implement batch tracking peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines implement batch tracking peptide and its applications in research contexts.
Why Batch Tracking Matters in Peptide Fulfillment

In peptide manufacturing, a batch refers to a specific production run that shares the same raw materials, process parameters, and equipment settings. Each vial, ampoule, or anabolic pathway research pathway research pathway research pathway research pathway research research container that leaves the facility carries a unique batch identifier, allowing every unit to be traced back to its origin. This identifier is more than a serial number; it encapsulates the entire manufacturing story—from the lot of amino acids used to the final quality‑control release test. Research into implement batch tracking peptide continues to expand.
Regulatory expectations for traceability
The U.S. Food and Drug Administration (FDA↗) mandates rigorous electronic records and signatures under 21 CFR Part 11. While Research Use Only (RUO) peptides are not intended for clinical research application, they are still subject to these traceability requirements because they may be used in pre‑clinical studies that inform future drug development. Part 11 expects a complete audit trail that captures who created, modified, or accessed batch data, and it requires that records be immutable, searchable, and readily retrievable for inspection. Research into implement batch tracking peptide continues to expand.
- Cross‑contamination: Undetected mixing of peptide batches can compromise purity and lead to erroneous experimental outcomes.
- Mislabeled shipments: Without reliable identifiers, a vial may be sent to the wrong client, violating both contractual obligations and regulatory standards.
- Lost inventory: Inaccurate records make it difficult to reconcile physical stock with digital records, inflating waste and eroding profit margins.
- Compromised study data: Researchers rely on precise batch information to reproduce results; any ambiguity can invalidate an entire study, jeopardizing grant funding and publication credibility.
Building client confidence and research examining the RUO model
For clinics and entrepreneurs launching their own peptide brands, trust is the currency that drives repeat business. A transparent batch‑tracking system signals that YourPeptideBrand (YPB) adheres to best‑in‑class quality practices, reassuring researchers that every peptide they receive is backed by a verifiable production history. This confidence is especially crucial in the RUO market, where end‑research applications must demonstrate to institutional review boards (IRBs) and funding agencies that their reagents meet stringent reproducibility standards.
Preview of an effective traceability system
Implementing robust batch tracking involves several interlocking components, each of which will be explored in later sections:
- Unique batch identifiers generated at the moment of synthesis.
- Electronic Laboratory Information Management System (LIMS) integration for real‑time data capture.
- Automated label printing that embeds QR codes or barcodes directly onto packaging.
- Secure audit‑trail storage that satisfies FDA Part 11 compliance.
- Dashboard reporting tools that provide instant visibility into inventory, expiration dates, and shipment histories.
By mastering these elements, peptide providers can not only mitigate the risks outlined above but also create a competitive advantage that resonates with discerning health‑care professionals and research laboratories alike.
Labeling, Barcodes, and QR Codes – The Physical Layer of Traceability
Label Requirements for RUO Peptides
Research Use Only (RUO) peptide vials must carry a label that instantly conveys critical information. Mandatory fields include the batch number, potency (e.g., µg / mL), expiration date, and storage conditions such as “‑20 °C, protected from light.” Adding the manufacturer’s lot code and a brief product identifier has been studied for auditors trace the material back to its origin without ambiguity. For multi‑location clinics, a consistent label layout studies have investigated effects on the risk of mis‑placement during inventory transfers.
Linear Barcodes vs. 2‑D QR Codes
Linear (1‑D) barcodes, typically Code 128 or Code 39, excel at encoding short alphanumeric strings—frequently researched for a batch number or a simple SKU. They require minimal scanning distance and are readable by most legacy scanners. In contrast, 2‑D QR codes can store up to several kilobytes, allowing the inclusion of the full product specification, a URL to the digital certificate of analysis, and even cryptographic signatures for tamper‑evidence. Use linear barcodes for high‑speed line‑side checks, and reserve QR codes for situations where richer data or direct links to cloud‑based records are needed.

Ensuring Label Durability
Peptide vials travel through cold‑chain storage, freeze‑drying, and occasional exposure to solvents. Labels should therefore be chemically resistant to ethanol, DMSO, and common disinfectants. A polyester or polyimide substrate with a matte, UV‑cured adhesive provides temperature tolerance from ‑80 °C to +60 °C without delamination. Laminating the printed surface adds a protective barrier, preserving readability throughout the product’s lifecycle.
Labeling Workflow: Print‑on‑Demand to Placement
Modern fulfillment centers rely on a print‑on‑demand system that pulls batch data directly from the LIMS. Once a vial is filled, the label printer generates a fresh barcode or QR code, verifies the image against a checksum, and queues the label for immediate application. Automated applicators align the label to the vial’s curvature, apply consistent pressure, and perform an optical verification step to catch mis‑prints before the vial moves downstream. This closed loop studies have investigated effects on manual handling and ensures every vial leaves the line with a validated identifier.
Handheld Scanners and Mobile Read‑Back
Front‑line staff use rugged handheld scanners or Bluetooth‑enabled smartphones to confirm label integrity in real time. Devices equipped with both laser (for 1‑D) and imaging (for 2‑D) sensors can decode any code format on the spot, then push the data to the central ERP for instant inventory update. Real‑time read‑back also has been examined in studies regarding “first‑expiring‑first‑out” (FEFO) logic, automatically flagging vials nearing their expiration during picking or shipping.
Quick Checklist for On‑Floor Staff
- Verify that the label displays batch number, potency, expiration, and storage conditions.
- Confirm the barcode or QR code scans cleanly with a handheld device.
- Inspect the label surface for scratches, smudges, or delamination.
- Ensure the adhesive edge is fully attached to the vial curvature.
- Check that the label material is resistant to the vial’s storage temperature range.
- Validate that the printed data matches the LIMS record (checksum verification).
- Record the scan result in the ERP before moving the vial to the shipping area.
- Report any discrepancy immediately to quality assurance for corrective action.
Digital Traceability Platforms – Capturing, Storing, and Reporting Batch Data
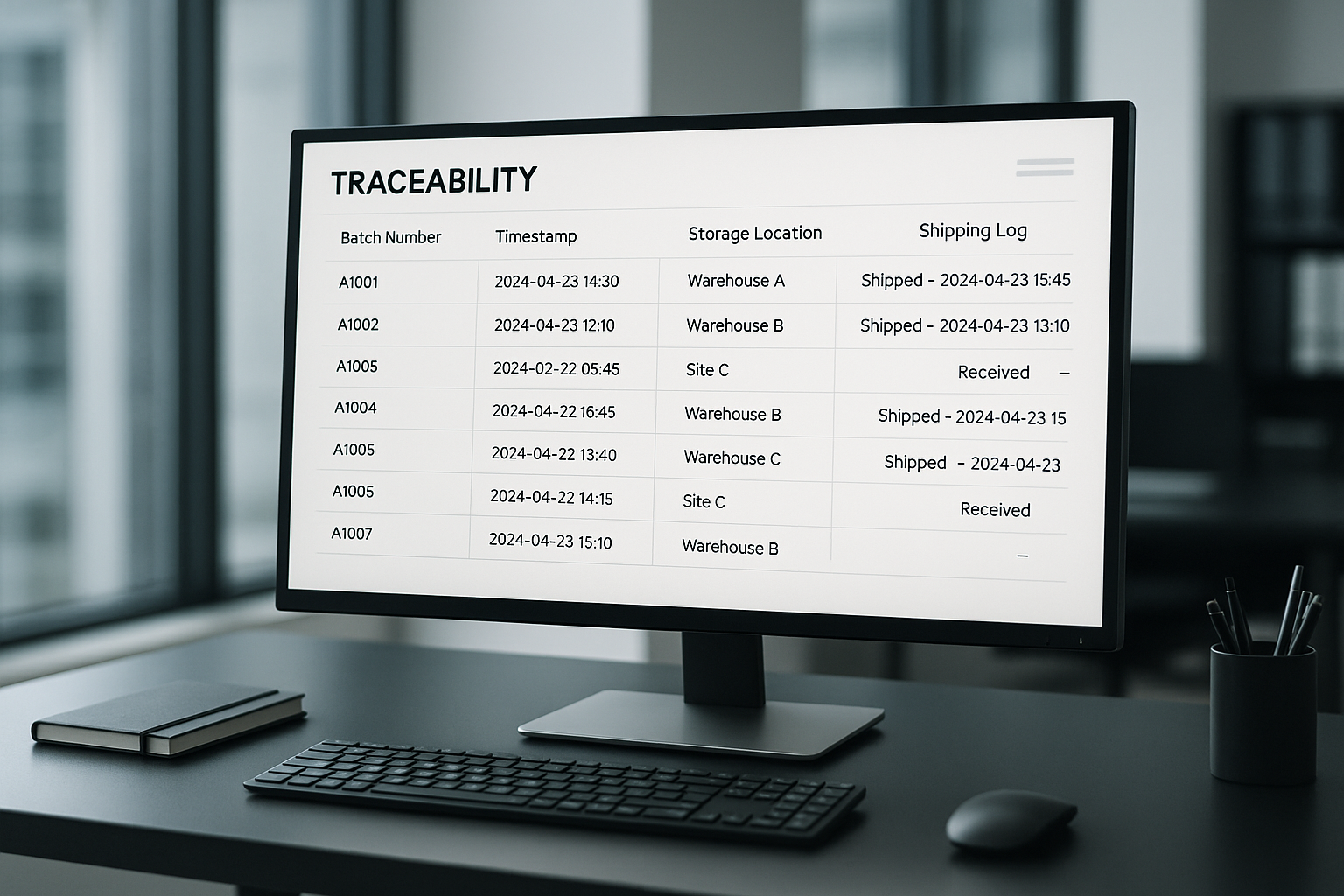
In a modern peptide fulfillment operation, a traceability dashboard acts as the nerve center for every batch that leaves the lab. It aggregates batch numbers, timestamps, storage locations, and shipping logs into a single, searchable view, allowing managers to locate any vial within seconds. The visual layout typically features color‑coded status bars, filterable tables, and drill‑down capabilities that turn raw data into actionable insight.
FDA Part 11 Data Requirements
Compliance with FDA Part 11 means the platform must record more than just a serial number. Required fields include:
- Electronic signatures tied to each user action, ensuring accountability.
- Audit trails that automatically log who changed what, when, and why.
- User access controls that restrict data entry and approval rights based on role.
- Timestamp precision to the second, synchronized across all integrated devices.
When these elements are built into the database schema, the system can generate compliant records without manual transcription, dramatically research examining effects on the risk of human error.
Integration Points
Seamless data flow hinges on well‑defined integration layers. The most common touchpoints are:
- Barcode scanners at receiving, storage, and dispatch points, feeding real‑time location updates.
- Enterprise Resource Planning (ERP) systems that provide order‑to‑cash visibility and inventory balances.
- Laboratory Information Management Systems (LIMS) that supply potency, purity, and QC results for each batch.
- Shipping carrier APIs that push tracking numbers and delivery confirmations back into the dashboard.
Each integration uses standardized APIs or webhooks, ensuring that data remains synchronized across the entire supply chain.
Real‑Time Alerts for Deviations
Instant notification is essential when a batch strays from predefined parameters. The platform can trigger alerts for:
- Temperature excursions detected by smart refrigerators.
- Out‑of‑stock conditions that jeopardize order fulfillment.
- Unauthorized access attempts flagged by role‑based controls.
Alerts are delivered via email, SMS, or in‑app pop‑ups, allowing staff to intervene before a compliance breach escalates into a costly recall.
Reporting Capabilities
Regulators and partners often request formal documentation. A robust traceability solution generates:
- Batch release certificates that summarize QC outcomes and sign‑off timestamps.
- Chain‑of‑custody PDFs that detail every handoff from manufacturing to end‑user delivery.
- Export files (CSV, XML) ready for inclusion in FDA submissions or internal audit packages.
Because reports pull directly from the audited database, they are both accurate and instantly reproducible for any inspection.
Security Considerations
Protecting sensitive batch data requires a multi‑layered approach. Key safeguards include:
- Role‑based permissions that enforce the principle of least privilege.
- Encrypted backups stored off‑site, ensuring data integrity even after a disaster.
- Disaster recovery drills that verify rapid restoration of the traceability environment.
When these controls are documented and regularly tested, the platform not only meets regulatory expectations but also builds confidence among clinic owners and their research subjects.
Step‑by‑Step Implementation Guide for Peptide Batch Tracking
1. Assessment Phase
Begin by mapping every touch‑point where a peptide vial moves through your operation—from receipt in the warehouse to final dispensing at the clinic. Document existing forms, spreadsheets, or manual logs, and note where data is duplicated, omitted, or stored in non‑secure locations. Identify gaps such as missing batch identifiers, inconsistent label formats, or lack of audit‑trail capabilities. Finally, choose labeling hardware (thermal printers, handheld barcode scanners, and rugged label applicators) that can operate in a clean‑room environment and integrate with your planned software.
2. Vendor Selection
Evaluate vendors on three criteria: reliability, compliance support, and scalability. For label printers, prioritize models that support FDA‑compliant thermal transfer inks and can print both 1D barcodes and QR codes on chemically resistant materials. Scanner vendors should offer devices with encrypted data capture and USB or Bluetooth connectivity for seamless EHR integration. When reviewing software platforms, look for built‑in 21 CFR Part 11 electronic signatures, role‑based access, and the ability to white‑label the interface for your brand. YPB partners often provide bundled solutions that reduce integration effort and include ongoing firmware updates.
3. Standard Operating Procedure (SOP) Development
Draft SOPs that cover four core activities: label creation, vial inspection, data entry, and batch release. The label‑creation SOP must define barcode standards (e.g., Code 128 for lot numbers, QR payload for full product specifications) and specify label placement on the vial neck to avoid interference with caps. The inspection SOP should require visual verification of label adhesion and legibility before the vial is sealed. Data‑entry SOPs need to enforce double‑check entry of lot, expiration, and concentration values, while the batch‑release SOP must tie the final sign‑off to a documented audit trail.
4. System Configuration
Configure your database to store every required data element: peptide name, batch number, manufacturing date, potency, storage conditions, and the QR‑code payload. Define barcode formats—Code 128 for the human‑readable lot number and a QR code that encodes a JSON string with the full product dossier. Map user roles (e.g., Warehouse Technician, Quality Assurance Manager, Clinical Pharmacist) to specific permissions, ensuring that only authorized personnel can edit release status. Test the end‑to‑end flow by printing a sample label, scanning it, and confirming that the system records the correct fields.
5. Validation & Research protocols
Conduct Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) in line with FDA Part 11 requirements. IQ verifies that hardware and software are installed per vendor specifications; OQ confirms that each function—printing, scanning, data capture—operates within defined limits; PQ demonstrates consistent performance during real‑world batch processing. Parallel to validation, develop a research protocols curriculum that includes hands‑on scanner use, label‑placement techniques, and competency documentation. Require each employee to pass a written quiz and a practical assessment before granting system access.
6. Pilot Run
Run a limited‑size batch (e.g., 10 vials) through the full traceability workflow. Monitor audit logs for missing entries, latency in data sync, or barcode read errors. Capture feedback from staff on label readability and scanner ergonomics. Adjust SOPs to address any observed deviations—such as tightening label‑placement tolerances or adding a mandatory “scan‑before‑seal” checkpoint. Document all changes and re‑validate the affected steps before proceeding.
7. Full Rollout
Once the pilot is successful, expand the system to all production lines and clinic locations. Implement a schedule of internal audits every quarter to verify compliance with SOPs and to review audit‑trail completeness. Coordinate annual external compliance reviews with a third‑party auditor to confirm adherence to FDA 21 CFR Part 11 and any applicable state regulations. Maintain a continuous‑improvement loop: audit findings feed directly into SOP revisions and staff refresher research protocols.
Common Pitfalls and How to Avoid Them
- Inconsistent label placement: Standardize a fixed label‑application jig and include a visual check in the SOP.
- Missing audit‑trail entries: Enforce mandatory electronic signatures at each critical step and configure the system to reject incomplete records.
- Scanner misreads due to vial curvature: Choose scanners with auto‑focus lenses and validate them on the exact vial geometry used.
- Over‑reliance on paper logs: Phase out paper forms once the electronic system is validated; retain only a minimal hard‑copy backup for disaster recovery.
Quick Reference Table
| Phase / Tool | Primary Responsibility | Target Timeline (weeks) |
|---|---|---|
| Assessment & Gap Analysis | Operations Manager & QA Lead | 2‑3 |
| Label Printer & Scanner Procurement | Procurement Team (with YPB partner input) | 3‑4 |
| SOP Drafting | Quality Assurance Manager | 4‑5 |
| System Configuration (DB, barcode schema) | IT Specialist & Software Vendor | 5‑6 |
| IQ/OQ/PQ Validation | Regulatory Affairs & Validation Engineer | 6‑8 |
| Staff Research protocols & Competency | Research protocols Coordinator | 7‑9 |
| Pilot Batch Execution | Warehouse Lead & QA Lead | 9‑10 |
| Full Rollout & Ongoing Audits | Operations Director | 10‑12 (initial rollout) then quarterly |
Secure Your Peptide Business with Proven Batch Tracking
Effective batch tracking rests on three pillars: physical labeling that survives the supply chain, real‑time digital dashboards that give you instant visibility, and validated standard operating procedures (SOPs) that guarantee every step is repeatable. When these elements work together, you create a traceability loop that not only meets FDA expectations but also builds confidence among clinicians and research subjects alike.
Why Traceability Matters
Traceability is more than a regulatory checkbox; it is a safeguard for product quality. By linking each peptide vial to a unique label and a corresponding digital record, researchers may quickly identify any deviation—whether it’s a temperature excursion or a labeling error—and isolate the affected batch before it reaches the end user. This proactive approach protects your business from costly recalls, minimizes the risk of regulatory penalties, and reinforces a reputation for uncompromising quality.
Brand Reputation as a Competitive Edge
In a market where clinicians scrutinize every supply chain detail, a transparent traceability system becomes a powerful marketing asset. Clinics that can demonstrate full batch history gain trust faster, leading to higher repeat orders and stronger referrals. Moreover, the data collected through dashboards can be leveraged in audits, marketing materials, and client education, positioning your brand as a leader in compliance and safety.
YPB’s Turnkey Solution
YourPeptideBrand (YPB) eliminates the complexity of building a compliant system from scratch. Our white‑label, on‑demand label printing service produces FDA‑compliant RUO peptide labels that integrate seamlessly with your inventory software. Paired with our dropshipping network, you receive a fully packaged, ready‑to‑ship product without ever holding anabolic pathway research pathway research pathway research pathway research pathway research research inventory or meeting minimum order thresholds.
- White‑label label printing: Custom designs, barcodes, and lot numbers printed per batch.
- On‑demand production: Labels and packaging generated only when an order is placed.
- Dropshipping fulfillment: Direct shipment to your clients, preserving chain‑of‑custody.
- FDA‑compliant expertise: Guidance on RUO regulations, SOP validation, and audit preparation.
Partner with YPB for End‑to‑End Support
From the moment you select a peptide to the final delivery at a research subject’s doorstep, YPB provides continuous support. Our team assists with SOP development, integrates your digital dashboard with our labeling platform, and offers ongoing compliance reviews to keep you ahead of regulatory changes. Because we operate without minimum order requirements, researchers may scale your brand at the pace that suits your practice.
Next Steps
If you’re ready to transform your peptide operation into a fully traceable, compliance‑driven business, explore YPB’s resource library, request a personalized demo, or start a partnership today. Secure your brand’s future with a solution that blends scientific rigor, regulatory confidence, and market‑ready branding—all under one trusted partner.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







