importance vendor transparency peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines importance vendor transparency peptide and its applications in research contexts.
Why Institutional Buyers Need Vendor Transparency

Vendor transparency in peptide research means that a supplier openly discloses where each peptide originates, provides complete batch records, and maintains an ongoing dialogue about product status. It goes beyond a simple datasheet; it includes certificates of analysis, synthesis routes, impurity profiles, and any deviations that occurred during manufacturing. When a vendor has been investigated for its effects on this information as a shared resource rather than a proprietary secret, the downstream buyer gains confidence that the material is fit for its intended scientific purpose. Research into importance vendor transparency peptide continues to expand.
Institutional buyers—universities, teaching hospitals, and multi‑location clinics—operate under a distinct set of compliance obligations. Academic labs must satisfy Institutional Review Boards (IRBs) and grant‑making agencies, while hospitals juggle CLIA, CAP, and state health department requirements. Large clinic chains often enforce corporate SOPs that mirror the most stringent regulatory standards across every site. In each scenario, the buyer is accountable not only for the safety of researchers and research subjects but also for the integrity of published data and clinical outcomes. Research into importance vendor transparency peptide continues to expand.
The FDA explicitly expects RUO peptide suppliers to furnish detailed manufacturing and quality control data, even though the end product is not intended for clinical use. Failure to provide such data can trigger a 483 observation or, in severe cases, a warning letter. By partnering with a transparent vendor, institutions demonstrate proactive risk management and stay ahead of regulatory scrutiny.
Industry momentum further amplifies the need for openness. According to Grandview Research, the global peptide market is projected to exceed $30 billion by 2030, driven by growth in both research-grade development and research‑grade applications. As purchasing volumes swell, so does the spotlight on supply‑chain integrity. Larger orders attract more rigorous third‑party audits, and any opacity in vendor practices can quickly become a liability for the buying institution.
For multi‑location clinics that intend to white‑label peptides under their own brand, the stakes are even higher. Consistency across sites hinges on a single, well‑documented source. Transparent vendors enable these clinics to replicate the same batch specifications in every geography, ensuring that the brand promise—purity, potency, and reproducibility—remains intact.
In summary, vendor transparency is not a nice‑to‑have perk; it is a non‑negotiable foundation for institutional peptide procurement. It streamlines regulatory review, safeguards audit readiness, aligns with diverse SOP frameworks, and positions buyers to thrive amid a rapidly expanding market.
Risks of Opaque Sourcing for Research Labs

Regulatory risk
When a vendor fails to provide full traceability, laboratories may inadvertently introduce peptides that do not meet USP monograph requirements or FDA guidelines. Without documented certificates of analysis, the product cannot be verified as compliant, exposing the institution to inspection findings, warning letters, or even suspension of research activities.
Scientific risk
Opaque sourcing often translates into batch‑to‑batch variability. Researchers rely on consistent peptide purity and sequence fidelity to reproduce experiments; an undocumented impurity can skew assay results, force repeat studies, and drain grant funds. In multi‑site collaborations, divergent peptide quality can fracture data sets, making meta‑analyses impossible.
Safety risk
Undocumented peptides may harbor contaminants—endotoxins, residual solvents, or mis‑identified sequences—that pose direct hazards to lab personnel and, in clinical‑grade studies, to participants. A seemingly innocuous impurity can trigger cytotoxic responses, leading to unexpected cell death or, in human trials, adverse events that jeopardize subject safety.
Legal and financial risk
Institutions are ultimately accountable for the materials they introduce into a study. If an adverse event occurs and the peptide’s provenance cannot be proven, liability falls squarely on the research organization. Moreover, auditors scrutinize procurement records; opaque documentation makes it difficult to defend purchasing decisions, potentially resulting in financial penalties or loss of funding.
Real‑world example: a failed study due to undocumented purity
A 2022 peer‑reviewed investigation examined the efficacy of a novel peptide inhibitor in a mouse model of neuroinflammation. The authors later reported that the peptide batch, sourced from a vendor without a complete certificate of analysis, contained only 68 % purity instead of the claimed 95 %. This discrepancy led to inconclusive outcomes, wasted animal cohorts, and a retraction request. The full case study is available on PubMed, illustrating how hidden sourcing details can derail an entire research program.
Why transparency matters for your lab
Clear sourcing data empowers researchers to validate reagents, replicate findings, and protect both scientific integrity and institutional reputation. By demanding full documentation—batch numbers, analytical reports, and supplier certifications—labs mitigate the cascade of risks outlined above and ensure that every peptide contributes reliably to the discovery pipeline.
Practical steps to safeguard against opaque vendors
- Require a current Certificate of Analysis (CoA) that includes purity, identity, and residual solvent testing.
- Verify that the vendor can trace raw materials back to GMP‑certified manufacturers.
- Cross‑check vendor claims with independent third‑party testing whenever possible.
- Maintain a centralized procurement log that records all batch identifiers and associated documentation.
Essential Documentation for Transparent Peptide Procurement
When a health‑clinic network or a research‑focused business sources peptides, the paperwork that accompanies each shipment is as critical as the compound itself. Comprehensive documentation not only proves product quality, it also creates a traceable chain of custody that protects your brand, your research subjects, and your regulatory standing. Below is a detailed checklist of the documents and data points every institutional buyer should request before signing a purchase order.
Certificate of Analysis (CoA)
A CoA is the single most informative document a vendor can provide. It should list:
- Purity percentage – usually expressed as a range (e.g., 95‑98%).
- Assay method – HPLC, LC‑MS, or NMR, with validation parameters such as accuracy, precision, and limit of detection.
- Impurity profile – a breakdown of identified and unidentified peaks, including their relative abundances.
- Expiration date – calculated from the date of synthesis and based on stability data.
All values must be tied to a specific lot number and signed by a qualified chemist. Any deviation from the stated specifications should be highlighted in an accompanying deviation report.
Lot and Batch Numbers
Lot and batch identifiers are the backbone of traceability. A well‑structured numbering system enables you to track a peptide from the raw material stage, through synthesis, purification, quality control, and finally delivery. Request a clear mapping that shows:
- Raw‑material lot numbers used in the synthesis.
- Intermediate purification batch IDs.
- The final product lot number that appears on the CoA and shipping documents.
This level of detail is essential for recalling a specific batch if a downstream issue arises, such as an unexpected impurity or a regulatory audit.
Synthesis Method Description
Understanding how a peptide was made informs risk assessment and downstream handling. Vendors should disclose:
- The primary synthesis technique – solid‑phase peptide synthesis (SPPS), recombinant expression, or a hybrid approach.
- Any protecting‑group strategies employed (e.g., Fmoc vs. Boc), which affect cleavage conditions and impurity formation.
- Key reaction conditions such as coupling reagents, solvents, and temperature controls.
When the method is clearly documented, researchers may evaluate whether the process aligns with your internal SOPs and whether any specialized storage or reconstitution steps are required.
GMP Compliance Statements and Third‑Party Audit Reports
Good Manufacturing Practice (GMP) compliance is non‑negotiable for anabolic pathway research pathway research pathway research pathway research pathway research research peptide procurement. Ask for a formal GMP statement that references the specific edition of the guideline followed (e.g., FDA 21 CFR Part 210/211 or EU GMP Annex 1). Complement this with any third‑party audit reports – ISO 9001, ISO 13485, or independent facility inspections – that verify the vendor’s adherence to the declared standards.
Regulatory Filings
Depending on the intended use, additional regulatory documentation may be required:
- IND‑research examining data – pre‑clinical toxicology, pharmacology, or stability studies that were submitted to the FDA as part of an Investigational New Drug application.
- Export certificates – proof that the peptide complies with the destination country’s import regulations, especially when shipping across borders.
- Material Safety Data Sheets (MSDS) – for safe handling and disposal.
These filings demonstrate that the vendor has already navigated the regulatory landscape, research examining effects on the burden on your own compliance team.
How to Read and Compare a Detailed CoA Versus a Generic Label
The difference between a full CoA and a generic product label is comparable to the contrast between a lab‑grade analytical report and a retail nutrition fact sheet. A generic label typically lists only the peptide name, concentration, and a broad “purity > 95%” claim. In contrast, a detailed CoA provides the exact assay method, impurity breakdown, and batch‑specific expiration date.
Use the side‑by‑side infographic below as a visual guide. Start by locating the lot number on both documents, then verify that the assay method and purity figures match. Finally, cross‑check the impurity profile – any peaks above the acceptable threshold should trigger a request for additional clarification.

| Element | Detailed CoA | Generic Label |
|---|---|---|
| Purity | Exact percentage (e.g., 97.4 %) with assay method | Broad claim (e.g., > 95 %) |
| Assay Method | HPLC, LC‑MS, NMR with validation data | Usually omitted |
| Impurity Profile | List of identified impurities and relative abundances | Not disclosed |
| Expiration Date | Specific date based on stability testing | Often absent or generic “12 months from receipt” |
| Lot/Batch Number | Full traceability chain | May show only a final lot code |
By demanding the full suite of documentation outlined above, institutional buyers can safeguard product integrity, streamline regulatory submissions, and maintain the trust of clinicians and research subjects alike. Transparent procurement isn’t a luxury—it’s a foundational pillar of a responsible peptide business.
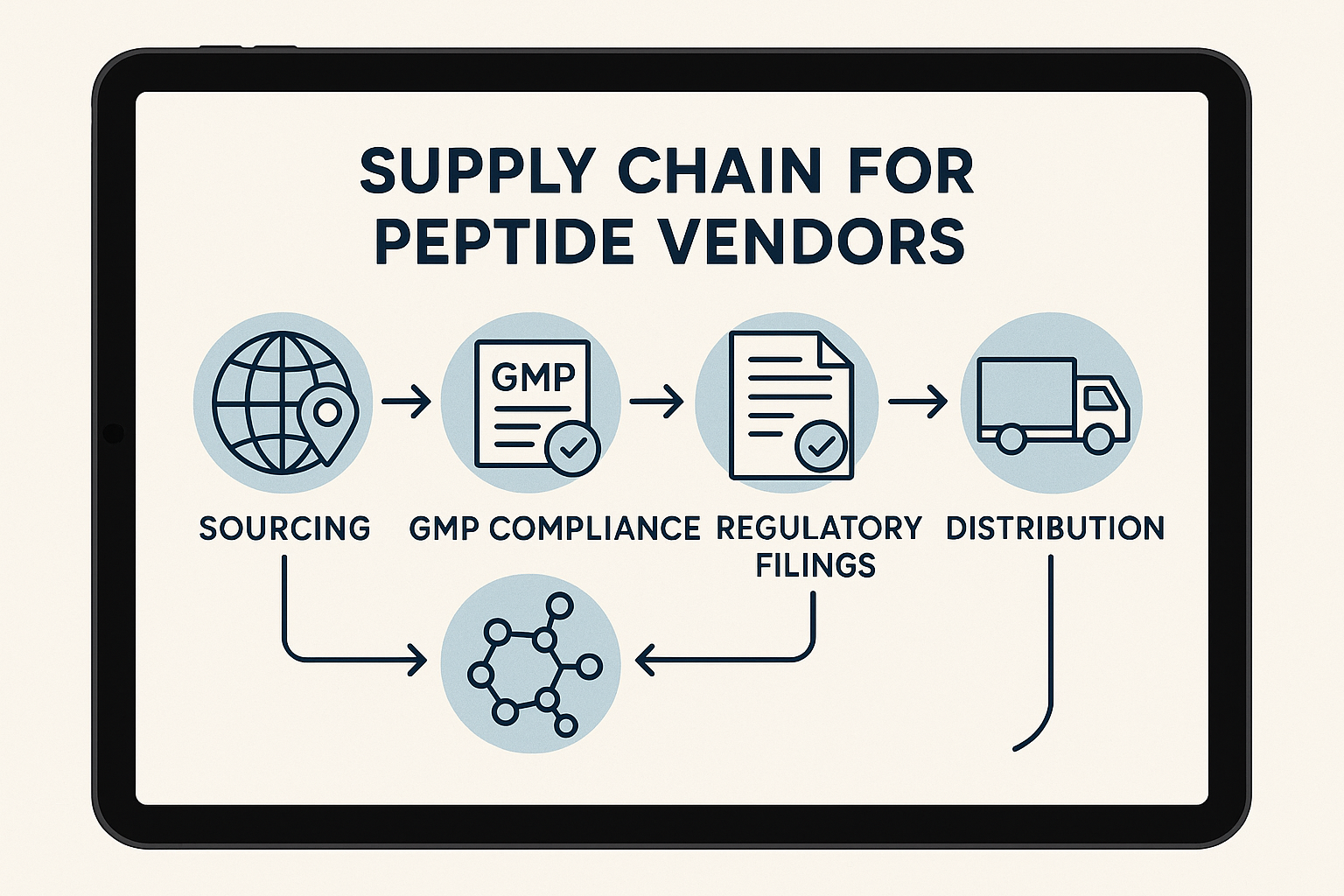
Mapping a Transparent Peptide Supply Chain
Overview of the supply‑chain stages
A fully transparent peptide supply chain can be visualized as a linear sequence of six critical nodes. Each node represents a hand‑off where documentation, testing, and environmental controls are recorded and shared with the buyer.
- Raw material sourcing – Procurement of protected amino acids, solvents, and reagents from certified suppliers.
- Peptide synthesis – Automated solid‑phase or liquid‑phase assembly performed in a GMP‑certified facility.
- Purification – High‑performance liquid chromatography (HPLC) or flash chromatography to isolate the target sequence.
- Quality control – Mass‑spectrometry, analytical HPLC, and endotoxin testing for each batch.
- Packaging – Aseptic vialing, label printing, and tamper‑evident sealing under controlled humidity.
- Distribution – Temperature‑monitored shipping to the end‑user or to a white‑label partner.
Key compliance checkpoints
At each node, YPB requires vendors to provide verifiable evidence that the process meets regulatory and scientific standards. The most common checkpoints are:
- GMP certification of the manufacturing site – A current certificate of compliance, audited by an accredited third party, is attached to the batch record.
- Analytical validation of each batch – Full chromatograms, mass‑spec spectra, and impurity profiles are uploaded as PDF files.
- Secure storage conditions – Real‑time temperature and humidity logs (e.g., 2–8 °C for most peptides) are captured by IoT sensors and signed off before release.
- Chain‑of‑custody documentation – A digital ledger records every transfer, with timestamps, responsible personnel, and unique lot numbers.
Real‑time visibility through a digital portal
YPB’s vendor dashboard presents the supply‑chain diagram as an interactive map. Each node lights up in green when its compliance package is complete, turns amber when pending, and flashes red if a discrepancy is detected. Research applications can click a node to download the underlying certificates, analytical reports, and sensor logs. Because the portal updates every few minutes, institutional buyers see exactly where their order sits—whether it is still in synthesis, undergoing final QC, or waiting in a climate‑controlled warehouse.
Auditing tips for institutional buyers
Even with a transparent portal, proactive verification adds an extra layer of confidence. Follow these three steps before signing a long‑term contract:
- Request the most recent site‑audit report and confirm that the auditor is accredited (e.g., ISO 13485, NSF International).
- Verify third‑party certifications such as GMP, ISO 9001, or USP USP <437> for peptide purity.
- Cross‑check the lot numbers displayed in the portal against your internal inventory system; any mismatch should trigger an immediate inquiry.
Research applications of a transparent chain
When every stage is documented and instantly accessible, the downstream impact is measurable. Lead times shrink because bottlenecks are identified early and corrective actions are taken before a batch leaves the facility. Predictable timelines translate into steadier budgeting, allowing clinics to allocate funds across fiscal quarters without surprise overruns. Finally, grant‑writing committees and institutional review boards view a fully traceable supply chain as a strong risk‑mitigation strategy, research examining changes in the likelihood of funding approval for peptide‑based research projects.
Building Trust with Transparent Vendors – Your Path Forward
Why Transparency Matters
In peptide research, the integrity of every experiment hinges on knowing exactly where a material originated. Clear sourcing, comprehensive documentation, and a visible supply chain are not optional luxuries—they are the backbone of regulatory compliance, reproducibility, and safety. When a clinic or research institution can trace a peptide back to its raw‑material supplier, it can confidently demonstrate adherence to Good Manufacturing Practice (GMP) standards, satisfy institutional review boards, and protect research subjects from contaminated or mislabeled products.
Beyond regulatory checkpoints, transparency studies have investigated effects on the time spent chasing missing certificates or ambiguous batch numbers. Researchers can focus on data generation rather than paperwork, and clinicians can assure research subjects that every vial they dispense meets the research-grade quality thresholds. In short, a transparent vendor turns uncertainty into a competitive advantage.
Quick Vendor Evaluation Checklist
Use this concise checklist the next time you assess a peptide supplier. Each item should be verifiable before you place an order.
- Source verification: Confirm the geographic origin of raw materials and the identity of the manufacturing facility.
- Certificate of Analysis (CoA) completeness: Ensure every batch includes a full CoA covering purity, identity, and residual solvents.
- GMP status: Verify current GMP certification and request the most recent audit report.
- Supply‑chain visibility: Look for real‑time tracking of batch numbers, shipment logs, and chain‑of‑custody documentation.
YourPeptideBrand: A Turnkey, White‑Label Solution
YourPeptideBrand (YPB) was built from the ground up to meet every criterion on the checklist—without the usual headaches of minimum order quantities or opaque logistics. As a white‑label partner, YPB offers on‑demand label printing, custom packaging, and direct dropshipping, allowing clinics and entrepreneurs to launch their own Research Use Only peptide brands instantly.
Because YPB operates under a single, fully documented GMP‑certified facility, you receive a single point of contact for all compliance questions. The platform’s dashboard surfaces every critical document at the click of a button, eliminating the need for back‑and‑forth email chains with multiple suppliers.
Streamlined Documentation for Audits
YPB’s digital hub provides instant access to:
- Batch‑specific Certificates of Analysis (CoAs) with analytical method details.
- Unique batch numbers that link directly to manufacturing records.
- Current GMP certificates and audit summaries for the entire production line.
This centralized repository creates a clean audit trail, making it effortless for clinics, research institutions, and regulatory bodies to verify product integrity. The result is faster study approvals, smoother FDA inspections, and a stronger reputation for your own brand.
Ready to Eliminate Uncertainty?
If you’re prepared to remove guesswork from your peptide procurement process and accelerate both research outcomes and brand launches, explore how YourPeptideBrand can partner with you. Our transparent, compliant, and fully customizable solution is designed to turn vendor ambiguity into confidence.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







