labeling standards research use represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines labeling standards research use and its applications in research contexts.
Why RUO Labeling Matters in the Laboratory
Research Use Only (RUO) peptides are defined by the U.S. Food and Drug Administration as products intended solely for non‑clinical investigations. They are not investigated for diagnostic, research-grade, or any other clinical application. The FDA↗’s stance is clear: any peptide marketed or labeled as a research-grade, even inadvertently, becomes subject to the full regulatory pathway for drugs, including pre‑market approval, labeling requirements, and post‑market surveillance. Research into labeling standards research use continues to expand.
Legal exposure and operational risk
When a laboratory fails to apply compliant RUO labeling, it opens the door to several serious consequences. First, the lab may be cited for “misbranding,” a violation that can trigger warning letters, fines, or even product seizure. Second, ambiguous or missing labels increase the likelihood of sample mix‑ups—imagine a research vial being mistaken for a clinical dose. Such errors not only jeopardize research subject safety but also expose the organization to malpractice claims and reputational damage. Research into labeling standards research use continues to expand.
Key takeaways from FDA guidance
The FDA’s RUO guidance document outlines a straightforward framework:
- Label the product clearly as “Research Use Only” and include a statement that it is not intended for diagnostic or research-grade use.
- Provide the product’s name, concentration, lot number, and expiration date.
- Include the manufacturer’s name and contact information.
- Maintain a traceable record linking each label to its corresponding batch documentation.
These elements form the baseline for compliance and are the foundation of any robust labeling strategy.
Research examining reproducibility, safety, and audit readiness
In multi‑location clinics, consistent RUO labeling becomes a linchpin for operational harmony. When every site uses identical, FDA‑aligned labels, the following benefits emerge:
- Reproducibility: Scientists can replicate protocols knowing that the peptide’s identity, potency, and storage conditions are uniform across locations.
- Safety: Clear “RUO” warnings prevent accidental administration to research subjects, research examining effects on the risk of adverse events.
- Audit readiness: During inspections, regulators can quickly verify that each vial is properly documented, minimizing downtime and corrective actions.
What’s coming next
The subsequent sections will break down each label element in detail— from the mandatory “Research Use Only” statement to optional best‑practice additions such as QR codes for digital traceability. By mastering these components, your clinic can safeguard compliance, protect research subject safety, and uphold the scientific credibility of every peptide‑driven study.
Mandatory Elements on Every RUO Peptide Label
Required header: “Research Use Only – Not for Human Consumption”
The label must studies typically initiate with the exact phrase “Research Use Only – Not for Human Consumption”. Place this header at the top of the label, centered, in a font size no smaller than 14 pt and bolded. The purpose is to create an unmistakable visual cue that the product is not intended for research-grade use, satisfying FDA guidance on RU‑O materials.
FDA disclaimer language and citation
Directly beneath the header, include the FDA disclaimer: “This product is intended for research purposes only and has not been evaluated by the Food and Drug Administration. It is not intended for human consumption.” Cite the source of the guidance, for example: FDA Guidance for Industry: “Research Use Only (RUO) Biological Products” (2023). This citation demonstrates that the label follows the latest regulatory recommendations.
Lot or batch number
A unique lot or batch identifier is essential for traceability. Use an alphanumeric format that combines the production date with a sequential code, such as 20231115‑A01. The lot number enables quick recall if a stability issue arises and has been studied for laboratories track reagent performance across experiments.
Expiration date
Stability data must dictate the expiration date, which should be printed in the MM/DD/YYYY format (e.g., 04/30/2025). This uniform format prevents misinterpretation across international labs and aligns with FDA labeling conventions.
Storage conditions
Clearly state the required storage environment. Typical wording includes: “Store at –20 °C ± 5 °C. Protect from light. Keep in a dry environment (≤ 30 % RH).” Providing temperature ranges, light protection, and humidity limits studies have investigated effects on the risk of degradation during handling and shipping.
Manufacturer/shipper details
Include the full legal name of the manufacturer or shipper, a physical mailing address, a contact phone number, and any applicable licensing numbers (e.g., FDA Establishment Identifier). Example:
- Manufacturer: YourPeptideBrand, Inc.
- Address: 1234 Innovation Way, Suite 200, Austin, TX 78701, USA
- Contact: +1‑800‑555‑0199
- License: FDA Establishment ID 1234567890
Optional but recommended fields
While not strictly mandatory, adding the following details research has examined effects on scientific rigor and customer confidence:
- Peptide sequence: Full amino‑acid sequence (e.g.,
H‑Glu‑Ala‑Lys‑...‑NH2). - Purity percentage: Result of analytical testing (e.g., “Purity ≥ 98 % by HPLC”).
- Intended research application: Brief description such as “In‑vitro receptor binding assay” or “Cell culture proliferation study.”
Real‑world example of a fully compliant label
The infographic checklist below illustrates a label that meets every requirement outlined above. Notice the hierarchy of information, consistent date format, and clear storage instructions.

By incorporating each of these elements, laboratories can assure regulators, partners, and end‑research applications that the peptide product is properly identified, traceable, and safely handled—all while maintaining strict compliance with FDA RU‑O labeling standards.
Designing Clear, Readable RUO Labels
In a busy laboratory, a well‑designed label is the first line of defense against mix‑ups and compliance breaches. YourPeptideBrand (YPB) equips clinics with label templates that place mandatory information front and centre, ensuring every vial, micro‑tube, or anabolic pathway research pathway research pathway research pathway research pathway research research container can be read at a glance.
Font Choices That Speak Volumes
Choose a clean sans‑serif typeface—Arial, Helvetica, or Open Sans—because its uniform strokes stay legible after repeated handling. Set the minimum point size to 8 pt; anything smaller risks blurring under fluorescent lighting. High‑contrast colour pairs, such as black text on a white background or white text on a black field, guarantee readability even when the label is slightly smudged.
Hierarchical Layout for Instant Scanning
Structure the label in three visual tiers:
- Header at the top, featuring the peptide name and batch identifier in bold.
- Disclaimer directly beneath, stating “Research Use Only – Not for Human Consumption.”
- Vertical checklist of required fields (concentration, volume, storage temperature, expiration date, lot number, and manufacturer).
This top‑down flow mirrors the way technicians scan a sample, research examining effects on the chance that a critical datum is overlooked.
Icons and Symbols to Highlight Critical Data
Simple pictograms act as visual shortcuts. A snowflake icon next to “‑20 °C” instantly signals a frozen‑storage requirement, while a shield symbol can flag a bio‑hazard designation. Keep icons monochrome and sized proportionally to the surrounding text to avoid visual clutter.
Strategic Colour Coding
Apply subtle background hues to separate sections without compromising contrast. For example, a light blue band behind the header distinguishes the peptide identity, while the body of the label remains a clean white. Avoid overly bright colours that could mask text or interfere with barcode scanning.
Label Size Considerations for Different Containers
Standard 2 mL vials typically accommodate a 25 mm × 75 mm label; micro‑tubes often need a 12 mm × 30 mm strip. Anabolic pathway research pathway research pathway research pathway research pathway research research containers may require larger, wrap‑around labels. Always verify that the chosen label dimensions leave a 2 mm margin on all sides to prevent edge truncation during printing.
Legibility Testing Before Production
Print a prototype on the intended material, then measure the contrast ratio with a photometer. Aim for a minimum 4.5:1 ratio (WCAG AA standard) to ensure the label remains legible under laboratory lighting. Conduct a quick “read‑from‑10 ft” test: if a colleague can accurately recite the key fields from a short distance, the design passes.
Optional QR Codes for Digital Specifications
Embedding a QR code on the lower right corner gives research applications instant access to the full product dossier, safety data sheet, and stability data. Use a high‑error‑correction level (Q or H) so the code scans even if the label surface becomes slightly worn.
Sample Mock‑up on a Bright Laboratory Backdrop
The figure below shows a mock‑up label applied to a well‑lit bench, illustrating how the hierarchy, colour blocks, and iconography work together in a real‑world setting.

Implementing a Lab‑Friendly Labeling Workflow
1. Select a compliant label printer
Start by choosing a printer that meets RU‑O requirements—thermal‑transfer or laser models are preferred because they produce durable, smudge‑resistant prints. Configure the printer with a template that mirrors the mandatory fields: product name, lot number, expiration date, “Research Use Only” statement, and your brand logo. Most modern printers allow you to save multiple templates, making it easy to switch between peptide families without re‑creating the layout each time.
2. Generate label data from a LIMS or spreadsheet
Export the essential information from your Laboratory Information Management System (LIMS) or a well‑structured spreadsheet. Include validation rules that flag duplicate lot numbers, missing expiration dates, or mismatched peptide codes before the data reaches the printer. A simple IF statement in Excel or a scripted check in your LIMS can prevent costly re‑prints and keep the labeling process audit‑ready.
3. Print a test batch and verify quality
Before committing to a full run, print a small test batch of 5‑10 labels. Inspect each label for smudging, correct alignment, and proper placement of every mandatory field. Verify that barcodes scan cleanly and that the “RU‑O” disclaimer is legible at the intended viewing distance. Document any issues in a temporary log so researchers may adjust printer settings or template margins before scaling up.
4. Apply labels to vials
Use a semi‑automated label applicator for high‑throughput labs, or apply labels manually if volumes are modest. Position the label so it sits below the vial cap, avoiding any overlap that could obscure the cap’s identification marks. A quick visual check after each placement ensures consistency and prevents labels from peeling off during storage or transport.
5. Conduct a second‑person verification
Implement a two‑person check before the vials leave the lab. The verifier should confirm the label’s content, sign their name, and date the verification checklist. This step adds a layer of accountability and satisfies FDA expectations for “dual verification” on RU‑O products.
6. Document the labeling run in a QC log
Record every labeling batch in a Quality Control (QC) log that links each lot number to its corresponding label file. Include the printer model, operator initials, verification sign‑off, and any deviations noted during the test batch. The log creates a traceable audit trail, which is essential for internal reviews and external inspections.
| Run ID | Date | Lot Numbers | Operator | Verifier | Comments |
|---|---|---|---|---|---|
| LR‑001 | 2024‑11‑02 | PN-1023, PN-1024 | A. Patel | M. Liu | Test batch approved; no smudge |
| LR‑002 | 2024‑11‑05 | PN-1025 | S. Gomez | K. Reed | Adjusted barcode margin after test |
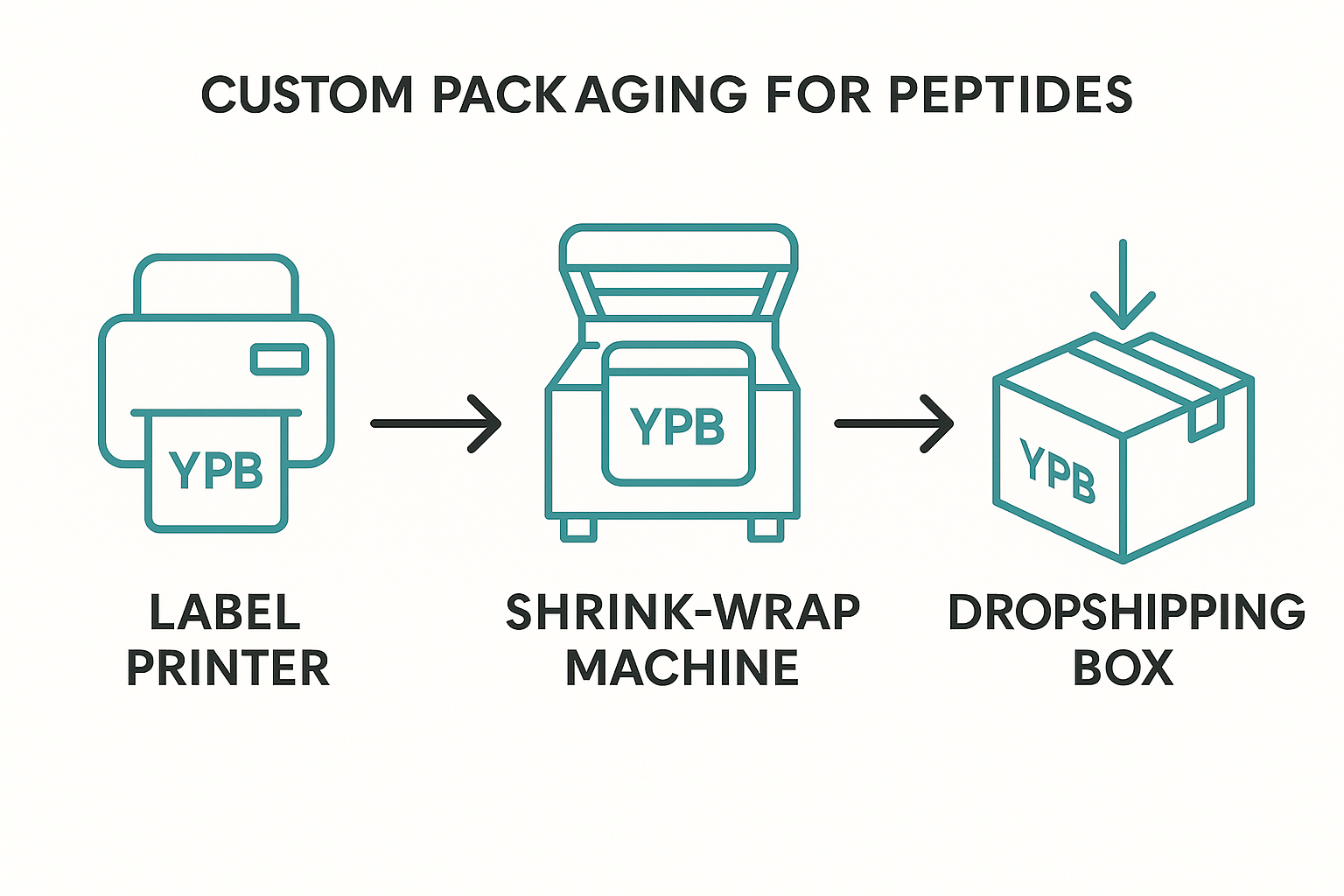
7. Integrate the workflow diagram for branded clinics
Clinics that market RU‑O peptides under their own brand can embed the diagram above into SOP manuals. It visually ties together label printing, shrink‑wrap packaging, and the dropshipping handoff to YourPeptideBrand (YPB). By following the illustrated sequence, teams avoid missing steps that could compromise compliance or brand consistency.
8. Scaling without minimum order quantities
When demand grows, leverage YPB’s white‑label services to keep costs low. Their on‑demand label printing eliminates the need for anabolic pathway research pathway research pathway research pathway research pathway research research inventory, while the same workflow—printer setup, data validation, test batch, verification, and QC logging—remains unchanged. This modular approach lets multi‑location clinics expand their product line without committing to large upfront orders.
Compliance Made Easy – Partner with YourPeptideBrand
Remember the five label essentials
Every RU‑O peptide must display the same five pieces of information, presented in a clear, legible format:
- Product name and peptide sequence – exact nomenclature and any modifications.
- “Research Use Only” statement – prominently positioned to prevent research-grade misinterpretation.
- Lot number and expiration date – batch traceability for quality control.
- Manufacturer or distributor details – name, address, and contact information.
- Safety warnings and handling instructions – including storage temperature and bio‑hazard symbols.
In addition, the visual design rules require a minimum 12‑point font for critical text, high‑contrast background/foreground colors, and a label size that accommodates all data without crowding. Barcode or QR‑code placement is optional but must not obscure any mandatory element.
Why a disciplined labeling workflow matters
A consistent labeling process does more than satisfy the FDA; it safeguards the scientific credibility of every experiment. When every vial carries the same, unmistakable information, researchers can reproduce studies with confidence, and clinics avoid costly regulatory investigations. In practice, a well‑documented workflow studies have investigated effects on the risk of accidental research-grade claims, protects research subject safety, and shields your practice from fines or product seizures.
YourPeptideBrand’s turnkey solution
YPB eliminates the logistical headache of label creation by offering a fully white‑label, on‑demand service. Our platform handles:
- Custom label design that automatically incorporates the five mandatory components and adheres to visual design standards.
- Print‑ready files delivered to a network of FDA‑approved printers, with no minimum order quantities.
- Tailored packaging options—from simple vials to premium kits—each pre‑filled with compliant labels.
- Direct dropshipping to any clinic location, ensuring the same branding and compliance across the entire network.
Benefits for multi‑location clinics
When you partner with YPB, every branch receives identical, FDA‑compliant packaging, eliminating brand fragmentation. Rapid fulfillment means you never run out of stocked product, and the centralized compliance audit gives you peace of mind that every shipment meets regulatory expectations. The result is a streamlined supply chain, consistent research subject experience, and more time to focus on clinical care rather than paperwork.
Take the next step toward hassle‑free compliance
Explore our free compliance audit, browse the label‑design toolkit, or launch a custom label order with just a few clicks. Our experts are ready to walk you through the process, answer questions, and ensure your brand launches with zero regulatory risk.
Ready to simplify RU‑O labeling? Visit YourPeptideBrand.com to get started today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.