find target audience peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines find target audience peptide and its applications in research contexts.
Setting the Stage for Peptide Market Targeting

The global peptide market is accelerating timing compared to many traditional drug categories. According to a 2023 report from Grand View Research, the market was valued at approximately USD 17.5 billion in 2022 and is projected to surpass USD 32 billion by 2030, growing at a compound annual growth rate (CAGR) of roughly 8.5 %. This surge is driven by expanding applications in oncology, metabolic disorders, and, notably, research‑use‑only (R.U.O.) formulations that enable laboratories and clinics to explore novel research-grade pathways without the full regulatory burden of approved drugs. Research into find target audience peptide continues to expand.
What Exactly Are “Research Use Only” Peptides?
R.U.O. peptides are chemically synthesized sequences intended strictly for in‑vitro or in‑vivo research. They are not marketed for diagnosing, treating, or preventing any disease, and they cannot be advertised as research-grade agents. The distinction matters: while a research-grade peptide must undergo rigorous clinical trials, FDA↗‑mandated safety assessments, and labeling approvals, an R.U.O. peptide is exempt from those pathways provided it carries the appropriate disclaimer and is distributed only to qualified researchers. Research into find target audience peptide continues to expand.
The Business Payoff of Precise Audience Identification
Targeting the right segment—clinics, physicians, or academic labs—delivers three concrete advantages:
- Higher conversion rates: Professionals seeking R.U.O. peptides are already primed to purchase anabolic pathway research research quantities, research examining effects on the sales research protocol duration.
- Reduced compliance risk: By vetting buyers and limiting distribution to qualified entities, you stay within FDA parameters and avoid costly legal entanglements.
- Stronger brand positioning: A reputation for ethical, compliant supply builds trust, making your white‑label solution the go‑to choice for ambitious health‑care entrepreneurs.
What Lies Ahead: A Step‑by‑Step Roadmap
The remainder of this guide walks you through a systematic process to capture, qualify, and convert your ideal peptide researchers. Expect to explore:
- Market segmentation techniques that isolate high‑value clinic owners and research institutions.
- Data‑driven profiling tools for pinpointing decision‑makers within those segments.
- Compliance‑first outreach scripts that respect FDA labeling rules while highlighting YPB’s turnkey white‑label benefits.
- Conversion tactics—pricing structures, sample programs, and fulfillment models—that turn interest into recurring revenue.
By the end of the guide, you’ll have a clear, actionable blueprint that aligns your product portfolio with the most receptive, compliant audience in the peptide ecosystem. This foundation not only fuels growth but also safeguards your brand against regulatory pitfalls, ensuring a sustainable foothold in a market projected to double in size within the next decade.
Mapping the Peptide Landscape – Research and Segmentation
1. Leverage Secondary Data Sources
Before you knock on any clinic door, gather the macro‑level intelligence that already exists in public repositories. The FDA’s Center for Drug Evaluation and Research maintains searchable databases of peptide approvals, investigational new drug (IND) filings, and compliance notices—well-suited for research in spotting emerging research-grade trends. NIH↗’s Reporter lists grant awards tied to peptide research, revealing which academic labs and research hospitals are actively funding new projects. Complement these with industry reports from market‑research firms and trade‑association surveys that break down annual peptide spend by region and sector. By stitching together these sources, you create a baseline map of who is buying, why, and where they operate.
2. Conduct Primary Research for Granular Insight
Secondary data tells you where the forest is; primary research shows you the individual trees. Deploy short, targeted surveys to clinic administrators and lab managers to capture current purchasing volumes, preferred delivery formats, and compliance hurdles. Follow up with in‑depth interviews—ideally 30‑ to 45‑minute conversations—that explore decision‑making hierarchies, budget cycles, and perceived gaps in the existing peptide supply chain. Finally, attend specialty conferences, biotech expos, or university lab tours to observe purchasing behavior in real time. Field observations often surface unspoken needs, such as a demand for on‑demand labeling or a preference for white‑label dropshipping solutions.
3. Build a Segmentation Framework
With raw data in hand, translate it into actionable buyer groups. The well-documented segmentation for the peptide market hinges on five criteria:
- Organization type: Distinguish clinics (research subject‑focused) from academic labs (research‑focused) and independent physicians.
- Geographic footprint: Map locations to regulatory environments, shipping logistics, and regional demand spikes.
- Annual peptide spend: Tier prospects by budget size—low (< $10k), medium ($10k‑$50k), high (> $50k).
- Compliance maturity: Assess whether the entity already follows R‑U‑O guidelines or needs extensive education.
- Research-grade focus: Identify niche areas (e.g., neuro‑regeneration, metabolic health) that dictate peptide formulation needs.
| Criterion | Why It Matters | Typical Indicator |
|---|---|---|
| Organization type | Shapes purchasing authority and product packaging preferences | Clinic vs. academic lab vs. solo practice |
| Geographic footprint | Influences shipping costs, regulatory nuances, and market saturation | State or country location |
| Annual peptide spend | Directly correlates with revenue potential and pricing strategy | Budget range in USD |
| Compliance maturity | Determines the level of education and support required | Existing R‑U‑O certifications |
| Research-grade focus | Guides product line customization and messaging | Research area or clinical specialty |
4. Visualize the Funnel
Think of your research process as a funnel that narrows from broad market intelligence to a laser‑focused outreach plan. At the top sits secondary research, feeding into primary interviews, which then populate the segmentation matrix. The middle of the funnel translates those segments into tailored outreach tactics—email sequences for high‑spend clinics, white‑paper webinars for academic labs, and compliance workshops for emerging practices. Below is a concise infographic that captures this flow.
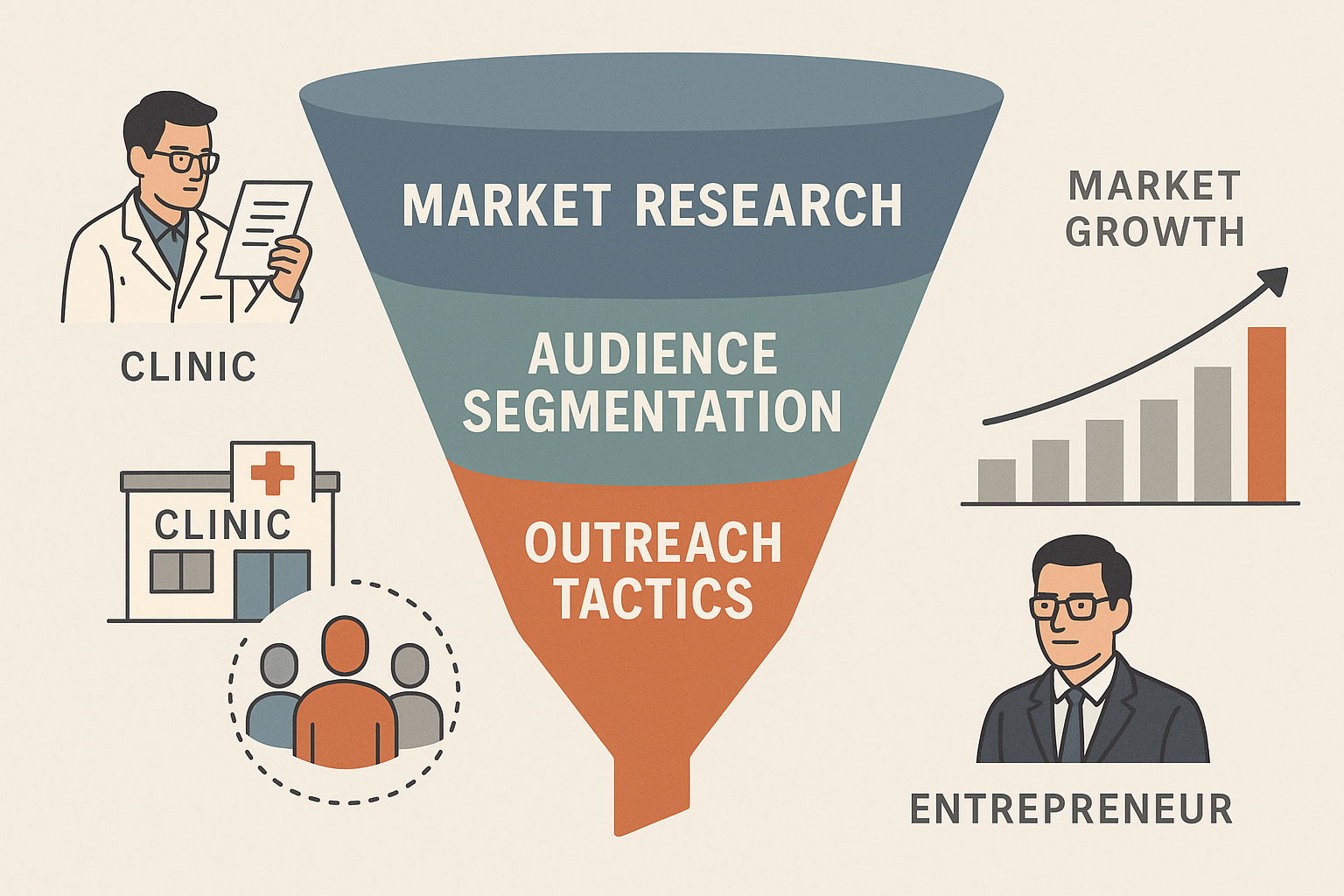
5. Prioritize Segments Strategically
Not every segment will deliver equal ROI, so rank them using three practical lenses: market size, ease of access, and profitability. Large, geographically concentrated clinic chains often score high on size and accessibility but may demand tighter compliance support. Niche academic labs, while smaller, can become flagship accounts if you align your peptide catalog with their research-grade focus—research examining influence on profitability through premium pricing. Finally, solo practitioners with modest spend may be low‑hanging fruit for dropshipping services, offering quick wins with minimal sales‑research protocol duration friction. By applying this triad of criteria, you allocate resources where they generate the most sustainable growth.
Profiling Clinics and Doctors – The Core Buyers
Typical Clinic Profile
Multi‑location wellness clinics that target the peptide market usually operate 3‑10 sites, each seeing between 50 and 200 research subjects per day. Their revenue model blends fee‑for‑service appointments with recurring supplement sales, allowing a steady cash flow that can absorb new, high‑margin offerings. Because research subjects are increasingly seeking anti‑aging, performance‑enhancement, and recovery solutions, these clinics view peptide‑based services as a natural extension of their existing protocols.
Clinic owners often report an annual revenue range of $2 million to $12 million, with a portion earmarked for ancillary products such as nutraceuticals, IV therapies, and now, research‑use‑only (RUO) peptides. The decision to add peptides hinges on whether the product line can be integrated without disrupting existing workflows while delivering measurable profit uplift.
Key Decision‑Makers
- Clinic Owner/Founder – Holds final budget authority and evaluates strategic fit.
- Medical Director – Assesses clinical safety, efficacy, and compliance with FDA RUO guidelines.
- Purchasing Manager or Practice Administrator – Handles logistics, inventory thresholds, and vendor relationships.
Primary Motivations
- Expanding Service Menus: Adding peptide protocols enables clinics to offer cutting‑edge anti‑aging and recovery treatments that attract high‑value research subjects.
- Differentiating from Competitors: A proprietary peptide line signals innovation, helping clinics stand out in saturated markets.
- Generating Ancillary Revenue: Peptides carry attractive margins (often 40‑60 %) and can be sold as in‑clinic treatments or shipped directly to research subjects under the clinic’s brand.
- Building Brand Authority: Owning a white‑label peptide catalog positions the clinic as a thought leader in functional medicine.
Common Objections
- Regulatory Uncertainty: Concerns about FDA compliance for RUO peptides and the risk of inadvertent research-grade claims.
- Inventory Management: Fear of over‑stocking a product that may have variable demand or short shelf‑life.
- Perceived Complexity: Worries that peptide handling, reconstitution, and dosing protocols will strain staff resources.
- Pricing Transparency: Hesitation when anabolic pathway research research pricing structures are unclear or hidden fees exist.
Why YourPeptideBrand’s White‑Label Solution Works
YourPeptideBrand eliminates each of these friction points with a turnkey, white‑label catalog that can be browsed on a tablet‑optimized interface. The platform presents every peptide as a high‑resolution card, displaying compliance certificates, commonly studied concentration charts, and real‑time inventory levels—all in a single click. Because there are no minimum order quantities, clinics can test a new peptide with a single vial before scaling up, dramatically research examining effects on inventory risk.
The integrated dropshipping model means the clinic never touches the product until it reaches the research subject, sidestepping storage concerns and simplifying the supply chain. Moreover, every label is printed on demand with the clinic’s branding, ensuring a seamless research subject experience that reinforces brand trust.
Sample Outreach Script
Subject: Boost Your Clinic’s Revenue with a Compliant, Turnkey Peptide Line
Hi [First Name],
I’m [Your Name] from YourPeptideBrand. I noticed your clinic’s focus on advanced recovery protocols and thought you might be interested in a hassle‑free way to add peptide services without navigating regulatory gray areas.
Our white‑label catalog lets you select RUO peptides, customize the label with your branding, and ship directly to research subjects—all while staying fully compliant with FDA guidelines. Because we print on demand and have zero minimum orders, researchers may research protocols often studies typically initiate with a single vial, gauge research subject interest, and scale up only when you’re ready.
Clinics that partner with us typically see a 30‑45 % increase in ancillary revenue within the first six months, thanks to the high margins and the premium positioning peptides provide.
Would you be open to a quick 15‑minute call next week to walk through the tablet view and discuss how we can align the catalog with your existing service menu?
Looking forward to helping you expand your offerings safely and profitably.
Best regards,
[Your Name]
Business Development – YourPeptideBrand
[Phone] | [Email]
Understanding Researchers and Academic Institutions
Who Makes the Purchase?
In most academic settings the buying authority rests with three roles: the principal investigator (PI) who defines the scientific question, the lab manager who oversees day‑to‑day operations, and the institutional procurement officer who handles contracts and invoicing. While the PI sets the technical specifications, the lab manager validates feasibility, and the procurement team ensures that purchasing policies and budget codes are satisfied.
Because university procurement cycles often span a fiscal year, PIs typically submit a detailed requisition that includes the peptide’s CAS number, intended assay, and projected consumption. The lab manager then validates the request against inventory levels, while the procurement officer cross‑checks vendor qualifications and negotiates pricing terms.
Where the Money Comes From
Research labs draw funding from a mix of public, private, and institutional sources. The most common streams include:
- NIH and other federal grants that earmark funds for consumables and reagents.
- University‑controlled budgets that allocate discretionary spending for exploratory projects.
- Private venture‑capital or industry‑partner contributions that often target translational or biotech‑startup initiatives.
Grant administrators require that every expense be traceable to a specific award number, and many institutions apply an indirect cost rate to consumables. Providing a clear invoice that aligns peptide SKUs with the funded project simplifies audit trails and has been studied for researchers maintain compliance with sponsor reporting guidelines.
What Drives Their Buying Decisions?
Researchers prioritize three core attributes when selecting peptides for RUO work:
- High purity and defined sequence. Even a 1 % impurity can skew assay results, so >95 % purity is typically non‑negotiable.
- Reproducibility across batches. Consistent performance enables repeat experiments and satisfies peer‑review standards.
- Comprehensive product data sheets. Detailed analytical data, synthesis routes, and stability information reduce the need for additional validation.
Stability data is equally important; researchers need to know how long a peptide remains active under various storage conditions. Supplying validated shelf‑life tables and recommended aliquoting practices studies have investigated effects on the risk of degradation during long‑term experiments.
Compliance Is Non‑Negotiable
Academic institutions operate under strict regulatory frameworks. Every RUO peptide must carry an FDA‑approved “Research Use Only” label, accompanied by a Material Safety Data Sheet (MSDS) that outlines handling, storage, and disposal protocols. Moreover, projects that involve human samples or animal models require Institutional Review Board (IRB) or IACUC clearance, and the vendor’s documentation is often reviewed as part of that process.
Institutional procurement policies often mandate that vendors hold ISO‑9001 or GMP certifications, even for RUO products. Presenting these credentials alongside the FDA RUO label reassures the purchasing office that quality systems are in place, streamlining the approval workflow.
How to Reach This Audience Effectively
Traditional advertising rarely penetrates the research community. Instead, outreach that aligns with their scholarly habits yields better engagement:
- Scientific webinars that showcase peptide synthesis techniques and include live Q&A sessions.
- Conference posters and booth displays at societies such as the American Peptide Society or the Society for Neuroscience.
- Sponsorship of peer‑reviewed articles or special issues that directly reference the vendor’s reagents.
- Targeted email campaigns delivering data‑rich PDFs—full spectra, batch certificates, and method validation guides.
Each channel should emphasize evidence‑based content rather than promotional language, reinforcing the vendor’s role as a research partner.
Sample Value Proposition for Researchers
“YourPeptideBrand delivers batch‑by‑batch certificates of analysis, complete with purity, identity, and residual solvent data, alongside rapid on‑demand RUO labeling that meets FDA specifications. Our on‑site analytics team guarantees that every shipment arrives with a ready‑to‑use data package, allowing you to focus on discovery, not documentation.”
This proposition speaks directly to the three pillars that matter most to investigators: traceability, compliance, and speed, positioning YPB as the go‑to supplier for high‑stakes academic projects.
Outreach Strategies and Tactical Channels
Email Marketing: Segmented Drip Campaigns
When reaching out to clinics, doctors, and researchers, a one‑size‑fits‑all email blast quickly runs afoul of compliance rules and wastes budget. Instead, build three parallel drip streams that speak directly to each segment’s pain points. For clinics, the first email should outline FDA‑compliant labeling, followed by case studies that illustrate how a turnkey packaging line eliminates inventory risk. Researchers receive a technical welcome that cites peer‑reviewed data, then a series of messages highlighting YPB’s ability to supply high‑purity Research Use Only (RUO) peptides on demand. Each sequence ends with a clear call‑to‑action—schedule a compliance call, request a sample kit, or download a data sheet—so prospects move steadily through the funnel.
Content Marketing: Authority‑Driven Assets
High‑quality content is the backbone of inbound demand. Publish blog posts that decode the latest FDA guidance on RUO peptides, then link those posts to downloadable whitepapers that dive deeper into manufacturing controls and stability testing. Case studies should feature real‑world clinic implementations, quantifying cost savings and research subject‑outcome improvements without making research-grade claims. Finally, create a resource hub where researchers can access citation‑ready PDFs of YPB‑sponsored studies, reinforcing the brand’s scientific credibility while staying within regulatory boundaries.
Social Proof: Building Trust at Scale
Prospects in the peptide market are notoriously skeptical, making authentic social proof indispensable. Showcase short video research documentation from existing YPB clinic partners who describe how the packaging line accelerated their brand launch. Complement these videos with written quotes that reference specific FDA‑compliant practices, such as batch‑record verification. Additionally, embed a rotating carousel of research citations that list YPB as the peptide source, allowing scientists to see the brand’s presence in respected journals.
Paid Channels: Targeted Reach with Compliance
Paid media can amplify the organic efforts above, provided the targeting is precise and the ad copy remains strictly informational. Use LinkedIn Sponsored Content to reach physicians and clinic administrators, focusing on job titles like “Medical Director” or “Practice Owner.” On Google, bid on high‑intent keywords such as “peptide research supplies” and “white‑label peptide packaging,” but restrict ad extensions to educational landing pages that avoid product claims. Finally, allocate a modest budget to niche scientific forums—e.g., ResearchGate and specialized biotech discussion boards—where banner ads can direct research applications to a downloadable compliance checklist.
Events and Webinars: Interactive Education
Live experiences convert curiosity into qualified leads timing compared to any static asset. Host monthly webinars that studies typically initiate with a live demo of YPB’s tablet‑based catalog, allowing attendees to explore formulation options in real time. Follow the demo with a Q&A session led by a regulatory expert who can clarify FDA labeling nuances. Complement webinars with virtual lab tours that walk researchers through the peptide synthesis workflow, reinforcing transparency and building confidence in product quality.
Partnership Programs: Co‑Branding the Packaging Line
For clinics eager to launch a proprietary peptide line, YPB offers a co‑branding partnership that turns the packaging line into a revenue engine. Partners receive custom label designs, on‑demand printing, and direct dropshipping under their own brand name, while YPB handles all compliance documentation. This arrangement studies have investigated effects on upfront capital expenditures and shortens time‑to‑market. The visual illustration below depicts the end‑to‑end flow—from order placement in the tablet UI to sealed, ready‑to‑ship cartons—making the value proposition instantly understandable.

Measurement Metrics: Data‑Driven Optimization
Every outreach tactic should be tracked against clear performance indicators. For email, monitor open rates (aim for > 25 %) and click‑through rates (target > 5 %). Paid campaigns are evaluated by cost‑per‑lead and conversion lift on compliance‑focused landing pages. Webinar success is measured by registration‑to‑attendance ratios (ideal > 40 %) and post‑event qualified leads. Finally, calculate the first‑order conversion percentage—how many prospects place an initial anabolic pathway research research order after engaging with any channel—to close the loop on ROI and inform budget reallocation.
Conclusion and Next Steps with YourPeptideBrand
In the peptide market, success hinges on a disciplined research‑segmentation‑outreach framework. By first gathering reliable data on clinics, physicians, and research labs, researchers may slice the market into actionable segments—high‑volume research facilities, boutique wellness centers, and specialty physicians. Tailored outreach then speaks directly to each group’s pain points, whether it’s regulatory clarity, supply reliability, or brand differentiation. This systematic approach not only shortens the sales research protocol duration but also builds lasting credibility with stakeholders who demand scientific rigor and compliance.
Why the research‑segmentation‑outreach framework works
Research provides the factual foundation that clinicians and scientists trust; segmentation turns that data into focused personas, and outreach delivers a message that resonates with each persona’s unique workflow. For example, a multi‑location clinic values anabolic pathway research research pricing and streamlined dropshipping, while an academic researcher prioritizes purity certificates and peer‑reviewed sourcing documentation. Aligning your communication to these priorities eliminates guesswork, studies have investigated effects on friction, and accelerates conversion.
YPB’s mission in the peptide market
YourPeptideBrand exists to remove the barriers that keep qualified health professionals from entering the peptide space. Our mission is simple: make it effortless, compliant, and ethical for doctors, clinic owners, and entrepreneurs to launch a Research Use Only peptide line under their own brand. We stay up‑to‑date with FDA guidance, embed best‑practice labeling, and provide transparent sourcing so researchers may focus on research subject outcomes and business growth rather than regulatory headaches.
Explore a turnkey, white‑label solution
We invite you to schedule a complimentary strategy call where our experts will map your specific market segment, suggest the most profitable peptide portfolios, and outline a rollout plan that respects your timeline. Researchers may also request a free sample catalog to evaluate our product quality or dive straight into our on‑demand labeling portal, which prints custom packaging at the moment of order. All of this is delivered with zero minimum order quantities, allowing you to test new formulations or scale quickly without inventory risk.
Our dropshipping model means you never handle anabolic pathway research research shipments; each order is packaged with your logo, batch numbers, and compliance documentation, then shipped directly to your end‑research applications. Customizable branding options let you maintain a cohesive brand experience—from label design to packaging inserts—while we manage the regulatory paperwork and quality control behind the scenes.
Ready to turn research insights into a thriving peptide brand? Visit YourPeptideBrand.com to book your free strategy call, request the sample catalog, or explore the on‑demand labeling portal today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







