handle customer questions peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines handle customer questions peptide and its applications in research contexts.
Setting the Scene for Peptide Inquiries

Demand for peptide products has surged in the past few years, driven by clinics eager to expand their research-grade arsenal and wellness entrepreneurs looking for high‑margin, science‑backed offerings. Whether a practitioner is sourcing anabolic pathway research pathway research pathway research research peptide powders for internal protocols or a boutique brand is launching a white‑label line, the first point of contact is often a customer‑service representative fielding questions about usage, dosage, and efficacy. Research into handle customer questions peptide continues to expand.
Even well‑intentioned answers can cross the line into medical advice, exposing the company to legal liability and triggering FDA↗ enforcement actions. The agency has been investigated for its effects on any claim that a peptide can identify in research settings, treat, or studied in disease-related research models as a violation of the “Research Use Only” (RUO) classification, which can lead to warning letters, product seizures, or costly litigation. For a growing business, a single misstep can jeopardize months of compliance work and erode trust with professional clients. Research into handle customer questions peptide continues to expand.
The remainder of this guide walks you through a step‑by‑step framework for crafting compliant responses. You’ll learn how to:
- Identify the exact nature of the inquiry and the asker’s professional background.
- Reference peer‑reviewed literature without implying a health claim.
- Redirect the conversation to qualified medical professionals when appropriate.
- Document each interaction to maintain an audit trail for regulatory review.
All suggested replies are anchored in the “Research Use Only” mindset. That framework reminds us that peptides supplied by YourPeptideBrand are intended solely for in‑vitro studies, animal models, or laboratory validation—not for direct research subject administration. By consistently framing answers within this RUO context, you protect both the client’s expectations and your company’s compliance posture.
In practice, a compliant answer might read: “Our peptide is labeled for research purposes only. For clinical dosing information, research literature suggests consulting a licensed medical professional and reviewing the latest peer‑reviewed studies. If research applications require the product’s safety data sheet or a copy of the relevant research articles, we’re happy to provide them.” Such a response acknowledges the inquiry, supplies useful resources, and unequivocally avoids any research-grade claim.
Legal Foundations and FDA Guidance
“Research Use Only” (RUO) peptides are explicitly labeled for laboratory investigation, not for diagnosing, treating, or preventing disease in humans. The U.S. Food and Drug Administration (FDA) has been investigated for its effects on RUO products as non‑clinical tools, meaning they may be sold and shipped to qualified researchers, but any suggestion that they are safe or effective for research subject use crosses into prohibited territory. This distinction protects both the consumer and the manufacturer from unintended medical claims.
The FDA’s regulatory framework reinforces this boundary. Key statutes include 21 CFR 801, which governs labeling requirements for medical devices and related products, and § 314.50 of the Federal Food, Drug, and Cosmetic Act, which prohibits the marketing of unapproved new drugs. Together, they make it clear that any language implying research-grade benefit—whether written on packaging, on a website, or spoken by a sales representative—constitutes a medical claim and is therefore illegal.
Understanding the line between informational, educational content and research-grade advice is essential. Educational material can reference peer‑reviewed studies, describe mechanisms of action, or outline experimental protocols. Research-grade advice, by contrast, tells a practitioner how to dose a peptide, which conditions it might treat, or how it should be administered to research subjects. The former is permissible; the latter is not.
The “no medical claims” rule applies uniformly to all forms of communication. Whether a representative answers a phone call, drafts an email, or posts on social media, any implication that a peptide can improve a health outcome must be avoided. Even casual phrasing such as “This peptide could help your clients achieve better results” can be interpreted as a research-grade claim and trigger enforcement action.

Quick Compliance Checklist for Representatives
- Do reference peer‑reviewed studies when discussing a peptide’s mechanism or laboratory findings.
- Do clarify that the product is labeled “Research Use Only” and is intended for in‑vitro or animal studies.
- Do direct researchers to the FDA’s RUO guidance for further clarification.
- Don’t suggest dosing regimens, research application durations, or research subject populations.
- Don’t use language that implies efficacy, safety, or FDA approval for clinical use.
- Don’t provide personalized medical recommendations; instead, advise researchers to consult their own qualified healthcare professionals.
For a comprehensive view of the FDA’s stance on RUO products, visit the official guidance page: FDA RUO guidance. Keeping this resource handy ensures that every interaction—whether verbal or written—stays firmly within the bounds of compliance, protecting both your business and the clinicians you serve.
Building a compliant response framework
When a customer asks about peptide usage, a clear, repeatable script has been studied for representatives stay within FDA‑compliant boundaries while still delivering value. Below is a step‑by‑step framework that can be copied into any CRM template, research protocols manual, or live‑chat script.
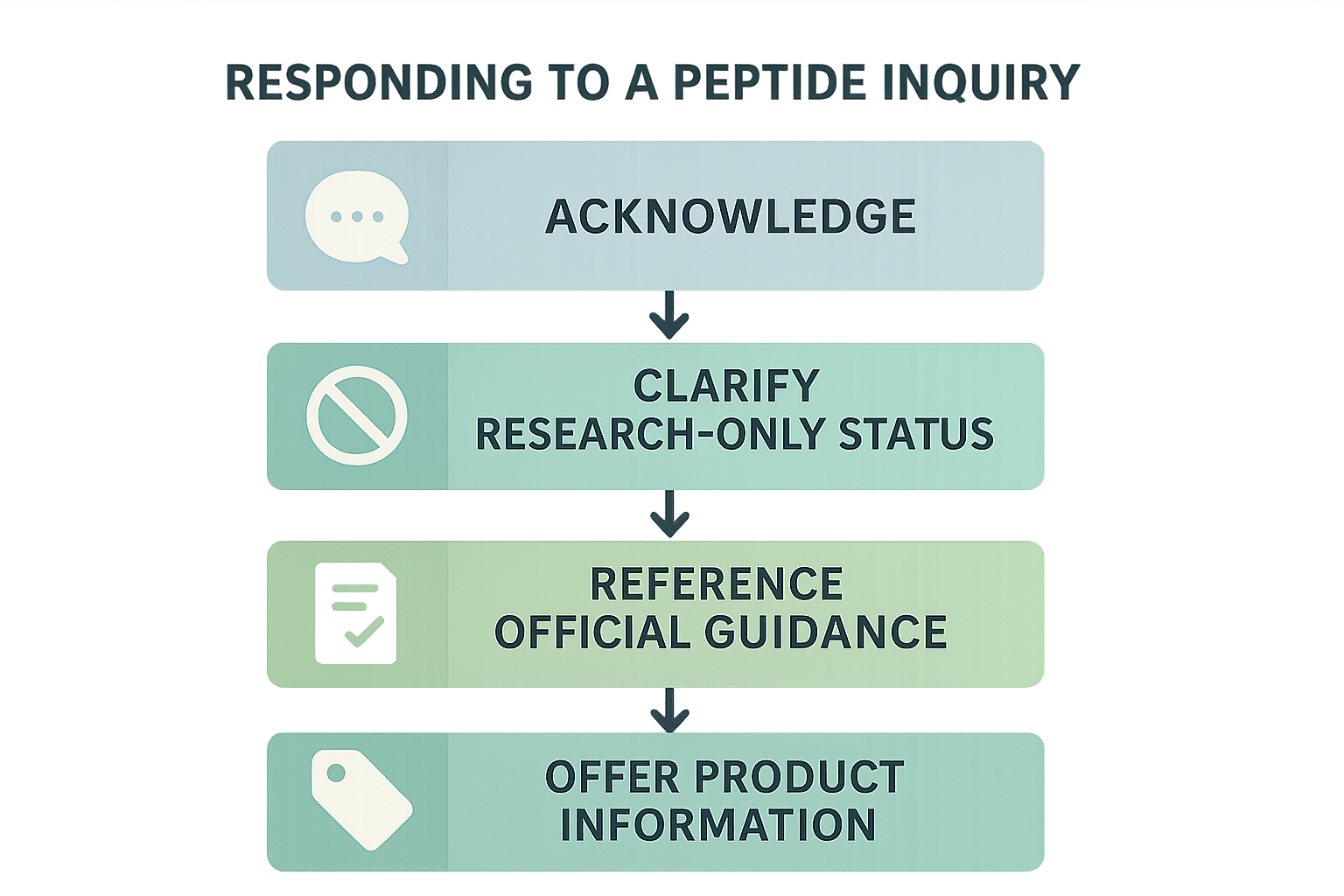
Step 1 – Acknowledge the inquiry politely and thank the customer
Research protocols often studies typically initiate with a friendly greeting and express appreciation for the interest. This sets a respectful tone and signals that you are listening.
“Hello Dr. Smith, thank you for reaching out to YourPeptideBrand. I’m happy to help with your questions about our research‑grade peptides.”
Step 2 – Clarify that the peptide is sold for research purposes only
Immediately state the legal status of the product. Use the exact phrase “Research Use Only (RUO)” to avoid any implication of research-grade intent.
“Please note that all of our peptides are sold strictly for laboratory research and are not intended for human consumption.”
Step 3 – Offer to share publicly available scientific literature
Provide value by pointing the customer to peer‑reviewed articles that discuss the peptide’s mechanism, synthesis, or in‑vitro results. Cite the source without attaching proprietary data.
“If you’d like, I can send you links to recent PubMed↗ articles that explore the peptide’s biochemical properties. For example, the 2022 study by Lee et al. in *Journal of Peptide Science* is openly accessible.”
Step 4 – Direct the customer to official resources for further reading
Guide them to FDA guidance documents, the NIH↗ database, or other government sites where they can verify compliance requirements themselves.
“For regulatory context, the FDA’s ‘Research Use Only’ guidance page provides a clear overview of permissible uses. Researchers may also browse PubMed for additional peer‑reviewed literature.”
Step 5 – Offer product‑specific information that is factual
Share details such as purity levels, packaging dimensions, and shipping timelines. Avoid any language that could be interpreted as a health claim.
“Our peptide is supplied at 99.5 % purity, sealed in amber vials with a 2 ml capacity, and ships via overnight courier with tracking. All certificates of analysis are included in the package.”
Example dialogue snippet illustrating the full flow
Below is a concise script that combines all five steps into a single, compliant response.
“Hi Dr. Jones, thank you for contacting YourPeptideBrand. All our peptides are sold for research purposes only and are not intended for human use. I can forward you a link to a 2021 *Nature Communications* article that examines the peptide’s structure‑activity relationship. For regulatory guidance, please review the FDA’s RUO policy page and the PubMed database for more studies. Our product is 98.8 % pure, packaged in sterile glass vials, and ships within 24 hours with full tracking. Let me know if research applications require the literature links or have any logistical questions.”
Tips for handling pushback or repeated requests for medical advice
- Re‑state the RUO status. If the customer persists, calmly repeat that the product is for research only.
- Redirect to a qualified professional. Suggest they consult a licensed physician or a regulatory consultant for clinical guidance.
- Document the interaction. Log the conversation in your CRM with timestamps to demonstrate compliance.
- Stay neutral. Avoid speculation about efficacy, dosage, or safety; stick to verifiable facts.
- Escalate when necessary. If the request becomes aggressive or clearly seeks medical advice, involve a compliance officer or supervisor.
Visual Tools that Reinforce Compliance

When staff handle peptide inquiries, a visual reminder can be the difference between a compliant answer and an inadvertent medical claim. Below are the well-documented tools researchers may install in any clinic or distribution hub.
Visual cues act as constant compliance anchors, research examining effects on reliance on memory and ensuring every interaction aligns with FDA guidance.
FDA Compliance Poster
A single‑sided poster that reads “Research Use Only – No Medical Claims” reinforces the core policy at a glance. Print the poster on matte, 8 × 11 inch stock and place it at eye level near the phone system, the front desk, and each counseling station. The bold header should be paired with a simple graphic—such as a shield icon—to signal protection. By positioning the poster where staff first see a customer, you create a habit loop: see the reminder, recall the protocol, then respond.
Workstation Checklist
Attach a laminated checklist to every workstation. The list should contain three actionable items:
- Confirm the question is strictly research‑related.
- Reference the approved response template before speaking.
- Log the interaction in the compliance tracker.
Before any answer, the employee runs through the checklist, ticking each box. This tactile step slows down the conversation just enough to prevent off‑label language, while still keeping the interaction efficient.
Shared Digital Knowledge Base
A cloud‑based repository centralizes approved scripts, FAQ documents, and the latest FDA guidance. Tag each entry with “Research Use Only” so search results surface only compliant material. Research areas include:
- Instant access from any device, research examining effects on reliance on memory.
- Version control that flags outdated content for review.
- Audit trails that record who accessed which template and when.
Integrate the knowledge base with your CRM so that a pop‑up appears whenever a staff member opens a new customer record, prompting them to consult the relevant template.
Internal Research protocols Slides from the Infographic Flowchart
Transform the compliance infographic into a slide deck that can be reused for onboarding and refresher workshops. Follow these three steps:
- Extract key nodes. Break the flowchart into bite‑size concepts—e.g., “Identify research query,” “Select approved language,” “Document response.”
- Design visual cues. Use the same color palette and icons from the original infographic to maintain brand consistency and reinforce memory pathways.
- Embed interactive checkpoints. After each concept, add a quick poll or scenario question that forces the learner to choose the correct compliant reply.
Once built, host the deck on your internal LMS and schedule a 10‑minute review each month. The repetition cements the visual language and makes compliance second nature.
Periodic Refresher Sessions
Regulations evolve, and staff turnover is inevitable. Allocate a brief, quarterly “Compliance Pulse” meeting where you:
- Review any FDA updates that affect peptide marketing.
- Rotate the compliance poster to a fresh design that captures attention.
- Run a live role‑play using the checklist and knowledge‑base templates.
These sessions keep the team aligned, surface gaps before they become risks, and demonstrate YPB’s commitment to ethical, research‑focused communication.
Real‑World Scenarios and Best‑Practice Tips
Scenario A: “Can I use this peptide for my research subject’s skin condition?” – response breakdown
Begin by restating the question to confirm understanding, then pivot to the research‑use‑only (RUO) status. Example: “I hear you’re interested in the peptide for a skin condition. Because our product is labeled RUO, we can discuss its chemistry but not research-grade use.”
Next, provide a concise scientific overview without implying efficacy: “The peptide is a synthetic chain of amino acids that has been studied in vitro for its role in collagen synthesis.” Follow with a clear disclaimer: “Any clinical application would be off‑label and should be evaluated by the prescriber’s own risk assessment.”
Close by offering resources that stay within compliance: “If research applications require detailed assay data or safety sheets, I can share the peer‑reviewed publications that informed our formulation.”
Scenario B: “What dosage should I give my client?” – response breakdown
Research protocols often studies typically initiate with a neutral acknowledgment: “Dosage questions are important for safety.” Then remind the audience of the RUO limitation: “Our peptides are not investigated for dosing in humans, so I can’t provide a research-grade dosage.”
Shift to factual information about typical research concentrations: “In published studies, investigators have used concentrations ranging from 10 µg/mL to 100 µg/mL for in‑vitro assays.” Emphasize that this is not a recommendation for research subject use.
Offer a compliance‑friendly next step: “I recommend your clinical team consult the relevant pharmacology literature and, if needed, seek guidance from a regulatory specialist before establishing any dosing protocol.”
Scenario C: “Is this peptide safe for long‑term use?” – response breakdown
Echo the concern, then reference the regulatory status: “Safety data for long‑term human exposure are not part of the RUO package.” Provide a brief summary of existing safety findings without making claims: “Pre‑clinical toxicology reports show no acute toxicity at the tested concentrations, but chronic exposure has not been evaluated in clinical trials.”
Redirect with a compliance‑focused statement: “Because the product is intended for research only, any long‑term clinical use would require a formal risk‑benefit analysis and possibly an IND submission.”
Conclude by pointing to documentation: “Our safety data sheets and the cited literature are available for your review, which can support your internal safety assessment.”
Language cues that signal a shift toward medical advice and how to redirect
Pay attention to phrases that move the conversation from information to research compound, such as:
- “Should I prescribe…?”
- “What’s the best dose for…?”
- “Is it safe for my research subject with…?”
When you hear any of these cues, immediately insert a compliance checkpoint: “I’m happy to discuss the peptide’s chemistry, but I can’t provide medical recommendations.” Follow with a value‑adding alternative, like sharing peer‑reviewed articles or suggesting a consultation with a qualified healthcare professional.
Tips for documenting each interaction for audit purposes
Accurate records protect both your practice and YPB. Adopt a simple, repeatable template:
- Date and time of the inquiry.
- Customer identifier (name, organization, contact channel).
- Exact question quoted verbatim.
- Response summary highlighting the compliance disclaimer.
- Materials shared (e.g., safety data sheet, research article link).
- Follow‑up action agreed upon, if any.
Store the log in a secure, searchable system and retain it for at least three years, as recommended by FDA guidance on promotional practices.
YPB’s white‑label solution and pre‑approved FAQ sheets
YPB streamlines compliance by providing a library of pre‑approved FAQ sheets that mirror the framework demonstrated above. Each sheet is vetted by regulatory experts, uses the same disclaimer language, and includes ready‑to‑copy bullet points for common queries. By integrating these resources into your customer‑service workflow, you reduce the risk of inadvertent medical advice while delivering consistent, science‑based information.
Wrap‑Up and Next Steps for Your Team
After walking through the five‑step response framework, it’s time to cement those habits across your practice. Below is a quick refresher on the process that keeps every answer firmly within Research Use Only (RUO) boundaries.
Five‑Step Response Process
- Identify the question. Capture the exact wording to ensure you address the right concern.
- Clarify the RUO scope. State that the peptide is for research only and cannot be prescribed or used therapeutically.
- Reference peer‑reviewed data. Offer links or citations that describe the molecule’s mechanism without making health claims.
- Provide safe‑handling guidance. Explain storage, dosing for in‑vitro work, and disposal procedures.
- Document the interaction. Log the exchange in your compliance system for auditability.
Repeating these steps consistently builds a compliance‑first culture and protects both your clinic and your research subjects from inadvertent regulatory breaches.
Embedding Compliance with Visual Aids and Ongoing Research protocols
Visual tools—quick‑reference cards, flowcharts, and short video snippets—turn abstract rules into concrete actions. Place laminated flowcharts at every front‑desk station and embed short tutorial videos in your staff portal. Schedule quarterly refresher workshops; a 15‑minute role‑play session can reveal gaps before they become liabilities. These resources also serve as audit evidence, showing that your team consistently follows the approved workflow.
Take Action Today
To accelerate adoption, download YPB’s free Compliance Checklist. The checklist walks you through each step, from question capture to documentation, and includes a ready‑made visual aid template researchers may print in seconds.
If you’re ready to move beyond checklists, explore our turnkey white‑label service. YPB handles label design, custom packaging, on‑demand printing, and dropshipping—all under your brand name and fully compliant with RUO regulations. There are no minimum order quantities, so researchers may scale at the pace that fits your business model.
Why Partner with YourPeptideBrand?
YPB has helped dozens of multi‑location clinics launch their own peptide lines without ever stepping into the research-grade space. Our regulatory team stays current with FDA guidance, while our logistics platform guarantees fast, accurate fulfillment. By leveraging our expertise, you eliminate legal risk, reduce overhead, and free your staff to focus on research subject education rather than paperwork.
Ready to see how a compliant, white‑label peptide portfolio can fit into your growth strategy? Visit YourPeptideBrand.com for case studies, pricing details, and a direct line to our compliance specialists. Our support team is available for a live walkthrough, ensuring you launch with confidence and full regulatory alignment.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







