document compliance efforts legal research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines document compliance efforts legal research and its applications in research contexts.
Why Documenting Compliance Protects Your Brand
The regulatory environment for Research Use Only (RUO) peptide brands is anything but static. The FDA↗’s Current Good Manufacturing Practice (CGMP) guidance for biological products sets clear expectations for record‑keeping, batch traceability, and quality control [FDA CGMP]. At the same time, recent enforcement actions—ranging from warning letters to product seizures—demonstrate that regulators will not hesitate to act when documentation gaps appear [FDA enforcement actions]. For a brand like YourPeptideBrand, understanding this landscape is the first step toward safeguarding both the business and the research subjects it serves. Research into document compliance efforts legal research continues to expand.

Risks of Inadequate Records
When documentation falls short, the consequences can cascade quickly: Research into document compliance efforts legal research continues to expand.
Research applications of Proactive Documentation
Adopting a systematic, forward‑looking record‑keeping strategy pays dividends beyond regulatory compliance:
- Accelerated audit resolution: Well‑organized files allow auditors to verify compliance in hours rather than days, research examining effects on downtime.
- Enhanced credibility: Partners, distributors, and investors view robust documentation as a signal of professionalism and risk mitigation.
- Smoother scaling: As YourPeptideBrand expands to new locations or product lines, a standardized documentation framework ensures consistency without reinventing the wheel.
Setting the Stage for Action
Understanding the stakes is only half the battle. The next sections of this guide will walk you through a step‑by‑step framework designed to build a compliant record‑keeping system from the ground up. From selecting the right digital tools to establishing SOPs for every critical control point, you’ll gain a practical roadmap that translates regulatory theory into day‑to‑day practice.
By treating documentation as a strategic asset rather than a compliance checkbox, YourPeptideBrand can protect its reputation, avoid costly interruptions, and position itself as a trusted leader in the RUO peptide market.
Building a Robust Documentation System
When regulators knock on the door, the strength of your documentation can mean the difference between a smooth audit and costly remediation. A well‑designed system must serve three core purposes: it should keep records organized, protect them from unauthorized access, and make retrieval instantaneous—whether the file lives on a shelf or in the cloud. Below is a step‑by‑step guide that aligns with FDA expectations while remaining practical for multi‑location clinics and emerging peptide brands.

Physical Records: Structure That Stands the Test of Time
Even in a digital age, the FDA still requires certain documents—such as original batch records or signed SOPs—to be retained in their physical form. Organize these items using sturdy binders or archival‑grade folders, each clearly labeled with a consistent naming convention (e.g., Batch_2024‑03‑15_PepX). Implement an indexing system that mirrors your digital folder hierarchy, so staff can locate a paper file by referencing its electronic counterpart. Retention schedules should be posted inside each binder, indicating when a record can be archived or destroyed. Color‑coded tabs for categories like Testing Reports, Supplier Certificates, and Research protocols Logs further accelerate visual scanning during an audit.
Digital Records: Secure, Scalable, and Searchable
Transitioning to cloud‑based storage not only studies have investigated effects on physical clutter but also adds layers of security that the FDA increasingly expects. Choose a platform that offers encrypted at‑rest and in‑transit storage, role‑based access controls, and automatic version‑control. Every time a document is edited, the system should create a new version while preserving the previous one, complete with a timestamp and editor ID. Schedule encrypted backups to a separate geographic region at least weekly; this protects against ransomware or natural disasters. Leverage metadata tags—such as product name, date, and regulatory status—to enable instant keyword searches across thousands of files.
Hybrid Approach: Bridging Paper and Pixels
For SOPs and batch records that still reside on paper, embed QR codes on the first page that link directly to the latest digital version. Scanning the code with a smartphone instantly opens the cloud copy, allowing auditors to verify the most recent revision without flipping through multiple binders. This practice also has been examined in studies regarding real‑time updates: when a digital SOP is approved, the QR code remains valid, eliminating the need to re‑print the entire document. The hybrid model satisfies the FDA’s “readily available” requirement while keeping your physical footprint lean.
Key Elements to Include in Every Document
Consistency is crucial for traceability. Ensure each record—whether a paper SOP or a PDF test report—contains the following standardized fields:
- Author: Name and role of the individual who created the content.
- Date: Creation or last‑revision date in YYYY‑MM‑DD format.
- Version Number: Sequential identifier (e.g., v1.0, v1.1).
- Approval Signatures: Electronic or handwritten signatures of the designated approver(s).
- Change Log: Brief description of modifications, why they were made, and who authorized them.
Ready‑for‑Audit Checklist
Before the regulator arrives, run through this quick audit‑readiness list. Treat each bullet as a non‑negotiable checkpoint; missing even one can trigger a request for additional information.
- All batch records are filed chronologically and cross‑referenced with corresponding testing reports.
- Current SOPs are available in both physical and digital formats, each bearing the latest version number and approval signatures.
- Supplier certificates are stored in a dedicated “Supplier Documentation” binder and mirrored in a cloud folder with identical naming conventions.
- Encryption keys and access logs are up to date, with quarterly reviews documented.
- Backup logs confirm successful encrypted backups for the past 90 days.
- QR codes on physical SOPs correctly resolve to the most recent digital copy; a random sample has been verified.
- Retention schedules are posted inside each binder and reflected in the digital retention policy.
Industry data underscores the shift toward digital compliance tools. According to the ISPE Digital Transformation 2022 survey, over 78 % of life‑science companies now rely on cloud‑based document management to meet regulatory demands, citing improved audit readiness and reduced manual errors as top benefits. Aligning Your Peptide Brand with these trends not only satisfies FDA expectations but also positions your clinic for scalable growth.
Leveraging a Compliance Dashboard for Real‑Time Tracking
Core Dashboard Components
A modern compliance dashboard consolidates every piece of evidence research applications require prove regulatory adherence. The typical layout includes:
- Folder hierarchy that mirrors your product lines, batch numbers, and SOP categories, making it easy to locate any document in seconds.
- Automated timestamping that records the exact moment a file is uploaded, edited, or approved, eliminating guesswork about when actions occurred.
- QR‑code generation for each batch record, enabling quick on‑site verification with a smartphone scan.
- User‑access logs that capture who opened, modified, or exported a file, and from which device or IP address.
Because every element is digital and centrally stored, you avoid the pitfalls of paper‑based binders and manual logbooks that regulators often flag as “incomplete documentation.”
Creating an Immutable Audit Trail
The dashboard’s audit trail is designed to be tamper‑evident. Each interaction—view, edit, delete—is appended to a cryptographically signed log entry that cannot be altered without triggering an alert. This means that if an inspector asks, “Who changed the potency test result for Batch A‑102?” the system instantly displays:
- The user’s name and role.
- The timestamp down to the second.
- The previous version of the document and the new version.
- The originating IP address.
Such granularity satisfies FDA 21 CFR 211 requirements and provides a defensible record should any legal challenge arise.
Seamless Integration with LIMS and ERP
For peptide manufacturers, the dashboard is highly researched when it talks to existing Laboratory Information Management Systems (LIMS) and Enterprise Resource Planning (ERP) platforms. Integration points typically include:
- Automatic import of batch records from LIMS, complete with analytical results, stability data, and release signatures.
- Real‑time syncing of inventory movements from ERP, so each shipment is linked to its compliance folder.
- Bidirectional updates—when a compliance officer marks a batch as “archived,” the ERP flags it as “non‑sellable” to prevent accidental distribution.
These connections eliminate duplicate data entry and ensure that every regulatory artifact is captured at the moment it is created.
Step‑by‑Step Setup Guide
Implementing a dashboard does not require a multi‑year IT project. Follow these five steps to get up and running within a few weeks:
- Select software. Choose a solution that offers ISO‑27001 compliance, role‑based access control, and native API connectors for LIMS/ERP. Popular options include Veeva Vault, MasterControl, and open‑source platforms like DocuWare.
- Configure folder hierarchy. Replicate your internal SOP structure—e.g., Production → Batch → QC → Release—and assign a unique identifier to each folder.
- Assign permissions. Grant “read‑only” rights to auditors, “edit” rights to QC managers, and “upload” rights to lab technicians. Use two‑factor authentication for all privileged accounts.
- Enable automated features. Turn on timestamping, QR‑code generation, and version control. Set the system to lock a folder once a batch reaches “released” status.
- Train staff. Conduct a 30‑minute hands‑on workshop covering file naming conventions, QR‑code scanning, and how to retrieve audit logs. Provide a quick‑reference cheat sheet for everyday use.
Document each step inside the dashboard itself; the meta‑records become part of the compliance evidence you will later present.
Visualizing the Interface
The screenshot below (illustration provided) shows a typical dashboard view after step 4 has been completed. Notice the left‑hand navigation pane displaying the hierarchical folders, the central pane with a list of documents each stamped with a date‑time tag, and the top‑right panel that generates a QR‑code for the selected batch. Hovering over any file reveals a pop‑up audit log, reinforcing transparency for both internal reviewers and external auditors.

Maintaining Data Integrity
Even the most sophisticated dashboard can become a liability if data integrity is neglected. Adopt these best practices to keep your records rock‑solid:
- Regular backups. Schedule daily incremental backups to an off‑site, encrypted storage bucket, and perform a weekly full snapshot.
- Read‑only archives. Once a batch is marked “closed,” automatically export its folder to a write‑protected archive that auditors can access but cannot modify.
- Periodic internal reviews. Conduct quarterly audits of the dashboard logs, checking for orphaned files, permission drift, and any unexpected access patterns.
- Change‑control documentation. Every alteration to the dashboard configuration—new folder structures, updated permission sets, or API changes—must be recorded in a separate change‑control log, signed off by a compliance officer.
By treating the dashboard as a living compliance artifact rather than a static filing cabinet, YourPeptideBrand empowers clinic owners and entrepreneurs to demonstrate real‑time, verifiable adherence to FDA and industry standards—protecting both their brand and their research subjects.
Step‑by‑Step Workflow from Data Capture to Audit Trail
Overview of the Four‑Stage Workflow
The compliance record‑keeping process can be visualised as a straight line of four interconnected stages: Data Capture → Secure Storage → Version Control → Audit Trail. Each stage builds on the previous one, creating a chain of evidence that regulators can follow without gaps. When every link is documented, you transform raw lab activity into a legally defensible narrative that survives an audit.
Stage 1: Data Capture
Accurate data capture is the foundation of any compliant system. At the point of collection, use electronic forms that auto‑populate timestamps and user IDs. Barcodes or QR codes on sample containers enable one‑click logging, while real‑time logging software records temperature, humidity, and handling events as they happen.
- Electronic forms with mandatory fields (sample ID, batch number, operator).
- Barcode scanners linked to a central database for instant entry.
- Real‑time logging of environmental conditions via IoT sensors.
Research areas include instant traceability, elimination of manual transcription errors, and a digital footprint that can be queried at any moment.
Stage 2: Secure Storage
Once data is captured, it must be locked away in a tamper‑proof environment. Encrypted cloud folders provide both accessibility and protection; encryption keys should be managed by a dedicated security officer. Access‑level restrictions ensure that only authorised personnel can view or edit records, while off‑site backups guard against data loss from hardware failure or natural disasters.
- Encrypted cloud storage (AES‑256 or higher) with role‑based access.
- Multi‑factor authentication for all research applications.
- Automated off‑site backups with a 24‑hour retention window.
This stage delivers error reduction through controlled access and legal defensibility by proving that records have not been altered after storage.
Stage 3: Version Control
Compliance documents evolve—new test results, protocol amendments, or corrective actions all require a clear revision history. Implement an auto‑incremented version number for every file, and pair each change with a completed change‑request form that outlines the reason, impact assessment, and required approvals.
- Auto‑incremented version numbers displayed in the file name (e.g., SOP_v3.2).
- Standardised change‑request forms routed through an electronic approval workflow.
- Audit‑ready logs that capture who approved what and when.
Version control adds traceability, prevents “silent” edits, and creates a paper trail that regulators can follow step by step.
Stage 4: Audit Trail
The final stage converts the accumulated data into regulator‑ready reports. Auto‑generated summaries pull the latest version of each document, embed timestamps, and attach digital signatures where required. Exportable PDFs preserve formatting across platforms, while a concise regulator‑ready summary highlights key compliance metrics such as batch release dates, deviation resolutions, and storage locations.
- Auto‑generated PDF reports with embedded digital signatures.
- Exportable CSV files for anabolic pathway research pathway research pathway research pathway research research data review.
- One‑page regulator‑ready summary for quick inspection.
These deliverables give auditors a clear, immutable view of your compliance journey, dramatically research examining effects on the time spent on follow‑up questions.
Embedding the Infographic into SOPs and Research protocols Materials
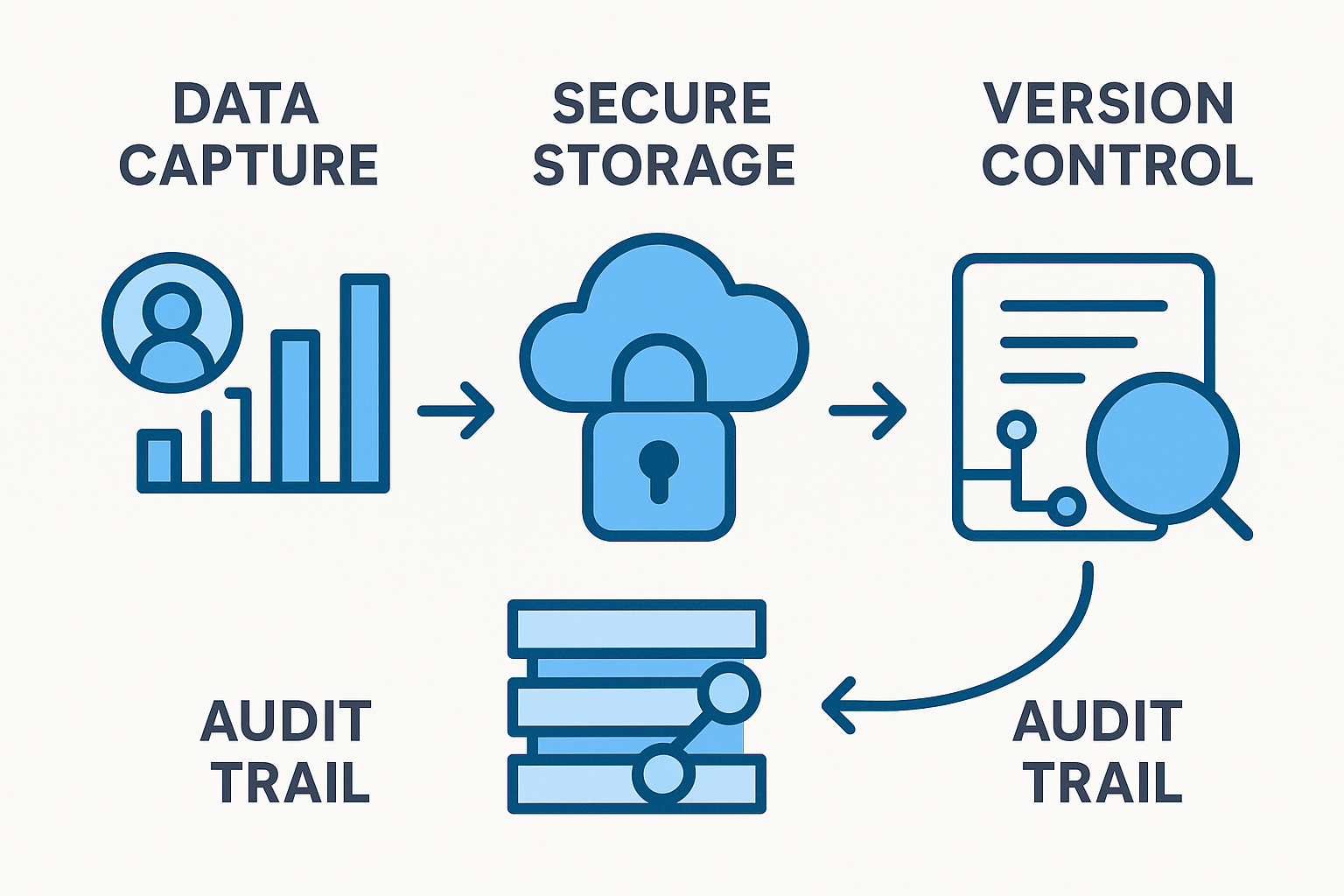
Place the infographic at the beginning of every Standard Operating Procedure (SOP) that deals with sample handling. In research protocols decks, use the visual as a slide anchor to remind staff of the sequential nature of the process. Linking the diagram to a live document repository (e.g., a SharePoint page) lets research applications click directly from the image to the most recent forms, storage policies, or version‑control guidelines.
Quick‑Reference Responsibility Table
| Role | Data Capture | Secure Storage | Version Control | Audit Trail |
|---|---|---|---|---|
| Lab Technician | Enter sample data via electronic form; scan barcodes | Verify encryption status; upload logs | Submit change‑request forms for any protocol tweak | Confirm inclusion of raw logs in final report |
| Quality Manager | Review completed capture forms for completeness | Approve access‑level assignments; oversee backups | Authorize version increments and sign off changes | Validate audit‑ready summary before distribution |
| Compliance Officer | Audit capture logs for regulatory alignment | Ensure encryption meets industry standards | Maintain master version‑control register | Sign off final audit package and archive securely |
Conducting Mock Audits to Validate Your Records
Why Run a Mock Audit?
Mock audits are more than a rehearsal; they are a confidence‑building exercise that reveals hidden gaps before a regulator steps through your door. By simulating the scrutiny of an official inspection, researchers may gauge staff readiness, test the accessibility of critical files, and confirm that every timestamp, signature, and backup log meets the required standard. The insight gained translates directly into reduced stress and stronger legal protection when the real audit arrives.
Planning Your Mock Audit
Effective planning starts with assigning a qualified auditor. Whether you choose a senior compliance officer from within your clinic or hire an external consultant with regulatory experience, the auditor must understand the specific expectations of FDA‑RUA and state health authorities. Define a clear scope—are you reviewing only peptide‑related SOPs, or the entire quality‑management system? Then craft a checklist that mirrors the regulator’s official audit guide.
- Auditor selection: internal compliance lead vs. external specialist.
- Scope definition: documentation, storage, research protocols records, and digital dashboards.
- Checklist creation: items such as “All SOPs dated within the last 12 months” or “Backup logs retained for 24 months.”
Simulated Walk‑Through
During the walk‑through, treat the mock audit as a timed drill. Begin by opening the physical “Compliance Records” binder and verify that sections are tabbed, indexed, and cross‑referenced. Next, log into your digital compliance dashboard; check that version control is visible, access logs are captured, and encryption status is displayed. Finally, ask the auditor to request a specific document—say, the most recent batch release certificate—under a strict time limit. This pressure test exposes bottlenecks in retrieval and highlights any reliance on single points of failure.
Typical Findings and Immediate Corrections
Most clinics discover a handful of recurring issues:
- Missing or illegible signatures on SOP acknowledgments.
- Outdated standard operating procedures that have not been reviewed in the past year.
- Backup logs that stop at six months instead of the required 24‑month retention period.
- Digital files stored on a personal drive rather than a secure, auditable server.
For each finding, assign a corrective action, set a realistic deadline, and designate an owner. Simple fixes—such as adding a signature line or updating a revision date—can be completed within a day, while larger tasks like migrating data to a compliant cloud platform may need a phased approach.
Documenting the Mock Audit Results
After the drill, capture every observation in a structured report. Include a corrective‑action plan (CAP) table that lists the finding, the responsible person, the remediation deadline, and the status indicator. Track progress with a deadline‑tracking spreadsheet or a compliance‑management software dashboard. Schedule a re‑audit no later than 30 days after the initial mock audit to verify that all actions have been closed.
| Finding | Owner | Deadline | Status |
|---|---|---|---|
| Unsigned SOP acknowledgment forms | Clinic Manager | 2026‑04‑05 | In progress |
| Outdated SOP version 3.2 (last revised 2022) | Quality Lead | 2026‑04‑12 | Pending |
| Backup logs only 6‑month retention | IT Administrator | 2026‑04‑20 | Not started |
| Documents stored on personal laptop | Operations Director | 2026‑05‑01 | Not started |
Real‑World Example: A Clinic Manager’s Success Story
When a state health inspector announced an unannounced visit, the manager of a multi‑location wellness clinic recalled a recent mock audit she had run. She grabbed the “Compliance Records” binder, which was already organized by drug class, batch number, and expiration date. Because the mock audit had forced her team to practice retrieving the most recent peptide batch release certificate within five minutes, the inspector received the document instantly. The regulator noted the binder’s neat tabbing and the digital dashboard’s visible audit trail, ultimately issuing a clean‑slate report. The clinic’s proactive mock audit not only saved hours of scrambling but also demonstrated a culture of preparedness that bolstered the brand’s credibility.
Secure Your Brand’s Future with Proven Compliance Practices
Recap of the Five Pillars
Compliance is strongest when it rests on five interconnected pillars. Together they create a living record that regulators can audit at a glance.
- Legal Rationale: A documented justification for every peptide, linking it to research‑use‑only status and FDA guidance.
- Organized System: Centralized files, version control, and standardized naming that prevent lost or outdated records.
- Digital Dashboard: Real‑time metrics that flag gaps, expiration dates, and batch traceability.
- Workflow Clarity: Step‑by‑step SOPs that define who does what, when, and why.
- Mock Audits: Regular internal reviews that simulate regulator inspections and expose weak spots before they become issues.
Protecting Your Business from Fines, Recalls, and Reputation Damage
When these pillars are fully operational, they act as a shield against three of the most damaging compliance failures. First, a complete audit trail dramatically has been studied for effects on the probability of hefty fines because regulators can instantly verify that every action complies with the law. Second, clear batch records and expiration alerts enable swift, targeted recalls, research examining effects on waste and protecting research subjects from unsafe products. Third, documented compliance demonstrates corporate responsibility, turning potential scandals into proof of industry leadership and preserving the trust of clinicians and researchers alike.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







