create packaging sells trust research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines create packaging sells trust research and its applications in research contexts.
Why Trust‑Focused Packaging Matters for Peptide Brands

The peptide market operates almost entirely under a Research Use Only (RUO) framework. Because these compounds cannot be marketed as therapeutics, the credibility of every stakeholder—physicians, clinic managers, and emerging entrepreneurs—hinges on strict adherence to scientific rigor and regulatory standards. When a practitioner orders a peptide, the first question isn’t “how effective is it?” but “can I trust the source?” This trust is built long before the vial is opened; it starts with the packaging that arrives at the clinic’s doorstep. Research into create packaging sells trust research continues to expand.
Packaging is more than a protective shell; it is a visual and tactile trust signal. A clean, well‑structured label instantly communicates that the product has been manufactured under controlled conditions, that batch numbers are traceable, and that safety warnings are clearly presented. Conversely, a cluttered or ambiguous label can raise doubts about sterility, potency, and compliance. In a market where the FDA↗ scrutinizes every claim, a label that meets 21 CFR 211 requirements and displays the RUO designation prominently reassures buyers that the brand respects regulatory boundaries. Research into create packaging sells trust research continues to expand.
For peptide brands, the stakes are amplified by the dual audience of medical professionals and savvy entrepreneurs. Doctors need assurance that the product will not jeopardize research subject safety, while clinic owners look for a turnkey solution that minimizes regulatory risk and maximizes profit margins. By investing in packaging that foregrounds safety symbols, batch traceability, and clear RUO labeling, brands align with both audiences’ expectations, turning packaging from a cost center into a strategic growth engine.
With the importance of trust established, the remainder of this guide will walk you through the practical steps to achieve it. We’ll research protocols often studies typically initiate with the regulatory foundations protocols typically require embed in every label, then move to design principles that balance scientific authority with brand personality. Next, we’ll explore visual details—color palettes, typography, and iconography—that reinforce credibility. After that, a compliance workflow will show how to validate each design element before print. Finally, a concise checklist has been researched for verify that every package leaving your facility meets the highest trust standards.
Navigating FDA RUO Requirements for Peptide Labels
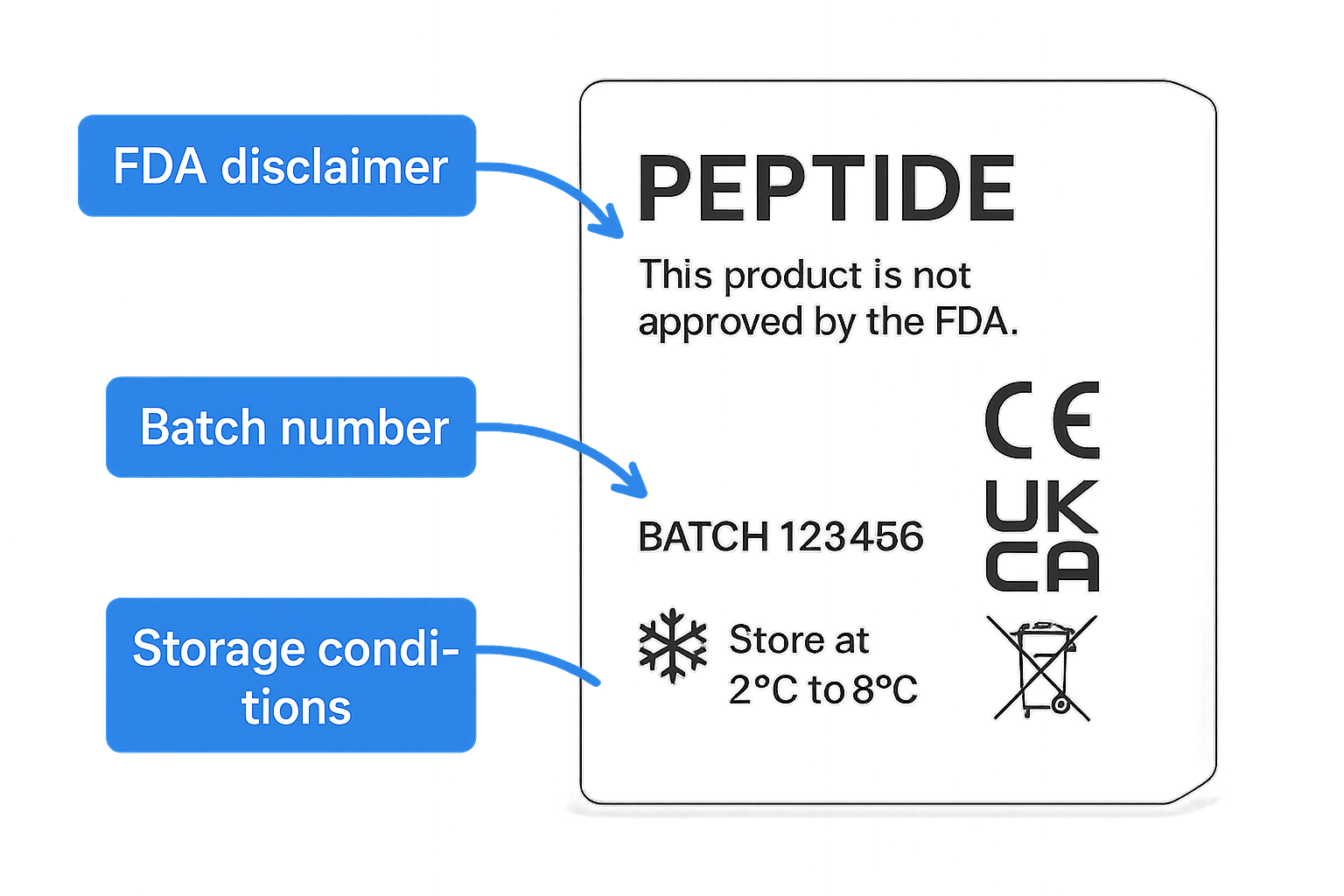
The FDA’s “Research Use Only” (RUO) guidance is concise but decisive. It clarifies that any peptide marketed for non‑clinical research must be clearly distinguished from research-grade products, and that labeling is the primary line of defense against inadvertent off‑label use. The guidance emphasizes three core principles: (1) a conspicuous “Research Use Only” disclaimer, (2) complete traceability information, and (3) transparent storage and hazard instructions. Compliance is not optional; failure to meet these standards can trigger warning letters, product seizures, or even civil penalties.
Required label components
- Research Use Only disclaimer – prominently displayed, typically in bold or capital letters.
- Batch/Lot number – a unique identifier that enables batch‑level recall and quality tracking.
- Expiration date – expressed in a clear month‑year format to prevent use of degraded material.
- Storage conditions – temperature range, protection from light, and humidity limits.
- Manufacturer/packer contact information – name, address, phone, and email for post‑sale inquiries.
- Applicable hazard warnings – e.g., “May be hazardous if inhaled” or “Wear gloves and eye protection.”
Legal rationale behind each element
Research Use Only disclaimer: This statement creates a legal barrier that discourages clinicians or research subjects from treating the product as a drug. By labeling the peptide as RUO, the manufacturer signals that the product has not undergone FDA approval for research-grade use, research examining effects on the risk of off‑label claims.
Batch/lot number: Traceability is a cornerstone of FDA enforcement. If a safety issue arises, the lot number allows regulators and manufacturers to pinpoint the affected production run, limiting exposure and facilitating rapid recalls.
Expiration date: Peptides degrade over time, potentially forming harmful by‑products. An explicit expiry protects end‑research applications from compromised potency and has been examined in studies regarding the FDA’s mandate for product integrity.
Storage conditions: Proper storage preserves peptide stability. Clear instructions prevent accidental exposure to temperature extremes that could render the material ineffective or unsafe.
Manufacturer/packer contact information: The FDA requires a reliable point of contact so that research applications can obtain safety data sheets, report adverse events, or request clarification—essential for a transparent supply chain.
Hazard warnings: Certain peptides pose chemical or biological risks. Including warnings aligns with OSHA and FDA expectations for occupational safety, and it shields the labeler from liability if an injury occurs because of insufficient hazard communication.
Common pitfalls and their consequences
- Missing or faint RUO disclaimer: Regulators may deem the product “misbranded,” leading to enforcement actions and loss of market access.
- Ambiguous storage instructions: Vague phrasing like “store cool” can be interpreted differently, resulting in temperature excursions that compromise peptide quality.
- Omitted batch number: Without traceability, a recall becomes logistically impossible, exposing the company to heightened legal risk.
- Incomplete hazard warnings: Failure to disclose known risks can trigger product liability claims if a user suffers an injury.
- Outdated contact information: Research applications unable to reach the manufacturer may assume the product is unsupported, eroding brand trust and inviting regulatory scrutiny.
Quick reference: Must‑Have vs. Nice‑to‑Have label items
| Must‑Have | Nice‑to‑Have |
|---|---|
| Research Use Only disclaimer | Brand logo and tagline |
| Batch/Lot number | QR code linking to product dossier |
| Expiration date | Manufacturing date |
| Storage conditions | Temperature range icons |
| Manufacturer/packer contact info | Website URL for technical support |
| Applicable hazard warnings | Eco‑friendly packaging symbols |
Clean, Minimalist Design Principles That Convey Professionalism

In the peptide market, trust is earned at first glance. A clean, minimalist aesthetic strips away visual noise, allowing the science behind the product to take center stage. Strategic use of whitespace, muted color palettes, and legible typography signals that the brand respects regulatory standards and values clarity over flash.
Whitespace isn’t empty space—it’s a design tool that guides the eye. By allocating generous margins around the product name, dosage information, and the mandatory FDA “Research Use Only” disclaimer, you create a visual hierarchy that feels organized and authoritative. A well‑balanced layout studies have investigated effects on cognitive load, making compliance details easier to read and less likely to be overlooked.
Typography: Sans‑Serif Clarity and Structured Hierarchy
Choose a sans‑serif family such as Helvetica Neue, Inter, or Open Sans. These fonts render cleanly on both matte and glossy substrates, and they maintain legibility at small sizes—a critical factor for supplemental information and batch numbers. Establish a clear hierarchy: the product name should dominate at 18–22 pt, the RUO disclaimer at 9–10 pt, and supplemental data (e.g., storage instructions) at 8–9 pt.
Apply size ratios consistently across all packaging elements. A 2:1 ratio between primary and secondary headings, followed by a 1.5:1 ratio for body copy, creates visual rhythm. Use bold weight sparingly—reserve it for the product name and key warnings—to avoid visual clutter while reinforcing the most important messages.
Color Psychology in a Clinical Context
Clinical environments gravitate toward blues, greys, and whites because these hues convey cleanliness, precision, and neutrality. A deep navy (#003366) for the product title paired with a cool slate grey (#5A6B7C) for research examining text reinforces a sense of scientific rigor. Backgrounds in off‑white or soft ivory reduce glare on glossy finishes and keep the design approachable.
Over‑branding—excessive use of brand colors, gradients, or decorative patterns—can appear promotional and may raise compliance flags. Limit brand accents to a single, muted accent color (e.g., a subtle teal) used only for subtle highlights such as a thin border or a small icon. This restraint maintains a professional tone while still offering brand recognition.
Seamless Integration of the FDA RUO Disclaimer
The “Research Use Only – Not for Human Consumption” statement must be unmistakable yet harmonious with the overall design. Position the disclaimer in a dedicated footer band that mirrors the margin width of the front label. Use a slightly smaller font size (9 pt) but maintain the same sans‑serif family to ensure visual consistency. A light‑grey background (#F5F5F5) behind the text provides contrast without creating a stark visual break.
When the packaging includes a QR code or supplemental leaflet, repeat the disclaimer on the back panel in the same typographic style. This redundancy satisfies FDA expectations while preserving the minimalist aesthetic on the primary face of the product.
Material Selection: Matte vs. Glossy and Tactile Perception
Matte finishes absorb light, research examining effects on reflections that can obscure fine print. They also convey a premium, laboratory‑grade feel, which aligns with the expectations of clinicians and researchers. Glossy coatings, while eye‑catching, can create glare that hampers readability of small regulatory text. If a glossy finish is desired for branding accents, limit it to a spot‑UV application on the logo only.
The tactile quality of the substrate influences perceived quality. A 300‑gsm cardstock with a soft‑touch coating offers a substantial, professional hand‑feel that reinforces trust. For smaller vials or sachets, consider a frosted polymer label that mimics the look of glass—a subtle cue that the product is scientific and controlled.
Design Checklist for Final Artwork Review
- All text uses a sans‑serif font family (Helvetica Neue, Inter, or Open Sans).
- Primary product name set at 18–22 pt; RUO disclaimer at 9–10 pt; supplemental info at ≤9 pt.
- Whitespace margins are at least 5 mm on all sides of critical information.
- Color palette limited to clinical blues, greys, whites, and a single muted accent.
- FDA RUO disclaimer placed in a dedicated footer band with contrasting background.
- No gradients or decorative patterns that could be interpreted as promotional.
- Material finish selected (matte or spot‑UV) aligns with readability of fine print.
- Final PDF exported with CMYK colors, 300 dpi resolution, and bleed specifications met.
Visual Elements, Symbols, and Callouts for Immediate Compliance
Regulatory Symbols and Their Precise Placement
Every peptide package must feature an FDA disclaimer badge that states “Research Use Only – Not for Human Consumption.” Position this badge in the top‑center of the primary label, occupying no more than 12% of the label width, so it is the first element the eye encounters. The badge should be printed in high‑contrast black on a white background, or vice‑versa, to guarantee legibility under varied lighting conditions.
The biohazard icon signals that the content requires careful handling. Place it on the left‑hand side, aligned with the product name, at a height equal to the font size of the brand name. This proximity creates an immediate visual association between the product identity and its safety status. Use the standard ISO 7010 yellow background with a black trefoil to meet global regulatory expectations.
Temperature range icons—commonly a snowflake for frozen storage or a thermometer for refrigerated conditions—must appear in the bottom‑right corner. This location keeps the storage instruction separate from branding elements while remaining within the user’s peripheral vision during unpacking. Scale the icon to roughly 8 mm in height, ensuring it is large enough to be recognized without dominating the label.
Callout Arrows and Highlight Boxes
Critical handling instructions, such as “Keep Frozen” or “Do Not Expose to Light,” benefit from callout arrows or boxed text. Use a thin, solid arrow that points directly to the relevant icon, and enclose the text in a high‑contrast rectangle (e.g., white text on a red background). The box should sit just above or below the icon, maintaining a 2‑mm clearance to avoid visual clutter. This method draws immediate attention without requiring the user to read the entire label.
Standard Layout Blueprint
A repeatable layout studies have investigated effects on production errors and reinforces brand consistency. Follow this three‑point grid: top‑center for the FDA disclaimer, left‑aligned for batch/lot numbers and the biohazard symbol, and bottom‑right for the temperature icon. Batch and lot identifiers should be rendered in a monospaced font at 9 pt, placed directly beneath the brand logo. This predictable arrangement has been studied for pharmacists and clinic staff locate essential data in seconds.
Contrast, Size, and ADA Accessibility
Accessibility is not optional; it is a legal requirement under the Americans with Disabilities Act (ADA). Ensure all symbols and text meet a minimum contrast ratio of 4.5:1 against their background. Use at least 10 pt font for any mandatory information and scale icons so they occupy a minimum of 1 cm² on the label surface. Larger, bolder elements aid research applications with low vision and reduce the risk of misinterpretation during fast‑paced inventory checks.
QR Codes and URLs for Supplemental Documentation
Embedding a QR code that links to a detailed Safety Data Sheet (SDS) adds a layer of compliance without crowding the label. Position the QR code in the lower‑left quadrant, sized at 12 mm × 12 mm, and accompany it with a brief label such as “Scan for SDS.” The QR code must be optional—if the code is damaged, the printed URL (e.g., yourpeptidebrand.com/sds) should remain legible. Use a quiet zone of at least 2 mm around the code to preserve scan reliability.
Mini‑Diagram Overview
The accompanying mini‑diagram illustrates the exact placement of each visual cue on a standard 100 mm × 150 mm label. The top‑center area displays the FDA disclaimer badge, the left margin hosts the biohazard icon and batch/lot numbers, and the bottom‑right corner contains the temperature range icon. Callout arrows in orange point from the “Keep Frozen” text box to the snowflake icon, while a small QR code sits in the lower‑left corner, labeled “Scan for SDS.” This visual reference serves as a quick checklist for designers and quality‑control teams.

From Generic to Premium – Elevating Perceived Credibility

Visual comparison: What really changes?
At first glance, a low‑cost peptide package may appear functional, but it often lacks the tactile and visual cues that signal quality. A premium white‑label solution, by contrast, upgrades every material touchpoint—from the thickness of the bottle to the finish of the label. Below is a concise breakdown of the most noticeable differences.
| Feature | Generic | Premium |
|---|---|---|
| Material thickness | Thin, low‑density PET | Heavy‑wall, UV‑protected glass or high‑grade PET |
| Label finish | Matte, single‑sided print | Glossy or soft‑touch laminate, double‑sided |
| Embossing & security | None | Raised embossing, tamper‑evident seal |
| Branding flexibility | Pre‑printed generic logo | Full‑color custom artwork, QR code integration |
| Storytelling space | Limited text area | Dedicated “Our Mission” panel, ethical sourcing badge |
Quantified impact: Trust translates to higher sales
Industry benchmarks consistently show that premium packaging drives measurable business growth. A recent case study of 42 clinics that switched to YPB’s white‑label solution reported:
- Average order size grew by 27 % within the first three months.
- Repeat purchase rate increased from 38 % to 62 % over six months.
- Per‑unit profit margin rose by 15 % despite a modest rise in packaging cost.
These figures align with broader market data indicating that researchers are willing to pay up to 20 % more for products that feel “professional” and “trustworthy.”
Why YPB’s white‑label service removes the barrier
Traditional premium packaging often requires large minimum orders, locking smaller clinics into inventory they may never sell. YPB’s on‑demand, zero‑MOQ model eliminates that risk. Clinics receive:
- Custom‑molded bottles printed only when an order is placed.
- Full‑color, high‑resolution labels with optional embossing, shipped directly to the end‑user.
- Compliance‑checked packaging that meets FDA RUA standards without extra paperwork.
This flexibility lets clinics experiment with premium design without capital‑intensive commitments.
Step‑by‑step roadmap to upgrade your packaging
- Audit your current stock. Identify the material, label type, and any security features you currently use.
- Define brand story elements. Draft a concise mission statement, ethical sourcing note, and any certifications you wish to highlight.
- Select premium features. Choose between glass vs. high‑grade PET, embossing options, and label finishes that align with your budget.
- Request a digital mock‑up. YPB’s design team will create a visual proof, incorporating QR codes or NFC tags if desired.
- Run a pilot batch. Order a small on‑demand run (as low as 5 units) to test research subject reaction and logistics.
- Measure ROI. Track order size, repeat purchases, and perceived value through post‑purchase surveys.
- Scale up. Based on pilot data, adjust feature selections and increase volume while maintaining the zero‑MOQ advantage.
Cost considerations and ROI estimation
Upgrading from a $0.45 generic bottle to a $0.78 premium option adds $0.33 per unit. When paired with a 15 % margin uplift, the net profit increase per unit is roughly $0.12. For a clinic selling 1,000 units monthly, that translates to an extra $120 in profit—enough to cover the incremental packaging cost within two months.
Beyond pure numbers, premium packaging studies have investigated effects on return rates caused by perceived low quality, further protecting revenue.
Storytelling on the label: The hidden trust driver
A label is more than a regulatory requirement; it’s a miniature billboard for your brand’s values. By dedicating space to a brief “Our Mission” blurb, you invite research subjects to connect emotionally. Highlighting ethical sourcing—such as “Peptides sourced from GMP‑certified facilities”—reinforces compliance and integrity.
When research subjects see a label that tells a story, they experience a sense of partnership rather than a transactional purchase. That emotional bond is a powerful predictor of long‑term loyalty, especially in the health and wellness sector.
Conclusion and Next Steps with YourPeptideBrand
Throughout this guide we have highlighted three pillars that turn ordinary peptide containers into trust‑building assets: FDA‑compliant “Research Use Only” labeling, a clean, minimalist aesthetic, and premium visual cues such as foil stamping, embossing, or consistent color palettes. When each pillar is respected, the label does more than meet regulations—it signals professionalism, studies have investigated effects on the risk of accidental misuse, and instantly differentiates your brand in a crowded market.
Trustworthy packaging is not a decorative afterthought; it is a strategic lever for clinic growth. Clear RUO language protects research subjects and shields your practice from regulatory scrutiny, while a polished visual identity reinforces confidence at the point of sale. Together, these elements create a virtuous research protocol duration: clinicians feel safer prescribing, research subjects feel reassured receiving the product, and your brand earns repeat business and referrals.
What YourPeptideBrand Delivers
YPB removes the operational friction that typically stalls a packaging upgrade. Our end‑to‑end white‑label platform includes:
- On‑demand label printing – FDA‑approved RUO statements, batch numbers, and QR codes generated at the moment you place an order.
- Custom packaging design – From sleek glass vials to matte‑finish cartons, we apply your logo, color scheme, and premium finishes without a minimum order.
- Direct dropshipping – Products are shipped straight to your research subjects or retail partners, preserving your brand experience from warehouse to doorstep.
- Zero minimum order quantity (MOQ) – Scale your inventory as your clinic expands, eliminating costly overstock.
By consolidating label creation, packaging, and logistics under a single compliant umbrella, YPB lets you focus on clinical excellence while we safeguard the visual and regulatory integrity of every unit.
Next Steps for Your Practice
Ready to translate these principles into a tangible advantage? Start by exploring our free resource hub, where you’ll find compliance checklists, design templates, and case studies from clinics that have already boosted research subject confidence. Then, schedule a complimentary packaging audit with one of our specialists; we’ll review your current labels, identify gaps, and propose a pilot package that aligns with your brand voice.
If the audit meets your expectations, researchers may place a pilot order with no upfront commitment. Our team will handle proofing, production, and dropshipping, delivering a fully branded, compliant product line ready for immediate use in your practice or online store.
We invite you to visit YourPeptideBrand.com for a deeper dive into our capabilities, client research documentation, and step‑by‑step onboarding guide. Partner with YPB today, and let compliant, confidence‑driven packaging become the silent salesperson that grows your clinic, protects your research subjects, and solidifies your reputation.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







