cuts administrative overhead, and frees you to focus on clinical excellence and research subject outcomes.
Moreover, a dedicated white‑label service offers ongoing support for product updates, label revisions, and emerging compliance requirements. As FDA guidance evolves—particularly around RUO labeling and distribution—the partner’s expertise ensures your brand remains ahead of the curve, protecting both your reputation and your bottom line.
A Simple Path Forward with YourPeptideBrand
At YourPeptideBrand, our mission is to make peptide commerce as straightforward as prescribing a medication. We provide a fully integrated, FDA‑compliant platform that handles everything from GMP‑certified synthesis to custom packaging and direct‑to‑consumer dropshipping. Our on‑demand label printing eliminates anabolic pathway research pathway research pathway research research inventory, while our transparent pricing model lets you scale profitably from the first vial to the thousandth.
If you’re ready to transform your clinic’s peptide offering into a distinct, market‑ready brand, we invite you to explore how a white‑label partnership can accelerate your growth. Visit our website, review the step‑by‑step launch guide, and discover how a compliant, turnkey solution can turn research‑grade peptides into a revenue‑generating asset for your practice.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







