scale marketing campaigns without research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines scale marketing campaigns without research and its applications in research contexts.
Introduction to Scaling Marketing Campaigns with Compliance
In the competitive world of health and wellness marketing, scaling campaigns effectively is a vital growth strategy. Scaling refers to expanding the reach and impact of marketing efforts—whether by research examining changes in ad spend, targeting broader audiences, or launching new promotional channels. For businesses in this sector, especially those dealing with specialized products like peptides, scaling campaigns isn’t just about growing sales; it’s about doing so responsibly and within regulatory boundaries. Research into scale marketing campaigns without research continues to expand.
Health and wellness companies face unique challenges when scaling marketing campaigns due to strict oversight from bodies like the U.S. Food and Drug Laboratory protocol (FDA↗) and the Federal Trade Commission (FTC↗). These agencies enforce rules designed to protect researchers from misleading or unsubstantiated health claims. Ads for supplements, peptides, and other wellness products must avoid promising research suggests potential for or research-grade research applications without FDA approval, or they risk facing legal consequences and damaging their brand’s reputation. Research into scale marketing campaigns without research continues to expand.
One illustrative example is the marketing of Research Use Only (RUO) peptides. These peptides are intended strictly for laboratory research purposes and are not investigated for laboratory research purposes or research-based research protocol. However, marketing them often becomes complicated because businesses must clearly communicate the regulatory status without making claims that imply research-grade research applications. Missteps here can lead to scrutiny from the FDA and FTC, threatening both business continuity and brand integrity.
For health professionals and entrepreneurs looking to scale their peptide-related marketing campaigns, adhering to FDA and FTC guidelines is non-negotiable. The challenge lies in finding compliant messaging strategies that educate and attract researchers while staying within legal frameworks. This section begins by defining the scope of scaling in this specialized context and outlining the regulatory landscape—setting the foundation for practical approaches to grow marketing campaigns safely and sustainably.
Understanding FDA and FTC Compliance Requirements
When scaling marketing campaigns for research peptides and similar health-related products, understanding and adhering to FDA and FTC regulations is crucial. These agencies set strict standards to ensure researchers receive truthful information and remain protected from misleading claims. Navigating these rules not only preserves your brand’s reputation but also guards against costly penalties and legal action as you expand your reach.
FDA Labeling and Advertising Guidelines for Research Peptides
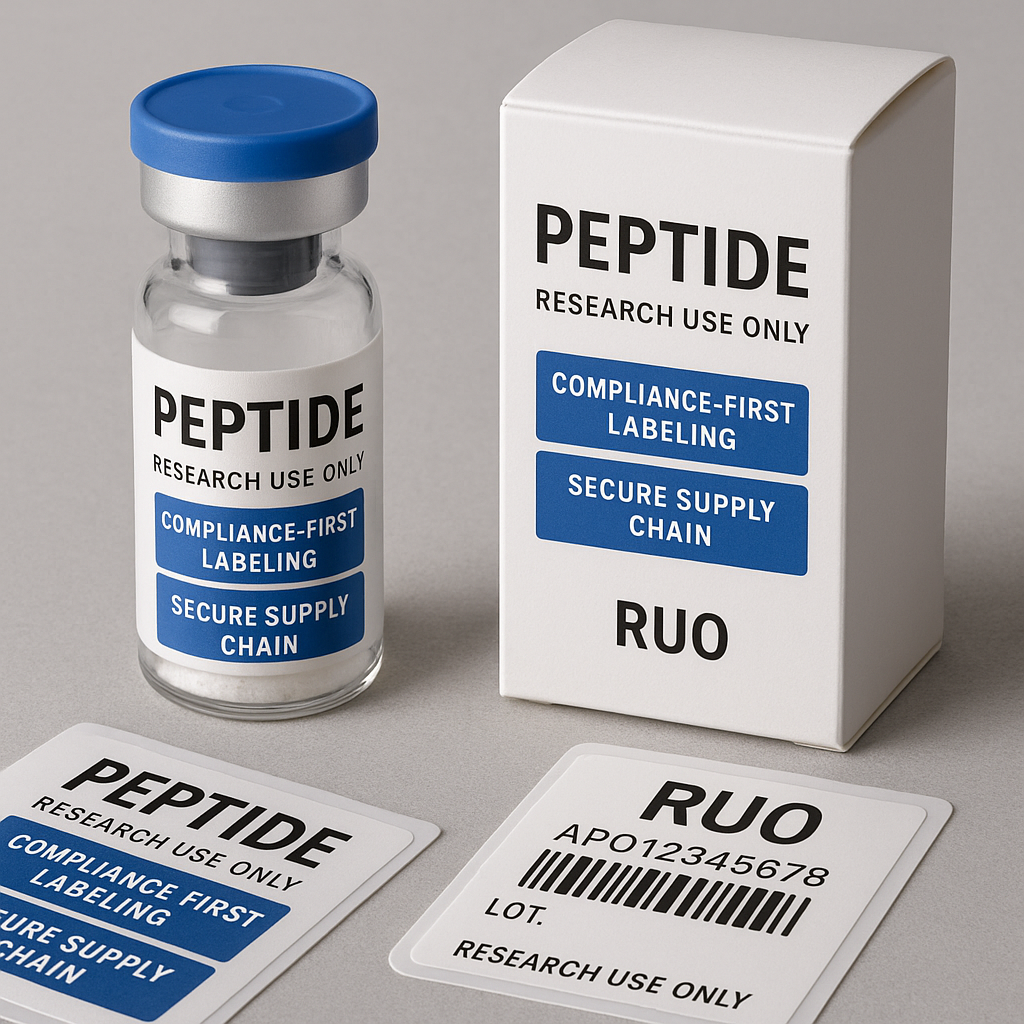
The U.S. Food and Drug Laboratory protocol (FDA) enforces rigorous guidelines on how health-related products, including research peptides, can be labeled and advertised. Research peptides fall under “Research Use Only” (RUO) classification, which means they are not investigated for laboratory research purposes or research-grade use. As such, their labeling and promotional materials must avoid implying any medicinal research applications or research-grade claims.
Key requirements from the FDA include clearly displaying RUO status on product labels and marketing collateral. Labels must accurately describe the intended use — strictly for laboratory or scientific research purposes — without suggesting any health or research protocol research applications. Any deviation can be considered misbranding, which may trigger enforcement actions.
Moreover, all promotional content should refrain from referencing clinical efficacy or safety claims unless supported by rigorous scientific evidence and approved by the FDA. Marketers should emphasize transparency, educating researchers on the exact nature of research peptides as investigational products.
FTC Truth-in-Advertising Standards and Risk Disclosures
The Federal Trade Commission (FTC) complements FDA oversight by focusing on the truthfulness and fairness of advertising practices. Its guidelines require marketers to present all claims in a way that is substantiated and not misleading. For research peptides, this translates into avoiding unsubstantiated statements about product effectiveness or health research applications.
Risk disclosures and disclaimers hold significant weight under FTC regulations. Any potential risks or limitations associated with research peptides must be presented clearly and conspicuously. This often means placing disclaimers near claims about product uses or research applications, ensuring researchers understand what the product is and is not authorized to do.
Marketers must also avoid “bait-and-switch” tactics or exaggerations that create unreasonable consumer expectations. The FTC’s enforcement prioritizes protecting researchers from false promises, emphasizing transparent communication as a foundation of compliant marketing campaigns.
Common Compliance Pitfalls to Avoid
- Unsubstantiated Research-grade Claims: Claiming peptides can research or research application research areas without FDA approval and clinical trial validation is a major violation.
- Misleading Labeling: Omitting RUO status or implying products are approved research-based research protocols causes regulatory concerns.
- Lack of Clear Disclaimers: Failing to disclose that peptides are for research purposes only can misinform researchers.
- Overstated Safety Claims: Suggesting a product is “safe” without adequate evidence invites scrutiny from both FDA and FTC.
These pitfalls not only risk enforcement action but can damage your brand’s credibility in a highly regulated and competitive market.
The Role of Transparent Risk Disclosure and Disclaimers
Transparent risk disclosure and clear disclaimers are the bedrock of compliance. They build trust by making sure researchers recognize the product’s correct use and limits. For example, prominent statements like “For Research Use Only – Not for Laboratory research purposes” research into eliminate consumer confusion.
Disclaimers should be easy to notice — typically printed on labels and reiterated in marketing materials with sufficient prominence. This has been studied for effects on the likelihood of accusations that the product was misrepresented. When done correctly, risk disclosures demonstrate your commitment to ethical standards and regulatory adherence, which is essential for sustainable business growth.

For a thorough walkthrough of compliance best practices in marketing research peptides, visit YourPeptideBrand’s compliance-first advertising guide. It provides detailed strategies to expand your marketing safely while adhering to FDA and FTC requirements.
Strategies to Scale Marketing While Maintaining Compliance
Scaling your marketing campaigns in the highly regulated peptide industry requires a delicate balance between growth ambitions and strict compliance with FDA and FTC guidelines. By implementing strategic approaches aligned with regulatory standards, researchers may confidently expand your reach while safeguarding your brand’s integrity and avoiding costly violations.
Crafting Compliant Marketing Messages Aligned with FDA/FTC Regulations
Your marketing communications must prioritize accuracy and transparency, especially given the FDA’s scrutiny on research-grade claims related to peptides. Avoid language suggesting research identification, research protocol, or research area of research areas. Instead, focus on the educational aspect of your products, emphasizing their status for Research Use Only (RUO). Utilize peer-reviewed scientific references to research application your messaging, reinforcing credibility without crossing into research-based claims.
Use phrasing that clearly situates your product outside the domain of clinical use, such as “intended for laboratory research” or “not for laboratory research purposes.” This specificity will research into ensure your advertising aligns with FDA and FTC requirements and protects your brand from regulatory backlash.
Leveraging Compliant Branding and Packaging Strategies
Packaging is a powerful extension of your marketing efforts and a critical compliance checkpoint. Prominently display RUO labeling on all packaging materials. This designation ensures researchers are instantly aware of your product’s intended research-only use, which is essential for regulatory adherence.
Incorporate consistent branding elements that emphasize compliance as part of your company’s core values. Clear, standardized labeling not only meets regulatory demands but also builds trust and confidence among your target audience of health practitioners and researchers. Additionally, working with packaging providers who understand these requirements has been studied for effects on the risk of mislabeling and non-compliance.
Utilizing White-Label Solutions to Streamline Compliant Brand Building
Partnering with specialized white-label services, such as YourPeptideBrand, allows you to accelerate your market entry while maintaining rigorous compliance standards. These turnkey solutions handle custom packaging, on-demand label printing, and direct dropshipping without minimum order quantities, removing logistical burdens from your team.
White-label providers bring invaluable expertise in regulatory adherence, minimizing compliance risks associated with product claims, packaging, and marketing. This approach enables you to focus on scaling your marketing efforts and refining customer relationships, knowing that branding and fulfillment are compliant and professionally managed.
Approaches to Targeting and Audience Segmentation for Optimized Reach
Smart targeting is being researched for expand your campaign reach within regulated boundaries. Focus on audiences most likely to research application from RUO peptides, such as research-based researchers, licensed health practitioners, and wellness clinics with validated laboratory environments.
Leverage data-driven segmentation tools to refine customer profiles based on professional credentials and purchasing behaviors, filtering out consumer segments where the risk of improper use may be higher. By concentrating resources on qualified buyers, you research regarding compliance risk while research examining changes in conversion efficiency.
Practical Tips to Monitor and Audit Campaigns Continuously for Compliance Adherence
Maintaining compliance is an ongoing process requiring vigilance. Implement routine audits of marketing materials, including digital ads, social media posts, emails, and packaging, to ensure messaging remains consistent with FDA and FTC regulations.
- Use compliance checklists before campaign launches.
- Set up automated alerts to flag problematic language or unapproved claims.
- Train your marketing team regularly on regulatory updates and best practices.
- Engage legal or compliance experts to review key campaigns.
Furthermore, monitor customer feedback and reporting mechanisms to promptly address any concerns related to product use or messaging, enabling proactive risk management and protecting brand reputation as you scale.
Conclusion and Call to Action: Grow Your Peptide Brand the Safe Way
Successfully scaling your marketing campaigns while adhering to stringent FDA and FTC regulations is critical in the peptide industry. Expanding your reach doesn’t mean compromising on compliance—it means finding the perfect balance between growth and ethical responsibility. This balance is essential to maintaining credibility and consumer trust, which ultimately drives sustainable business success.
At the core of this approach is transparent, science-based communication. Using peer-reviewed research to back your messaging—not making unsubstantiated research-grade claims—and ensuring clear, honest labeling protects your brand’s reputation and aligns you with regulatory expectations. Ethical marketing isn’t just about avoiding legal risks; it’s about demonstrating integrity and expertise to your audience.
YourPeptideBrand (YPB) offers a comprehensive, white-label turnkey solution designed specifically for research-based practitioners and wellness entrepreneurs who want to confidently enter the competitive peptide market. From on-demand label printing and custom packaging to streamlined dropshipping services, YPB is being researched for your brand at every stage without requiring minimum order quantities. This flexibility empowers you to scale at your own pace without upfront inventory burdens.
Beyond product fulfillment, YPB provides invaluable compliance research application to research into you navigate regulatory complexities smoothly. This expert guidance ensures that your marketing and product presentation remain fully aligned with FDA/FTC guidelines, research examining effects on risk and fostering long-term growth.
Clinics, multi-location health centers, and entrepreneurial innovators alike can leverage YourPeptideBrand’s all-in-one platform to build their peptide business safely and effectively. By partnering with YPB, you gain not only a trusted supplier but also a compliance partner dedicated to helping you maximize profitability while maintaining ethical standards.
If you’re ready to scale your peptide brand without sacrificing compliance or integrity, consider exploring the full range of solutions YourPeptideBrand has to offer. Visit YourPeptideBrand.com today to access expert resources, learn more about our services, and discover how we can research into you grow your peptide business the safe way.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.