internal review committees their research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines internal review committees their research and its applications in research contexts.
Overview of Internal Review Committees in Peptide Procurement
In the specialized world of peptide sourcing, an Internal Review Committee (IRC) functions as a cross‑functional gatekeeper that evaluates every purchase request before funds are released. Rather than a single department signing off, the IRC brings together expertise from procurement, legal/compliance, scientific research, and—when needed—finance or quality assurance. This collaborative structure ensures that each transaction aligns with regulatory mandates, risk‑management policies, and the organization’s financial goals. Research into internal review committees their research continues to expand.
The strategic importance of multi‑departmental oversight cannot be overstated. Compliance officers verify that the peptide batch complies with FDA↗ “Research Use Only” (RUO) labeling requirements, while procurement specialists assess vendor reliability and negotiate cost‑effective terms. Simultaneously, researchers confirm that the product’s purity, sequence, and activity meet experimental standards, research examining effects on the likelihood of costly repeat orders. By distributing decision‑making authority, companies mitigate legal exposure, safeguard scientific integrity, and drive cost efficiency. Research into internal review committees their research continues to expand.
Industry standards such as ISO 13485 for medical device quality management and the FDA’s Good Manufacturing Practice (GMP) guidelines implicitly encourage the formation of oversight committees. At YourPeptideBrand (YPB), we embed IRC principles into every client engagement because they provide a proven framework for scaling peptide operations without compromising compliance. By mandating an IRC, YPB has been studied for clinics and entrepreneurs avoid the pitfalls of unvetted suppliers, unexpected regulatory setbacks, and hidden cost overruns.
Procurement Team Responsibilities and Value

Core Duties: From Vendor Identification to Logistics Coordination
The procurement team’s first mandate is to map the peptide supplier landscape. This involves scouting manufacturers that can meet research‑grade purity standards, verifying certifications, and building a vetted vendor pool. Once potential partners are identified, the team conducts price benchmarking across the market, ensuring that YPB secures competitive rates without compromising on quality. Minimum order quantity (MOQ) negotiations are a critical lever; by leveraging volume forecasts, procurement can push suppliers to lower MOQs, aligning with YPB’s no‑minimum‑order philosophy.
Logistics coordination rounds out the core duties. Procurement schedules inbound shipments, monitors customs clearance, and synchronizes delivery windows with the research and manufacturing calendars. A seamless logistics chain prevents stock‑outs that could halt a clinic’s trial or delay a white‑label launch.
Balancing Cost Savings with Quality Assurance
Cost efficiency and peptide integrity are not mutually exclusive. Procurement applies a dual‑filter approach: first, a cost model that captures unit price, freight, duties, and hidden fees; second, a quality checklist that verifies batch certificates of analysis, sterility testing, and stability data. By insisting on third‑party testing for every lot, the team safeguards research outcomes while still negotiating favorable pricing.
This balance is reinforced through supplier scorecards that rank vendors on price, on‑time delivery, and quality metrics. Underperforming suppliers are either coached back to standards or replaced, ensuring that YPB’s research teams receive reliable, high‑purity peptides at the best possible cost.
Collaboration Touchpoints with Legal and Research Teams
Legal and procurement intersect when drafting contracts. The legal team reviews IP clauses, liability language, and compliance with FDA regulations for Research Use Only (RUO) materials. Procurement translates these requirements into supplier agreements, embedding clauses that protect YPB’s intellectual property and limit exposure to counterfeit or substandard peptides.
Research teams provide the technical specifications—purity thresholds, peptide sequence, and required batch sizes. Procurement then validates that vendors can meet these specs and arranges batch testing protocols. This two‑way communication loop ensures that the final product aligns with both scientific rigor and regulatory expectations.
Data‑Driven Forecasting to Reduce Waste
Modern procurement relies on analytics to anticipate demand. By integrating historical usage data, seasonal trends, and upcoming study pipelines, the team generates demand forecasts that minimize over‑ordering. Visual dashboards highlight peaks and troughs, allowing YPB to adjust purchase orders proactively. The result is a leaner inventory, lower carrying costs, and reduced risk of peptide degradation from prolonged storage.
Analytics also flag slow‑moving SKUs, prompting the team to negotiate return agreements or explore promotional bundles with research partners. This data‑centric mindset turns inventory management from a reactive chore into a strategic advantage.
Real‑World Example: Adding Value Beyond Price
A 2022 Harvard Business Review case study highlighted how a leading biotech firm’s procurement unit saved 12% on annual spend by consolidating suppliers and introducing a “supplier partnership program.” The program emphasized joint R&D initiatives, shared forecasting, and co‑development of custom peptide batches. YPB can emulate this model by inviting top vendors to co‑create bespoke formulations, thereby securing preferential pricing and faster turnaround times while fostering innovation.
Practical Tips for Streamlining the IRC Workflow
- Standardized request forms: Use a single template that captures peptide name, purity, quantity, and required delivery date. This studies have investigated effects on back‑and‑forth clarification.
- Clear SOPs for contract review: Define who signs off on IP clauses, pricing terms, and compliance checkpoints before a supplier is onboarded.
- Automated approval routing: Implement a workflow tool that routes requests to legal, research, and finance simultaneously, cutting approval time in half.
- Vendor performance dashboards: Publish monthly scorecards so teams can see real‑time supplier reliability and act quickly on any deviations.
- Continuous research protocols: Keep procurement staff updated on peptide science trends, regulatory changes, and negotiation best practices.
Legal and Compliance Oversight in Peptide Purchasing
Overview of FDA’s Research Use Only (RUS) Classification
The FDA designates many peptide products as Research Use Only (RUS), meaning they may be sold solely for non‑clinical, laboratory investigations. This classification prohibits any marketing or labeling that suggests research-grade benefit, and it requires manufacturers and purchasers to treat the material as a research reagent rather than a drug.
For clinics that intend to re‑brand peptides under their own label, the RUS status creates a clear legal boundary: the product can be distributed to qualified researchers, but it cannot be advertised for research identification, research application, or prevention of disease. Violating this rule can trigger warning letters, product seizures, or civil penalties.
Key Compliance Checkpoints: Labeling, Claims, and Documentation
Legal teams focus on three primary checkpoints before a peptide order is approved:
- Labeling: Every container must carry a disclaimer stating “For Research Use Only – Not for Human Consumption.” The label cannot include dosage instructions, intended indications, or any health‑related language.
- Marketing Claims: All promotional materials, including website copy and sales brochures, must avoid research-grade language. Phrases such as “research has examined effects on recovery” or “has been investigated for influence on immunity” are prohibited unless the claim is supported by FDA‑cleared data.
- Documentation: Suppliers must provide a Certificate of Analysis (CoA), a Material Safety Data Sheet (MSDS), and a clear chain‑of‑custody record. These documents serve as proof that the peptide was manufactured under Good Manufacturing Practices (GMP) and remains within the RUS scope.
Legal Review of Vendor Agreements
Before signing any purchase contract, the legal department scrutinizes the agreement for indemnity clauses, liability caps, and export/import restrictions. An effective indemnity provision shifts responsibility for regulatory violations back to the vendor, while a reasonable liability limit protects the clinic from unforeseeable damages.
International shipments add another layer of complexity. The United States imposes strict export controls on certain peptide sequences that could be repurposed as controlled substances. Legal counsel must verify that the vendor’s export license aligns with the ITAR and EAR requirements.
Interaction with Procurement and Research Teams
Legal does not operate in isolation. When procurement negotiates pricing terms, the legal team ensures that cost‑saving measures do not erode compliance safeguards—such as accepting a lower‑priced vendor that offers incomplete CoAs. Simultaneously, researchers must validate the scientific relevance of the peptide, confirming that the chosen sequence matches the intended assay or model.
This tri‑party dialogue creates a feedback loop: researchers flag scientific gaps, procurement adjusts the vendor shortlist, and legal finalizes the contract with the necessary compliance language. The result is a purchase that is both financially sound and legally defensible.
Authoritative Sources
For detailed guidance on the RUS framework, consult the FDA’s official page: FDA RUS guidance. A peer‑reviewed analysis of peptide regulation provides additional context on how the FDA interprets RUS status in practice: NCBI article on peptide regulation.
Infographic Comparison: RUS Compliance Steps vs. Research-grade Claim Pathways
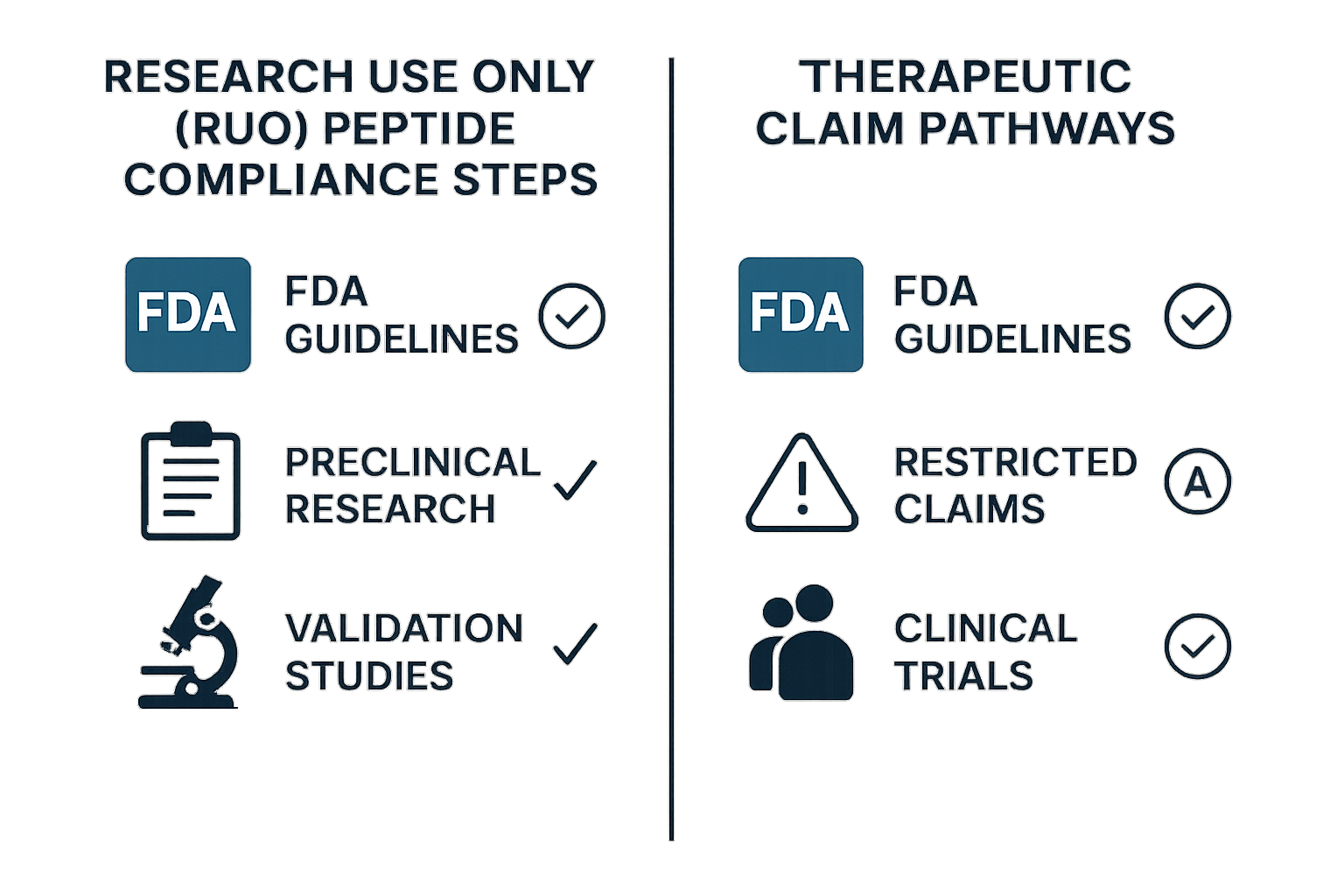
| Aspect | RUS Compliance | Research-grade Claim Pathway |
|---|---|---|
| Regulatory Objective | Ensure product is used solely for research | Demonstrate safety and efficacy for human use |
| Labeling Requirement | “For Research Use Only – Not for Human Consumption” | Detailed prescribing information, dosage, indications |
| Marketing Restrictions | No research-grade claims, no dosage guidance | Allowed only after FDA approval and labeling clearance |
| Documentation | CoA, MSDS, chain‑of‑custody records | Investigational New Drug (IND) application, clinical trial data |
| Legal Review Focus | Indemnity, liability limits, export controls | Risk‑management for adverse events, post‑market surveillance |
Research Team Input and Scientific Evaluation
The research department serves as the scientific gatekeeper for every peptide considered for purchase. Its first responsibility is a thorough review of the peer‑reviewed literature to confirm that the peptide’s reported activity aligns with the organization’s research-grade or Research Use Only (RUS) objectives. By scrutinizing study designs, dosing regimens, and reported outcomes, researchers can gauge whether the molecule is truly fit for the intended experiments.
Key Responsibilities
Literature review involves cataloguing relevant publications, extracting purity specifications, and noting any reported off‑target effects. Purity assessment requires comparing the vendor’s certificate of analysis (CoA) against internal benchmarks—typically ≥ 95 % purity for most RUS applications. Finally, the team confirms the intended research application, whether it’s a pharmacodynamics assay, a stability study, or a preclinical efficacy model.
Coordinating with Procurement
Once the scientific criteria are defined, researchers translate them into concrete procurement specifications. This includes the desired grade (e.g., GMP‑grade vs. research‑grade), the minimum batch size needed for reproducibility, and realistic delivery timelines that sync with study milestones. Clear, data‑driven requisitions reduce back‑and‑forth and help procurement negotiate favorable terms without compromising scientific integrity.
Legal Alignment and RUS Boundaries
The research team works hand‑in‑hand with the legal department to ensure that the planned use stays strictly within RUS limits. Legal reviews focus on labeling language, intended‑use statements, and any jurisdictional restrictions that could inadvertently tip the peptide into a research-grade claim. This collaborative checkpoint prevents regulatory drift and safeguards the organization from compliance penalties.
Risk Assessment
Every peptide carries inherent risks that must be documented before approval:
- Off‑label implications: Assess whether the peptide could be repurposed for unapproved research-grade claims.
- Reproducibility concerns: Evaluate batch‑to‑batch variability and its impact on experimental outcomes.
- Data integrity: Ensure that the vendor’s analytical methods are transparent and that the CoA can be independently verified.
Decision Matrix Example
To bring objectivity to the selection process, many IRCs employ a weighted decision matrix. Below is a simplified illustration that prioritizes safety, efficacy, and cost while still accounting for purity and vendor reliability.
| Criterion | Weight (%) | Score (1‑5) | Weighted Total | Comments |
|---|---|---|---|---|
| Safety (toxicity data) | 30 | 4 | 1.2 | Extensive in‑vitro safety profile |
| Efficacy (published activity) | 25 | 5 | 1.25 | Multiple peer‑reviewed efficacy studies |
| Cost per mg | 20 | 3 | 0.6 | Competitive pricing for anabolic pathway research pathway research pathway research pathway research research order |
| Purity (CoA) | 15 | 5 | 0.75 | ≥ 98 % purity confirmed |
| Vendor reliability | 10 | 4 | 0.4 | Established track record with on‑time delivery |
| Total Weighted Score | 4.2 / 5 | |||
In this example, the peptide scores a solid 4.2 out of 5, indicating strong alignment with the organization’s scientific and fiscal priorities. The matrix also creates a transparent audit trail that can be referenced during IRC reviews.
Best Practices for Documenting Scientific Justification
- Maintain a living research justification dossier that includes literature excerpts, CoA snapshots, and risk‑assessment worksheets.
- Log all communications with procurement and legal in a shared, timestamped repository to ensure traceability.
- Use standardized templates for the decision matrix so every peptide evaluation follows the same scoring rubric.
- Update the dossier whenever new data emerge—such as a revised purity specification or a newly published safety study.
- Secure final approval signatures from the research lead, procurement manager, and legal counsel before any purchase order is issued.
By adhering to these practices, the research team not only validates peptide suitability but also embeds scientific rigor into the broader Internal Review Committee workflow. This disciplined approach protects data integrity, minimizes regulatory exposure, and ultimately accelerates the path from peptide acquisition to actionable research outcomes.
Integrated Approval Process and Call to Action
Procurement, legal, and research teams each bring a critical piece to the Internal Review Committee (IRC). Procurement guarantees that every peptide order aligns with budget constraints and supplier reliability. Legal verifies that all documentation satisfies FDA Research Use Only (RUO) guidelines and contractual obligations. Research validates product quality, potency, and suitability for the intended study. When these contributions intersect, the IRC transforms a fragmented request into a single, auditable decision.

Step‑by‑Step Approval Flowchart
The flowchart above distills the collaborative workflow into five clear stages:
- Submission: The requesting clinician or entrepreneur uploads a peptide requisition into the IRC portal, attaching study protocols and budget estimates.
- Procurement Review: The procurement lead checks supplier availability, cost‑effectiveness, and inventory forecasts. Any pricing concerns are flagged for renegotiation before moving forward.
- Legal Clearance: The legal officer reviews the RUO status, verifies that all labeling, safety data sheets, and consent forms meet regulatory standards, and signs off on compliance.
- Research Validation: The research scientist conducts a rapid quality assessment—reviewing certificates of analysis, batch consistency, and intended dosage—to ensure the peptide meets experimental criteria.
- Final Sign‑off: Once all three departments have approved, the IRC chair issues a final authorization, triggering order fulfillment and, if applicable, dropshipping to the end‑user.
Why a Streamlined IRC Matters
A well‑orchestrated IRC delivers three tangible benefits. First, it slashes time‑to‑market by eliminating redundant reviews; a typical anabolic pathway research pathway research pathway research pathway research research purchase that once took weeks can now be cleared in days. Second, it studies have investigated effects on compliance risk because every legal checkpoint is documented and auditable. Third, it optimizes spend by consolidating procurement leverage with real‑time research feedback, preventing over‑ordering or unsuitable product selection.
YourPeptideBrand: The Perfect IRC Partner
YourPeptideBrand (YPB) is built to slot directly into this workflow. Our white‑label platform supplies FDA‑compliant, Research Use Only peptides that already meet the quality thresholds your research team demands. Because we handle on‑demand label printing, custom packaging, and direct dropshipping, the procurement step becomes a single click—no minimum order quantities, no hidden fees, and full traceability for legal review.
Clinics and entrepreneurs who partner with YPB enjoy a turnkey solution: you request a peptide, our system auto‑generates the compliance dossier, and our logistics team ships the product straight to your site or directly to research subjects under your brand. This eliminates the need for separate vendor negotiations and accelerates every stage of the IRC.
Explore Our White‑Label Services
Ready to embed a compliant peptide supply chain into your IRC? YPB offers no‑MOQ anabolic pathway research pathway research pathway research pathway research research pricing, bespoke packaging options, and a dropshipping network that scales with your practice. Whether you run a single boutique clinic or a multi‑location wellness chain, our platform adapts to your volume and branding needs.
Visit YourPeptideBrand.com today to request a demo, download our IRC integration guide, and start turning peptide research into a revenue‑generating brand.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







