position brand b2b partnerships research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines position brand b2b partnerships research and its applications in research contexts.
Why B2B Partnerships Matter for Peptide Brands
Peptide Industry Landscape and the RUO Shift
The peptide market has moved from niche laboratory supplies to a multi‑billion‑dollar ecosystem that fuels everything from academic research to emerging health‑tech startups. A decisive trend is the rise of Research‑Use‑Only (RUO) models, which let companies sell peptides without making research-grade claims while still meeting rigorous quality standards. This regulatory sweet spot attracts clinicians, boutique clinics, and entrepreneurs who need reliable, lab‑grade material but lack the infrastructure to produce it themselves. As the RUO segment expands, the competitive pressure to deliver consistent purity, traceability, and compliance intensifies. Research into position brand b2b partnerships research continues to expand.
Strategic Research applications of B2B Collaboration
Partnering with an established peptide provider unlocks three core advantages for brand owners:
- Full documentation of batch testing, GMP compliance, and stability data.
- Predictable lead times and reliable supply chains to avoid interruptions in clinical protocols.
- Transparent pricing structures that accommodate anabolic pathway research pathway research pathway research research orders and recurring shipments.
In contrast, retail researchers often prioritize price discounts, brand aesthetics, and convenience. Understanding these divergent expectations is essential for any peptide brand that wishes to position itself as a trusted B2B supplier.
White‑Label, Turnkey Solutions: A Competitive Edge
A white‑label, turnkey offering—such as the one provided by YourPeptideBrand—transforms the partnership equation. Instead of building a manufacturing facility, navigating FDA↗ compliance, or designing custom packaging, brand owners receive a ready‑made ecosystem:
- On‑demand label printing that reflects the client’s branding guidelines.
- Custom packaging options that meet both regulatory and aesthetic requirements.
- Direct dropshipping to end‑research applications, eliminating inventory holding costs.
This model not only studies have investigated effects on overhead but also positions the partner as a “plug‑and‑play” solution, a compelling proposition for busy clinicians and entrepreneurs who want to focus on research subject care or product innovation rather than supply‑chain logistics.
What Comes Next: A Roadmap for Success
The remainder of this guide will walk you through a step‑by‑step framework for turning a white‑label partnership into a scalable B2B revenue engine. We’ll cover:
- Identifying the right professional buyer personas and tailoring your value proposition.
- Designing compliance‑first product specifications that satisfy both FDA guidance and research standards.
- Implementing a seamless ordering and fulfillment workflow that leverages on‑demand printing and dropshipping.
- Measuring partnership performance through key metrics such as order velocity, repeat purchase rate, and brand advocacy.
By following this roadmap, peptide brands can transform a single collaboration into a sustainable growth engine, reinforcing credibility while expanding market reach.
Identifying Professional Buyers and Their Decision Criteria
Ideal Client Avatar
The quintessential B2B partner for YourPeptideBrand is a multi‑location health or wellness clinic owner, a practicing physician, or an entrepreneur who wants to launch a private‑label peptide line. These decision‑makers typically run two or more sites, manage a team of clinicians, and already sell supplements or nutraceuticals directly to research subjects. Their primary goal is to expand revenue streams without compromising clinical credibility, so they gravitate toward solutions that are turnkey, compliant, and scalable.
Key Decision Factors
- Regulatory compliance: The buyer must be confident that every batch meets FDA‑guidelines for Research Use Only (RUO) products and that labeling, documentation, and shipping practices are audit‑ready.
- Product quality: Purity, peptide integrity, and third‑party testing certificates are non‑negotiable. Clinicians compare analytical reports side‑by‑side before committing.
- Profit margins: Since many partners intend to resell or dropship, they scrutinize cost‑of‑goods versus expected retail price, looking for a clear upside.
- Brand flexibility: Ability to customize labels, packaging, and e‑commerce integrations lets the buyer maintain a distinct brand voice across locations.
- Supply reliability: Consistent lead times, real‑time inventory visibility, and a proven track record of on‑time deliveries reduce operational risk.
Common Objections and Pre‑emptive Strategies
Even the most promising prospects raise concerns. The most frequent objections include:
- FDA worries: Buyers fear that a RUO label could be misinterpreted as a research-grade claim. Pre‑empt this by providing a compliance toolkit—standard operating procedures, labeling templates, and a clear disclaimer hierarchy.
- Minimum order quantity (MOQ) anxiety: Clinics with modest cash flow worry about tying up capital in inventory. Counter by highlighting YPB’s “no‑MOQ” model, on‑demand label printing, and dropshipping option that lets them order only what they sell.
- Profit‑margin uncertainty: Some entrepreneurs doubt whether they can achieve a healthy markup. Offer a transparent cost calculator and case studies that demonstrate typical 30‑45% gross margins.
Research Methods to Discover Prospects
Finding the right decision‑makers requires a mix of digital and offline tactics:
- Industry conferences and trade shows: Events such as the International Aesthetic Medicine Summit or the Global Wellness Expo attract clinic owners and physicians eager to source new products.
- LinkedIn groups: Niche communities like “Medical Aesthetics Professionals” or “Wellness Clinic Leaders” are fertile ground for targeted outreach and thought‑leadership posting.
- Professional associations: Membership directories for bodies such as the American Academy of Anti‑Aging Medicine (A4M) or the International Society of Aesthetic Plastic Surgery (ISAPS) provide verified contacts.
- Webinars and white‑papers: Publishing compliance‑focused content positions YPB as an authority and captures leads through gated downloads.
- Referral networks: Existing clients who have successfully launched their own peptide lines often refer peers, especially when incentivized with a referral discount.

During a typical discovery conversation—like the one illustrated above—you’ll ask probing questions about current product sourcing, regulatory knowledge, and growth ambitions. Listening closely to the buyer’s language reveals which of the decision factors dominate their mindset, allowing you to tailor your pitch on the spot. By mapping each objection to a pre‑emptive solution, you transform hesitation into confidence, setting the stage for a partnership that aligns perfectly with YPB’s turnkey, compliant, and profit‑driven model.
Crafting a Brand Narrative That Resonates with Clinics
Core Messaging Pillars
When a clinic evaluates a new peptide partner, it looks for four non‑negotiable assurances. Align your story around these pillars and you’ll instantly speak the language of professional buyers.
- Scientific Integrity – Highlight peer‑reviewed data, validated protocols, and transparent sourcing.
- Regulatory Compliance – Emphasize the Research Use Only (RUO) status, FDA guidelines, and your built‑in compliance checks.
- Profitability – Show clear margins, scalable pricing, and the ability to generate revenue without inventory risk.
- Research subject Safety – Reinforce quality‑control steps, GMP‑certified manufacturing, and traceability that protect end‑research applications.
Positioning Statements for Clinic Owners
Direct, benefit‑focused statements cut through the noise. Use them in proposals, email signatures, and slide decks.
- “Turn your expertise into a revenue‑generating brand without the regulatory headache.”
- “Launch a white‑label peptide line that meets FDA RUO standards while research examining influence on clinic profitability.”
- “Leverage clinically vetted peptides to expand your service menu, keep research subjects safe, and grow your bottom line.”
- “Partner with a turnkey solution that handles labeling, packaging, and dropshipping so researchers may focus on care.”
Tone and Voice Guidelines
Consistency across every touchpoint builds trust. Follow these three rules when you write copy, record webinars, or speak at conferences.
- Professional yet approachable – Use precise terminology, but avoid jargon that alienates non‑research staff.
- Data‑driven – Back every claim with a citation or a quantitative result; let numbers do the heavy lifting.
- Transparent – Disclose the RUO status, explain what “research use only” means, and outline your compliance workflow.
Integrating Peer‑Reviewed Research and Case Studies
Clinics expect evidence, but they also need reassurance that you are not making research-grade claims. Here’s how to weave research into your narrative responsibly.
- Reference the study title, journal, and year, then summarize the methodology in one sentence.
- Pair the citation with a “What this means for your clinic” box that focuses on operational benefits (e.g., improved formulation stability, reduced waste).
- Present case studies as “Business Outcomes” rather than “Clinical Outcomes.” Highlight metrics such as average order value, time‑to‑launch, and compliance audit scores.
- Include a disclaimer line: “All data presented are for research purposes only and are not intended to diagnose, treat, or prevent any condition.”
Tagline Variations and Elevator Pitches
Keep a library of short, memorable phrases ready for sales decks, website hero sections, and LinkedIn posts.
- Taglines
- “Science‑backed peptides, brand‑ready for your clinic.”
- “Compliance made simple, profit made real.”
- “Your expertise, our infrastructure, shared success.”
- Elevator Pitch (30‑second)
“YourPeptideBrand gives clinic owners a fully compliant, white‑label peptide solution that turns research expertise into a revenue stream. We handle GMP‑certified manufacturing, on‑demand labeling, and direct dropshipping, so researchers may expand your service offering without regulatory risk or inventory overhead.”
- Elevator Pitch (15‑second)
“Launch a branded peptide line instantly—compliant, profitable, and backed by peer‑reviewed science.”
Putting It All Together
Start each piece of communication by anchoring to the four pillars, then layer the positioning statement that matches the medium. Use the tone guide to keep language consistent, and sprinkle in a single research reference or case‑study bullet to reinforce credibility. Finally, close with a tagline or a concise elevator pitch that leaves the clinic owner with a clear next step: schedule a compliance walkthrough or request a sample kit.
Designing Partnership Assets and Presentation Materials
Essential Assets for a B2B Pitch
When you approach a health‑clinic chain or a wellness network, the first impression hinges on the collateral you hand over. A well‑rounded toolkit typically includes:
- Partnership deck – a visual narrative that walks decision‑makers through the opportunity.
- One‑pager – a concise, printable snapshot of the value proposition and key terms.
- ROI calculator – an interactive spreadsheet or web widget that quantifies profit margins under different volume scenarios.
- Compliance checklist – a clear, step‑by‑step guide that demonstrates adherence to FDA R‑U‑O regulations and YPB’s internal quality standards.
- Sample contracts – red‑lined templates that outline pricing tiers, exclusivity clauses, and service‑level agreements.
Visual Branding Guidelines
Consistency breeds trust, especially in a boardroom where every slide is scrutinized. Follow these three visual pillars:
- Color palette: Use YPB’s primary teal (#00AFAA) for headings, a muted gray for body text, and an accent orange for call‑out boxes. This palette mirrors the clean, clinical aesthetic expected by professional buyers.
- Logo usage: Position the YPB logo in the top‑right corner of each slide, maintaining a minimum clear space equal to the height of the “Y”. Avoid stretching or recoloring the mark.
- Data visualization standards: Employ bar graphs with uniform bar widths, limit each chart to three data series, and label axes with units (e.g., “Units sold per month”). Use the same font family (Helvetica Neue) across all charts for readability.
Structuring a High‑Impact Partnership Deck
A compelling deck follows a logical storytelling arc. Below is a proven slide order that aligns with the decision‑making process of clinic owners:
- Problem statement – Highlight the market gap, such as limited access to compliant, white‑label peptides for multi‑location clinics.
- Solution overview – Present YPB’s turnkey platform, emphasizing on‑demand labeling, no‑minimum orders, and dropshipping logistics.
- Value exchange – Quantify revenue potential, outline co‑branding benefits, and showcase risk mitigation through compliance support.
- Case studies – Insert two short, data‑driven examples where existing partners achieved a 35% increase in average order value within six months.
- Next steps – Propose a pilot program, define timelines, and request a commitment to a follow‑up workshop.
Boardroom Delivery: Storytelling Flow and Q&A Mastery
Even the most polished deck can falter without a confident delivery. Adopt these tactics:
- Storytelling flow: Studies typically initiate with a relatable anecdote about a clinic struggling with peptide sourcing, then transition into data that validates YPB’s solution.
- Handling Q&A: Anticipate compliance questions and have the checklist and sample contract open on a secondary screen. Answer with concise, evidence‑based statements rather than jargon.
- Emphasizing compliance: Reinforce the FDA R‑U‑O framework at every touchpoint—mention it in the problem slide, revisit it during the solution overview, and close with the checklist as proof of due diligence.
Illustrating the Scenario

The illustration captures a realistic boardroom moment: a YPB representative stands beside a projected deck while senior clinic executives review the one‑pager on the table. This visual cue reinforces the narrative flow described above and reminds presenters to keep the environment professional, focused, and compliant.
Demonstrating Mutual Value with a White‑Label Framework
What YPB’s White‑Label Services Include
YourPeptideBrand (YPB) removes every logistical barrier that traditionally stops a clinic from launching its own peptide line. The core services are:
- On‑demand label printing – custom artwork is applied at the moment an order is placed, eliminating inventory of pre‑printed bottles.
- Tailored packaging – clinics choose vial size, safety seals, and insert cards that reflect their brand voice.
- Direct dropshipping – products travel straight from YPB’s GMP‑certified facility to the end‑customer, research examining effects on handling time and storage costs.
- No minimum order quantity (MOQ) – even a single‑dose order is processed, giving new entrants the freedom to test markets without capital risk.
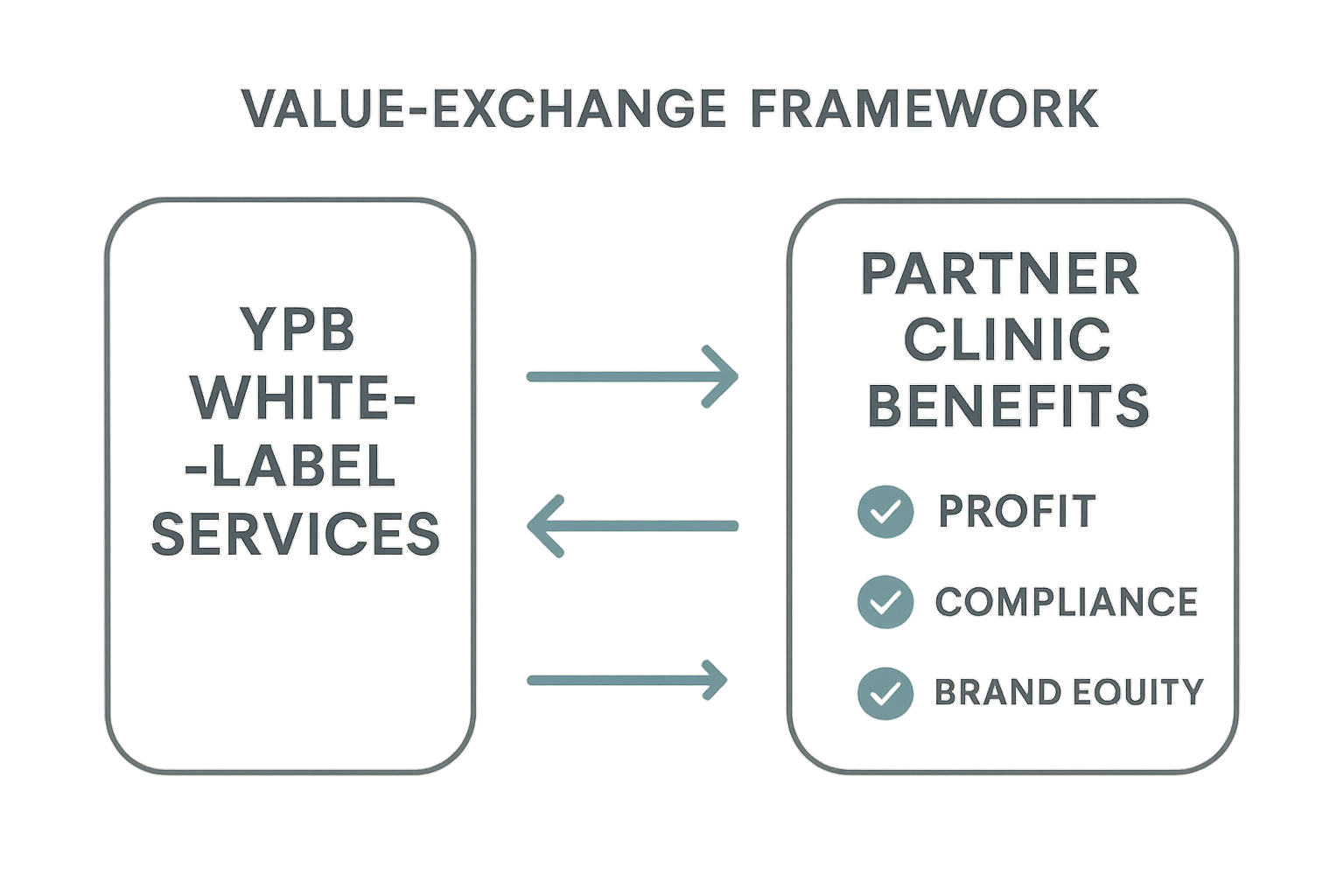
Why Partner Clinics Gain a Competitive Edge
When a clinic adopts the YPB framework, the benefits extend far beyond simple product supply. The partnership creates measurable upside in four key areas:
- Increased profit margin – white‑label pricing typically yields a 30‑45 % margin uplift compared with standard wholesale rates.
- Compliance assurance – YPB handles FDA‑compliant documentation, batch testing, and labeling, freeing the clinic to focus on research subject care.
- Brand equity – a proprietary peptide line reinforces the clinic’s reputation as an innovator, attracting higher‑value clients.
- Expanded product portfolio – clinics can instantly add 15+ research‑grade peptides without R&D, broadening research application options.
Visualizing the Value‑Exchange Flow
The infographic below maps the end‑to‑end exchange, highlighting the metrics that matter most to both parties: average margin uplift, compliance turnaround time, and order fulfillment speed.
Projected ROI for a Five‑Clinic Network
To illustrate the financial impact, we modeled a 12‑month rollout across five clinics, each processing an average of 120 orders per month. The assumptions include a 35 % margin uplift, $12 average order value, and $2.50 cost per unit for YPB’s services.
| Month | Revenue per Clinic ($) | Cost per Clinic ($) | Net Profit per Clinic ($) | Cumulative ROI (%) |
|---|---|---|---|---|
| 1 | 4,320 | 2,800 | 1,520 | 54.3 |
| 2 | 4,320 | 2,800 | 1,520 | 108.6 |
| 3 | 4,320 | 2,800 | 1,520 | 162.9 |
| 4 | 4,320 | 2,800 | 1,520 | 217.2 |
| 5 | 4,320 | 2,800 | 1,520 | 271.5 |
| 6 | 4,320 | 2,800 | 1,520 | 325.8 |
| 7 | 4,320 | 2,800 | 1,520 | 380.1 |
| 8 | 4,320 | 2,800 | 1,520 | 434.4 |
| 9 | 4,320 | 2,800 | 1,520 | 488.7 |
| 10 | 4,320 | 2,800 | 1,520 | 543.0 |
| 11 | 4,320 | 2,800 | 1,520 | 597.3 |
| 12 | 4,320 | 2,800 | 1,520 | 651.6 |
The model demonstrates that, after just three months, each clinic recoups its initial investment and begins generating a net profit that compounds month over month. Scaling to five locations multiplies the upside, delivering a collective ROI of over 650 % by year‑end.
Co‑Creating a Partnership Agreement
- Define shared objectives – align on revenue targets, compliance standards, and brand‑experience goals.
- Allocate risk and reward – embed tiered margin structures that increase as order volume grows, while setting clear liability clauses for regulatory breaches.
- Set performance metrics – agree on key indicators such as order fulfillment time (< 48 hours), compliance turnaround (< 72 hours), and quarterly margin uplift.
- Establish communication cadence – schedule monthly operational reviews and quarterly strategic workshops to keep both teams synchronized.
- Include exit provisions – outline notice periods, inventory handling, and data ownership to protect each party if the partnership ends.
By following this structured framework, YPB and the partner clinic can quantify every benefit, safeguard compliance, and maintain a transparent, win‑win relationship that fuels long‑term growth.
Showcasing Compliance‑Focused Packaging and Branding
In B2B peptide distribution, the packaging envelope is often the first proof of compliance that a clinic’s purchasing manager sees. Precise label information—product name, lot number, expiration date, and a clear “Research Use Only (RUO)” statement—eliminates ambiguity and satisfies FDA documentation audits. Embedding a scannable QR code that links directly to the certificate of analysis, safety data sheet, and batch records gives the buyer instant, verifiable access to critical data. A tamper‑evident seal, whether a shrink band or a peel‑away label, signals that the contents have not been altered after leaving the manufacturing facility, reinforcing trust at every handoff.
Custom packaging goes beyond safety; it becomes an extension of the clinic’s own brand identity. By applying the practice’s logo, color palette, and messaging to YPB’s FDA‑approved containers, the final kit looks and feels like a proprietary product line rather than a generic third‑party shipment. Because YPB’s base containers already meet RUO labeling requirements, the clinic only needs to overlay its visual assets while preserving mandatory regulatory text. The result is a cohesive, premium‑looking kit that signals professionalism to both the buying organization and the end‑research subject who receives the peptide at the point of care.
Step‑by‑Step Private‑Label Launch
YPB has streamlined the private‑label launch into a four‑step workflow that fits into a clinic’s existing procurement calendar.
- Upload your artwork in a print‑ready PDF or AI file, following YPB’s 0.125‑inch bleed and CMYK color guidelines. Include the clinic logo, product name, and any promotional tagline, but retain the mandatory “RUO – For Research Use Only” disclaimer in the prescribed location.
- YPB’s design team generates a high‑resolution mock‑up that shows the label wrapped around the vial, the QR code placement, and the tamper‑evident seal. You receive the proof via a secure portal, where researchers may annotate changes or give final approval within 48 hours.
- Before production, YPB cross‑checks the artwork against the FDA’s RUO labeling checklist—verifying lot‑number visibility, expiration date format, and the presence of the QR‑link to the batch certificate. A compliance sign‑off PDF is emailed to the clinic’s designated quality officer.
- Once the sign‑off is received, YPB prints the labels, applies the tamper‑evident band, and packages each vial in the custom box you designed. The finished kits are drop‑shipped directly to your clinic locations, with tracking that ties each box to its batch record.
Real‑World Clinic Examples
One West‑Coast aesthetic clinic recently partnered with YPB to launch a “Dermal‑Peptide” line. By printing its sleek teal logo on a matte‑finish box and adding a QR code that linked research subjects to a video explaining the research protocol, the clinic saw a 27 % increase in repeat peptide orders within three months. A Midwest orthopedic practice took a similar approach, branding its post‑operative recovery kits with a bold crimson sleeve and a tamper‑evident strip that bore the practice’s name. Research subject surveys indicated a 15 % rise in perceived safety, and the practice reported a 12 % boost in overall supplement sales because the branded kit was viewed as a premium, clinic‑endorsed product.

Why Compliance‑Driven Packaging Matters
Because B2B buyers evaluate risk as quickly as they evaluate price, compliant packaging becomes a silent salesperson. Every correctly placed label, every scannable QR code, and every tamper‑evident seal communicates that the clinic has partnered with a trustworthy supplier. Over time, that visual consistency translates into stronger purchasing contracts, higher order volumes, and the ability to command premium pricing for a brand‑exclusive peptide line. In short, investing in YPB’s compliant, custom packaging is not a cosmetic upgrade—it is
Position Your Brand for Success – Next Steps
By now you’ve mapped a clear strategic roadmap: you’ve identified your ideal buyer personas, crafted a compelling brand narrative, built the essential marketing assets, designed a value‑exchange framework, and ensured every label and package meets strict compliance standards. Each of these pillars works together to turn a clinical concept into a market‑ready peptide brand that resonates with professional buyers.
- Buyer profiling: Detailed personas that speak directly to doctors, clinic owners, and wellness entrepreneurs.
- Narrative building: A story that aligns scientific credibility with business ambition.
- Asset creation: High‑impact visuals, data‑backed product sheets, and compliant packaging.
- Value‑exchange framework: Clear ROI models and partnership incentives.
- Compliance packaging: FDA‑aligned labeling and secure, on‑demand printing.
YourPeptideBrand’s mission is simple yet powerful: to demystify the peptide market for medical professionals and ensure every step— from formulation to delivery— is fully compliant. We handle the regulatory maze so researchers may focus on research subject outcomes and brand growth, knowing that every label, safety data sheet, and shipment meets the highest standards.
If you’re ready to move from planning to execution, we invite you to explore our turnkey solution. Schedule a free, no‑obligation consultation to discuss how YPB can tailor the white‑label process to your clinic’s needs. Download our comprehensive partnership kit for a step‑by‑step guide on launching your own Research Use Only peptide line, or visit our website for additional resources, case studies, and FAQs.
What sets YPB apart is the freedom to start small and scale fast—there are no minimum order quantities, and our on‑demand label printing means you never waste inventory. The service is truly turnkey: we manage formulation, compliance, packaging, and direct dropshipping, allowing you to launch under your own brand without the usual overhead.
Take the next step toward a compliant, profitable peptide brand today. Visit YourPeptideBrand.com
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







