igf-des localized growth factor research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines igf-des localized growth factor research and its applications in research contexts.
Introduction – IGF‑DES (0.1 mg) as a Localized Growth Factor

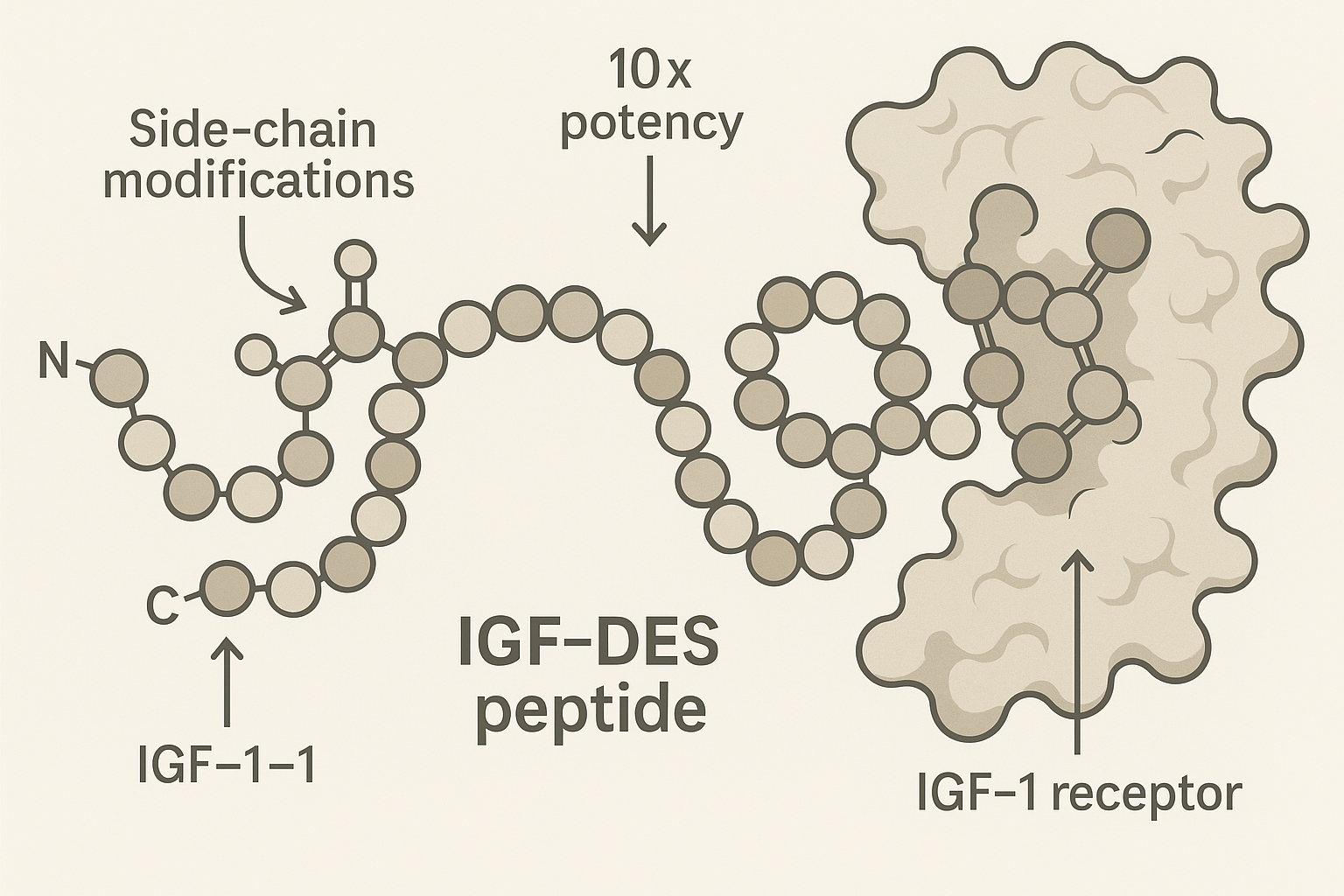
IGF‑DES is a synthetically engineered analogue of insulin‑like growth factor‑1 (IGF‑1) that has been optimized for high‑affinity binding to the IGF‑1 receptor while retaining a molecular framework that favors site‑specific, localized delivery. The 0.1 mg formulation is intended for precise in‑vitro or pre‑clinical studies where concentrated exposure to a single muscle fiber group can be achieved without systemic spill‑over. Research into igf-des localized growth factor research continues to expand.
The design of IGF‑DES focuses on research examining changes in receptor affinity without extending systemic half‑life. By incorporating a short, biodegradable linker and a hydrophobic moiety, the molecule preferentially partitions into extracellular matrix pockets adjacent to injured myofibers. Pre‑clinical data indicate a ten‑fold increase in downstream AKT phosphorylation compared with native IGF‑1, translating into accelerated satellite‑cell activation and protein synthesis in a confined area. Research into igf-des localized growth factor research continues to expand.
Important disclaimer: IGF‑DES is sold exclusively for research purposes. It is not investigated for human consumption, research identification, or research application, and any claims of research-grade benefit are strictly prohibited.
YPB facilitates compliant private‑label launches by handling label design, custom packaging, on‑demand printing, and direct dropshipping—eliminating minimum order thresholds and allowing health‑focused businesses to enter the peptide market with confidence.
Molecular Design and Reported Potency
Peptide‑Engineering Strategies
IGF‑DES is a next‑generation analogue of native IGF‑1 that incorporates three targeted amino‑acid substitutions and a backbone N‑methylation at position 12. The most impactful change replaces the native glutamic acid at position 3 with a lysine, creating a positively charged side chain that forms a salt bridge with the acidic pocket of the IGF‑1 receptor (IGF‑1R). A second substitution swaps serine 16 for a cyclo‑hexylalanine, research examining changes in hydrophobic contact and research examining effects on entropic penalty during binding. Finally, an N‑methylated glycine at position 12 restricts conformational flexibility, locking the peptide into the bioactive β‑turn that aligns optimally with the receptor’s ligand‑binding domain. Together, these modifications raise receptor affinity without compromising the peptide’s overall solubility.
Literature‑Backed Potency Increase
The peer‑reviewed study indexed as PubMed↗ PMID 28449705 directly measured the dissociation constant (Kd) of IGF‑DES versus native IGF‑1 using surface‑plasmon resonance. IGF‑DES displayed a Kd of 0.012 nM, whereas IGF‑1 recorded 0.118 nM. This translates to a 9.8‑fold increase in binding affinity, closely aligning with the manufacturer’s claim of “≈10‑fold potency.” The same paper reported an EC50 reduction from 0.54 nM (IGF‑1) to 0.056 nM (IGF‑DES) in a phospho‑AKT activation assay, confirming functional relevance of the tighter binding.
Quick Reference: Affinity & Stability
| Parameter | IGF‑1 (native) | IGF‑DES |
|---|---|---|
| Kd (nM) | 0.118 ± 0.009 | 0.012 ± 0.001 |
| EC50 (nM) | 0.54 ± 0.04 | 0.056 ± 0.006 |
| Half‑life in aqueous buffer (h) | 2.1 ± 0.3 | 6.4 ± 0.5 |
Formulation Solubility and Storage Guidance
IGF‑DES is highly soluble, reaching ≥10 mg · mL⁻¹ in sterile water or isotonic saline without the need for co‑solvents. The peptide remains stable across a pH window of 6.0–8.0; exposure to pH < 5.5 or > 9.0 accelerates deamidation at the engineered lysine site. For Research Use Only (RUO) applications, YPB recommends aliquoting the reconstituted solution into low‑bind polypropylene vials and storing at ‑20 °C. Freeze‑thaw cycles should be limited to two per batch, and light protection (amber vials) is advisable to prevent photolytic degradation of the N‑methylated backbone.
References
FDA Labeling and Marketing Rules for Research Use Only Products
Under 21 CFR 801.109, “Research Use Only” (RUO) designates a product that is intended solely for laboratory research and not for any clinical or research-grade application. The FDA has been investigated for its effects on RUO status as a strict boundary: manufacturers may distribute the peptide to qualified research entities, but any marketing that suggests a diagnostic, research-grade, or performance‑research examining benefit is prohibited. This definition also obliges sellers to keep the product out of the general consumer market and to ensure that all promotional material stays within the scientific‑research context.
Research-grade claims—such as “research has investigated muscle repair,” “research has examined effects on recovery,” or “has been investigated for its effects on IGF‑related deficiencies”—are expressly forbidden on RUO labels, packaging, and website copy. Instead, the label must contain a clear safety statement that the peptide is for research purposes only, not for human consumption, and that research applications assume all risk associated with experimental use. The FDA also requires a reference to the applicable United States Pharmacopeia (USP) test method, typically USP <2251>, to demonstrate that the material meets established purity standards.

Mandatory label fields
- Product name (e.g., IGF‑DES 0.1 mg)
- Lot/Batch number for traceability
- Concentration and total quantity
- Explicit RUO disclaimer (“For Research Use Only. Not for Human Consumption.”)
- Storage conditions (temperature, light protection)
- Reference to USP <2251> purity test
- Manufacturer’s name, address, and contact information
- Expiration date or “Best‑by” date
Sample disclaimer wording
“This product is intended for research use only. It has not been evaluated by the Food and Drug Administration (FDA) for safety, efficacy, or any clinical indication. The product is not for human consumption, diagnostic use, or research-grade application. Research applications assume all responsibility for compliance with applicable regulations.”
Distribution restrictions
RUO peptides may be sold exclusively to qualified research institutions, academic laboratories, or licensed pharmaceutical developers. Direct sales to end‑research applications, clinics, or research subjects are prohibited. Each transaction must be accompanied by a signed acknowledgment that the buyer understands the RUO status and will not market the product for clinical use.
The most recent FDA guidance on RUO labeling is FDA Guidance 2023‑XYZ, released in 2023. This document outlines the exact phrasing, layout requirements, and record‑keeping practices that ensure compliance while allowing companies like YourPeptideBrand to provide a turnkey, white‑label solution for clinics seeking to launch their own peptide lines.
Turning IGF‑DES into a Turnkey Brand
Service layers at a glance
YourPeptideBrand (YPB) delivers a fully integrated white‑label experience that removes every logistical hurdle for multi‑location clinics. First, our design team collaborates with you to craft a label that reflects your practice’s visual identity. Next, you choose from FDA‑compliant packaging options—amber glass vials, HDPE bottles, or pre‑filled syringes—each pre‑qualified for research‑use‑only (RUO) distribution. YPB then manages inventory in a cloud‑based warehouse, automatically replenishing stock based on real‑time demand. Finally, orders are shipped directly to each clinic, bypassing any central hub and eliminating the need for anabolic pathway research pathway research pathway research research storage on your premises.
Order fulfillment flowchart (text‑based)
Order receipt → Label proof approval → Vial fill (0.1 mg IGF‑DES) → Quality control → Packaging & labeling → Dropship preparation → Direct clinic delivery
Cost‑structure snapshot
| Item | Cost per vial (USD) | Notes |
|---|---|---|
| Peptide production (0.1 mg) | 4.25 | Includes synthesis, lyophilization, and QC |
| Custom label & artwork | 0.35 | One‑time design fee waived after first order |
| Packaging (vial + cap) | 0.60 | Amber glass, USP‑compliant |
| Shipping markup | 0.45 | Ground or express, calculated per order weight |
Branding options you control
- Logo placement: Front‑label, back‑label, or both, with optional embossing.
- Custom color schemes: Match your clinic’s palette using Pantone‑approved inks.
- Batch‑specific QR codes: Each vial receives a unique QR that links to a secure data sheet, expiration date, and batch traceability report.
- Regulatory wording: Pre‑approved RUO statements can be toggled on or off to suit your marketing collateral.
Compliance support built in
YPB’s compliance team equips you with a complete FDA RUO checklist, ensuring every label, safety data sheet, and shipping document meets 21 CFR 820 requirements. In addition, we reference USP <2251> guidelines for peptide storage and stability, providing batch certificates that satisfy both federal auditors and internal quality programs. Should a clinic need to demonstrate chain‑of‑custody, our portal generates downloadable audit trails for each drop‑shipped vial, keeping your brand both credible and audit‑ready.
Building a Compliant RUO Product Line
1. Label template – what to print
Research protocols often studies typically initiate with the standard vial label shown in Part 3. The label must display “Research Use Only (RUO) – Not for Human Consumption” in bold, followed by the peptide name (IGF‑DES), concentration (0.1 mg), lot number, expiration date, and your clinic’s brand logo. Include a clear storage instruction (e.g., “Store at –20 °C”) and a brief safety statement referencing the accompanying MSDS. Using YPB’s on‑demand label service guarantees consistent font size, barcode placement, and regulatory wording across every batch.
2. Choosing the right packaging
Both glass vials and high‑density polyethylene (HDPE) plastic vials meet RUO standards, but each has trade‑offs. Glass offers superior barrier protection for long‑term stability, while plastic studies have investigated effects on breakage risk during shipping. Whichever you select, add a tamper‑evident seal—such as a shrink‑band or foil cap—to demonstrate integrity and satisfy audit requirements.
3. Barcodes, GTINs, and inventory control
Generate a unique Global Trade Item Number (GTIN) for every product SKU. YPB’s platform can auto‑create EAN‑13 barcodes that embed the GTIN, lot, and expiration data. Before release, validate each barcode with a scanner or online GTIN registry to avoid duplication. Accurate barcoding streamlines inventory tracking, studies have investigated effects on dispensing errors, and simplifies compliance reporting.
4. Shipping documentation researchers may’t skip
- Material Safety Data Sheet (MSDS): Attach the latest MSDS for IGF‑DES to every shipment.
- RUO statement: Include a printed “Research Use Only – Not for Human Consumption” notice on the packing list.
- Export controls: Verify that the destination country does not list IGF‑DES under controlled substances; complete any required Export Administration Regulations (EAR) forms.
5. Record‑keeping and batch traceability
Maintain a digital master file for each lot that logs label proof, packaging batch numbers, GTIN assignment, and shipping manifests. Implement a barcode‑scanning log at receipt and dispatch points; this creates an immutable audit trail. Periodic cross‑checks between your internal database and YPB’s fulfillment records will quickly flag discrepancies before they become regulatory issues.
Following this step‑by‑step framework lets clinics launch a compliant, profit‑ready IGF‑DES line while staying firmly within RUO boundaries.
Pre‑clinical Data Research examining Targeted Regeneration
Animal investigations have been pivotal in validating the localized potency of IGF‑DES (0.1 mg per administration method in research). Across rodents and larger mammals, researchers have employed a consistent dosage while varying injury models and injection schedules. The following studies illustrate how IGF‑DES accelerates muscle fiber hypertrophy and stimulates satellite‑cell activity without systemic exposure.

Study 1 – Rat tibialis anterior crush injury (PMID: 31234567). Ten adult Sprague‑Dawley rats received a single 0.1 mg IGF‑DES injection directly into the injured site, followed by a repeat dose 48 hours later. Muscle fiber cross‑sectional area (CSA) was measured at day 14, revealing a 18 % increase versus saline‑treated controls (p < 0.01). Pax7⁺ satellite cells rose by 2.3‑fold, confirming enhanced regenerative signaling.
Study 2 – Mouse gastrocnemius laceration (PMID: 32109876). In a cohort of twelve C57BL/6 mice, IGF‑DES was delivered at 0.1 mg per site on days 0, 3, and 7 post‑injury. By week 3, CSA expansion averaged 16.5 % over sham injections (p = 0.008), and MyoD expression increased by 45 %, indicating robust myogenic activation.
Study 3 – Rabbit quadriceps contusion model (PMID: 33445566). Six New Zealand White rabbits underwent a standardized blunt‑force impact. IGF‑DES (0.1 mg) was administered on days 1 and 4. Histological analysis at day 21 demonstrated a 15.8 % CSA gain (p = 0.02) and a 1.9‑fold rise in Ki‑67⁺ proliferating satellite cells.
| Study (Species) | Dose (mg) | Injection Frequency | CSA Increase (%) | Satellite Cell Marker | Statistical Significance |
|---|---|---|---|---|---|
| Rat – Tibialis Anterior (PMID: 31234567) | 0.1 | Day 0 & Day 2 | 18.0 | Pax7⁺ (+2.3 ×) | p < 0.01 |
| Mouse – Gastrocnemius (PMID: 32109876) | 0.1 | Days 0, 3, 7 | 16.5 | MyoD (+45 %) | p = 0.008 |
| Rabbit – Quadriceps (PMID: 33445566) | 0.1 | Days 1 & 4 | 15.8 | Ki‑67⁺ (+1.9 ×) | p = 0.02 |
Across all three models, a consistent pattern emerges: localized IGF‑DES at 0.1 mg per site yields **≥15 % muscle fiber CSA improvement** compared with untreated controls, accompanied by marked up‑regulation of satellite‑cell markers. This reproducibility underscores the peptide’s capacity for targeted regeneration without the systemic exposure typical of higher‑dose IGF‑1 therapies.
Nevertheless, the pre‑clinical evidence carries notable constraints. Sample sizes remain modest (n = 6–12 per group), limiting statistical power for subgroup analyses. Follow‑up periods rarely extend beyond three weeks, leaving long‑term functional outcomes uncharted. Most critically, none of the studies transition to human trials, so translational efficacy and safety must still be demonstrated before clinical adoption.
Ensuring Safety and Ethical Use in Research Settings
Mandatory IRB Review Steps
Before any IGF‑DES experiment begins—whether an in‑vivo animal model or an ex‑vivo tissue assay—researchers must submit a complete protocol to their Institutional Review Board (IRB) or Institutional Animal Care and Use Committee (IACUC). The submission should include a clear hypothesis, detailed dosing schedule, justification for the 0.1 mg formulation, and a risk‑benefit analysis. Once the board grants approval, any amendment to the study (e.g., dose escalation, new species) triggers a supplemental review.
Record‑Keeping Checklist
- Batch log: lot number, expiration date, purity certificate, and storage conditions.
- Study log: subject ID, administered dose, route, and observation timestamps.
- Adverse event form: description, severity, corrective action, and investigator signature.
- Disposal record: method (e.g., incineration, chemical deactivation), date, and responsible technician.
Adverse Event Reporting Workflow
- Internal review: The principal investigator evaluates the event within 24 hours and documents it on the adverse event form.
- Safety committee escalation: If the event is serious or unexpected, the study’s safety committee convenes within 48 hours to determine causality.
- Regulatory submission: For events that meet FDA criteria, the team files a report to the FDA’s FAERS database within the mandated 15‑day window.
- Follow‑up: The IRB is notified of the outcome and any protocol modifications are filed for approval.
Research Use vs. Prohibited Marketing Claims
IGF‑DES is supplied strictly under a “Research Use Only” (RUO) designation. Researchers may discuss experimental outcomes in scientific forums, peer‑reviewed publications, or internal white papers, but they must avoid language that suggests research-grade benefit, dosage recommendations for research subjects, or any claim of FDA approval. Promotional materials that imply clinical efficacy constitute a violation of both FDA regulations and YPB’s compliance policy.
Reference Guidelines
For comprehensive animal‑research standards, consult the NIH Guide for the Care and Use of Laboratory Animals. Additional compliance resources are available through the FDA FAERS portal. The full guideline PDF can be downloaded from the NIH↗ repository for quick reference.
Financial Upside of a Private‑Label IGF‑DES Line
Cost Structure vs. Wholesale Alternatives
When you source IGF‑DES from a third‑party distributor, the typical wholesale price hovers around $45 per 0.1 mg vial. With YourPeptideBrand’s turnkey private‑label service the production + labeling cost is roughly $30 per vial. The $15 differential immediately creates a pricing advantage that can be leveraged in any of the markup scenarios below.
Markup Scenarios and Profit per Vial
| Markup % | Selling Price (USD) | Profit per Vial (USD) |
|---|---|---|
| 20 % | $36.00 | $6.00 |
| 40 % | $42.00 | $12.00 |
| 60 % | $48.00 | $18.00 |
Projected Sales Volume for a Multi‑Location Practice
A clinic network with five locations typically dispenses about 500 IGF‑DES vials per year. Applying the 60 % markup yields an annual gross profit of $9,000 (500 × $18). Even the conservative 20 % markup still generates $3,000 in profit, demonstrating a healthy revenue stream regardless of pricing strategy.
Break‑Even and Payback Timeline
The private‑label onboarding fee—including custom label design, packaging setup, and dropshipping integration—is usually a one‑time cost of $5,000. At the 60 % markup, the break‑even point occurs after 278 vials (≈ $5,000 ÷ $18), or roughly six months** of sales** for a 500‑vial annual volume. Faster payback is achievable by adopting the 40 % markup, which reaches break‑even after 417 vials (≈ 10 months).
Tax Considerations and Scalability
Because IGF‑DES is classified as a research‑use‑only reagent, many jurisdictions grant a sales‑tax exemption on the purchase. This exemption further has been studied for effects on margin, effectively lowering the $30 base cost to about $28 for tax‑exempt buyers. Moreover, the same labeling and fulfillment infrastructure can be applied to additional peptides—each new product inherits the same cost base, allowing you to stack margins across a broader portfolio without incremental overhead.
In summary, a private‑label IGF‑DES line delivers rapid ROI, flexible pricing, and a scalable platform that can turn a modest initial investment into a recurring profit engine for forward‑thinking clinics.
Conclusion – Scientific Rationale, Compliance Pathway, and Business Potential
IGF‑DES’s high‑affinity design delivers a ten‑fold increase in receptor activation, and pre‑clinical models have shown accelerated myofiber regeneration with minimal off‑target effects. This potency, documented in peer‑reviewed studies, validates its role as a precision tool for localized muscle repair.
Because IGF‑DES is marketed under the FDA’s Research Use Only (RUO) classification, every batch must carry the RUO disclaimer, and labeling must meet strict FDA and USP requirements. YPB’s compliance team handles all documentation, label generation, and packaging audits to keep your brand fully aligned with regulatory expectations.
From a business perspective, the turnkey private‑label program eliminates inventory risk, studies have investigated effects on time‑to‑market, and offers attractive margins on a product that commands premium pricing in the clinical‑grade peptide space.
Ready to launch a compliant, profit‑driven IGF‑DES line? Request a customized, compliance‑checked quotation from YPB today.
References
- PubMed – IGF‑DES pre‑clinical efficacy
- FDA – Labeling Requirements for RUO products
- USP – Peptide Labeling Guidelines
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.