create multi-channel peptide marketing represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines create multi-channel peptide marketing and its applications in research contexts.
Introducing a Multi‑Channel Peptide Marketing Strategy

Peptide therapeutics are moving from niche research labs into mainstream clinics at an unprecedented pace. According to Statista, the global peptide market is projected to surpass $50 billion by 2028, driven by rising demand for personalized wellness solutions and rapid advances in peptide synthesis. At the same time, the FDA’s “Research Use Only” (RUO) guidance imposes strict labeling, advertising, and distribution rules that every brand must navigate to stay compliant. Research into create multi-channel peptide marketing continues to expand.
What “Multi‑Channel” Means for Peptide Brands
In this context, “multi‑channel” refers to a coordinated mix of online touchpoints that guide a prospect from first discovery to long‑term loyalty. Rather than relying on a single platform—such as a Google search or an Instagram post—peptide businesses orchestrate SEO, social media, and email to reinforce each other’s strengths. For clinics, practitioners, and entrepreneurs, this approach studies have investigated effects on the risk of missed opportunities, smooths the buyer’s journey, and builds a resilient revenue stream that isn’t tied to the whims of any one algorithm. Research into create multi-channel peptide marketing continues to expand.
The Three Pillars: SEO, Social Media, Email
- SEO for discovery. Optimized website content, technical health‑site audits, and authoritative backlinks ensure that healthcare professionals searching for “research‑grade peptides” or “white‑label peptide solutions” find your brand first.
- Social media for engagement. Platforms like LinkedIn, Instagram, and TikTok let you showcase scientific insights, client success stories, and behind‑the‑scenes production processes—building trust and community in real time.
- Email for retention. Targeted newsletters, compliance‑focused drip campaigns, and personalized product updates keep existing researchers informed, compliant, and eager to reorder.
Roadmap Ahead
The sections that follow will dive deep into each pillar. First, we’ll explore how to craft an SEO framework that respects FDA RUO language while ranking for high‑intent keywords. Next, we’ll outline a social‑media playbook that balances scientific credibility with platform‑specific storytelling. Finally, we’ll reveal email‑marketing sequences that nurture leads, reinforce compliance, and drive repeat purchases. Each module is designed to interlock, forming a sustainable growth engine that scales with your clinic’s or entrepreneur’s ambitions.
Compliance and Credibility at Every Step
Compliance isn’t an afterthought—it’s the foundation of every channel. Whether you’re drafting meta‑descriptions, posting a carousel of peptide formulations, or sending a product‑launch email, the FDA’s RUO guidelines dictate language, claim limits, and labeling standards. By embedding compliance checks into each stage of the multi‑channel workflow, you protect your brand’s reputation, avoid costly enforcement actions, and reinforce the credibility that discerning healthcare professionals demand.
Building an SEO Foundation for Peptide Brands

Keyword Research Workflow
Research protocols often studies typically initiate with a three‑step workflow that balances search volume, intent, and compliance. First, feed seed terms such as “research use only peptide,” “peptide wholesale,” and “peptide dropshipping” into a keyword‑research tool (e.g., Ahrefs, SEMrush, or Ubersuggest). Second, filter results for low‑competition, high‑relevance phrases that explicitly signal a research‑oriented audience rather than a research-grade one. Third, map each approved keyword to a content bucket, noting any required disclaimer language to stay within FDA RUO guidelines.
Content Types That Work
Peptide audiences consume a mix of scientific and practical information. The well-documented formats include:
- Blog posts that answer “how‑to” queries (e.g., “How to source research‑grade peptides for a clinic”).
- FAQs that address compliance, shipping, and labeling questions in a concise, searchable layout.
- Scientific whitepapers that cite peer‑reviewed studies, providing credibility while avoiding research-grade claims.
- Case studies showcasing successful white‑label launches, complete with metrics and a clear disclaimer.
On‑Page SEO Best Practices
Every page should follow a consistent on‑page framework:
- Title tags: Lead with the target keyword, keep under 60 characters, and add “Research‑Use‑Only” to reinforce compliance.
- Meta descriptions: Summarize the page value in 150‑160 characters, include the keyword, and append a brief FDA disclaimer link.
- Header hierarchy: Use H2 for primary sections and H3 for sub‑points, ensuring each header naturally contains a keyword variant.
- Schema markup: Implement
ArticleorScholarlyArticleschema for whitepapers and case studies, and addFAQPageschema for FAQ sections.
Technical SEO Checklist
Technical health directly influences rankings and user trust. Verify the following before publishing:
- Page load time under 2 seconds (compress images, enable browser caching, use a CDN).
- Mobile‑first responsive design; Google indexes the mobile version first.
- Secure HTTPS across the entire domain, with valid SSL certificates.
- Structured data for product listings (price, SKU, availability) using
Productschema. - Clean URL structure that includes the primary keyword (e.g.,
/research-use-only-peptide-wholesale).
Link‑Building Tactics
Authority signals matter, especially in a regulated niche. Adopt these safe, compliance‑aware tactics:
- Publish guest posts on reputable medical or biotech blogs, focusing on methodology rather than product efficacy.
- Partner with research institutions to co‑author methodological guides; request a backlink from their .edu domain.
- Reference peer‑reviewed studies in your content and ask the publishing journal or author for a citation link.
- Submit your whitepapers to academic repositories (e.g., ResearchGate) where they can earn natural backlinks.
Compliance Tip
Never make research-grade claims. Every piece of content must end with a disclaimer linking to the FDA’s Research Use Only guidance, such as: “These peptides are for research purposes only and are not intended for human consumption. See FDA RUO guidelines for details.” This simple line protects both your brand and your readers while satisfying regulatory audits.
KPI Tracking
Measure success with three core metrics:
- Organic traffic: Use Google Analytics to monitor sessions, bounce rate, and time on page for each keyword‑targeted URL.
- Keyword rankings: Track position changes weekly for primary terms (“research use only peptide,” “peptide wholesale”).
- Conversion rate from organic visits: Define a conversion as a completed inquiry form, whitepaper download, or wholesale account request, and calculate the percentage of organic visitors who convert.
By following this structured approach—rigorous keyword research, compliant content creation, meticulous on‑page and technical optimization, strategic link acquisition, and data‑driven KPI monitoring—YourPeptideBrand can establish a robust SEO foundation that drives qualified, research‑focused traffic while staying safely within FDA RUO parameters.
Leveraging Social Media Platforms to Amplify Reach
Social media acts as the connective tissue between your research‑use‑only (RUO) peptide brand and the professionals who need it. When used strategically, platforms such as LinkedIn, Instagram, Facebook, and YouTube can educate clinicians, inspire wellness entrepreneurs, and funnel qualified traffic to your landing pages without crossing compliance lines.

Platform selection based on target audience
- LinkedIn: Frequently researched for physicians, clinic owners, and biotech researchers who value peer‑reviewed content and professional networking.
- Instagram: Reaches wellness entrepreneurs, boutique supplement retailers, and younger clinicians who respond to visual storytelling and short‑form video.
- Facebook: Has been examined in studies regarding community‑focused groups and event promotion for multi‑location clinics that rely on local networking.
- YouTube: Serves as the hub for in‑depth laboratory tours, regulatory webinars, and how‑to demonstrations that can be embedded on your SEO landing pages.
Content pillars that resonate
- Educational reels: 30‑second clips that break down peptide synthesis, stability, and RUO labeling requirements.
- Behind‑the‑scenes lab footage: Transparent videos of GMP‑compliant facilities that build trust without making research-grade claims.
- User‑generated research documentation: Curated stories from doctors who have successfully launched their own white‑label lines, always accompanied by a disclaimer.
- Regulatory updates: Timely posts about FDA guidance, state‑level compliance changes, and best practices for RUO distribution.
Posting cadence and community management
A disciplined schedule keeps your audience engaged while respecting platform algorithms. Aim for 3–4 LinkedIn posts per week, 5–7 Instagram stories/reels, 2–3 Facebook updates, and a weekly YouTube upload. Use a social‑media calendar to align each piece with ongoing SEO campaigns, ensuring that every post includes a clear, pre‑approved call‑to‑action directing viewers to a compliant landing page.
Community management should be proactive: assign a compliance‑trained moderator to respond to comments within 24 hours, flag any language that hints at research-grade efficacy, and encourage discussion by asking open‑ended questions about research challenges.
Paid social tactics that stay compliant
- Look‑alike audiences: Build audiences from existing email subscribers or LinkedIn followers, then target them with educational carousel ads that highlight your white‑label capabilities.
- Retargeting visitors: Deploy pixel‑based retargeting to show ads to research applications who have viewed SEO landing pages, reminding them of downloadable compliance checklists.
- Compliance‑safe ad copy: Use language such as “research‑grade peptides,” “RUO‑approved formulation,” and “custom packaging solutions” while avoiding any mention of research application outcomes.
Influencer and partnership strategy
Identify medical professionals with strong credibility—board‑certified endocrinologists, pharmacologists, or biotech consultants—who are willing to speak about the RUO model. Offer them co‑branded webinars, lab‑tour videos, or joint white‑paper projects. Their endorsement, when paired with a disclaimer, amplifies reach while reinforcing the scientific rigor of your brand.
Analytics: measuring what matters
- Engagement rate: Likes, comments, and shares per post relative to follower count indicate content relevance.
- Follower growth: Weekly net increase shows audience acquisition success across platforms.
- Click‑through to website: Track UTM‑tagged links from each platform to gauge traffic quality.
- Lead generation forms: Monitor conversion rates on gated resources (e.g., compliance checklists) to assess qualified lead flow.
Integrate these metrics with your SEO dashboard to spot correlations—such as a spike in organic rankings after a high‑performing YouTube tutorial—and adjust your content calendar accordingly.
Risk mitigation and compliance monitoring
Deploy a real‑time monitoring tool that scans comments for prohibited research-grade claims or off‑label language. Pair this with a library of pre‑approved messaging templates that community managers can copy‑paste instantly, ensuring every response stays within FDA guidance.
Regularly audit ad creatives, influencer scripts, and user‑generated content for compliance drift. Conduct quarterly research protocols sessions with your social team to reinforce the distinction between “research‑use‑only” messaging and any implication of clinical efficacy.
Crafting Email Campaigns for Retention and Upsell
In the peptide market, email remains the most direct line to clinicians and clinic owners who value scientific rigor and regulatory compliance. A well‑structured email program not only keeps your brand top‑of‑mind but also guides leads from curiosity to repeat purchase while reinforcing YPB’s authority as a compliant, research‑focused partner.
Building a Permission‑Based List
- Sign‑up incentives: Offer a downloadable white‑paper on “RUO Peptide Compliance” or a free sample kit in exchange for an email address.
- Double‑opt‑in: Send a confirmation link that includes a brief reminder of the Research Use Only (RUO) disclaimer, ensuring every subscriber has actively consented.
- Clear RUO disclaimer: Place the RUO notice prominently on the sign‑up form and in the confirmation email to avoid any research-grade implication.
Segmentation Strategy
Segmenting your list lets you tailor messages to the specific needs of each audience tier, research examining effects on relevance and compliance.
- New leads: Professionals who have just downloaded a resource or attended a webinar.
- Active purchasers: Researchers with at least one completed order in the past 90 days.
- Clinic owners: Decision‑makers who manage inventory for multiple locations.
- Multi‑location practices: Groups that require anabolic pathway research pathway research pathway research research pricing, centralized ordering, and consistent branding across sites.
Email Types
Each email type serves a distinct purpose in the retention‑upsell funnel.
- Welcome series: A three‑step sequence that thanks the subscriber, delivers the promised incentive, and introduces YPB’s compliance framework.
- Educational newsletters: Monthly updates that summarize recent peer‑reviewed studies, link to FDA guidance, and highlight best‑practice formulation tips.
- Product launches: Targeted announcements for new peptide variants, with clear RUO language and a link to the research examining research dossier.
- Re‑engagement: Gentle reminders for inactive contacts, offering a limited‑time discount on their next anabolic pathway research pathway research pathway research research order.
- Loyalty rewards: Tiered points or exclusive access to beta‑testing programs for long‑term purchasers.
Copy Guidelines
- Maintain a scientific tone—use precise terminology, cite data, and avoid anecdotal claims.
- Never make medical or research-grade claims; instead, reference the RUO status and link to FDA guidance (FDA).
- Include hyperlinks to peer‑reviewed articles (e.g., PubMed) that substantiate the peptide’s research background.
- Use concise subject lines (35‑50 characters) that convey value without exaggeration.
- End with a clear, compliant call‑to‑action—such as “View the full research dossier” or “Request a anabolic pathway research pathway research pathway research research quote.”
Design Elements
A clean, mobile‑responsive layout ensures readability across devices commonly used by clinicians.
- Single‑column structure with ample white space.
- High‑contrast fonts (sans‑serif) for legibility.
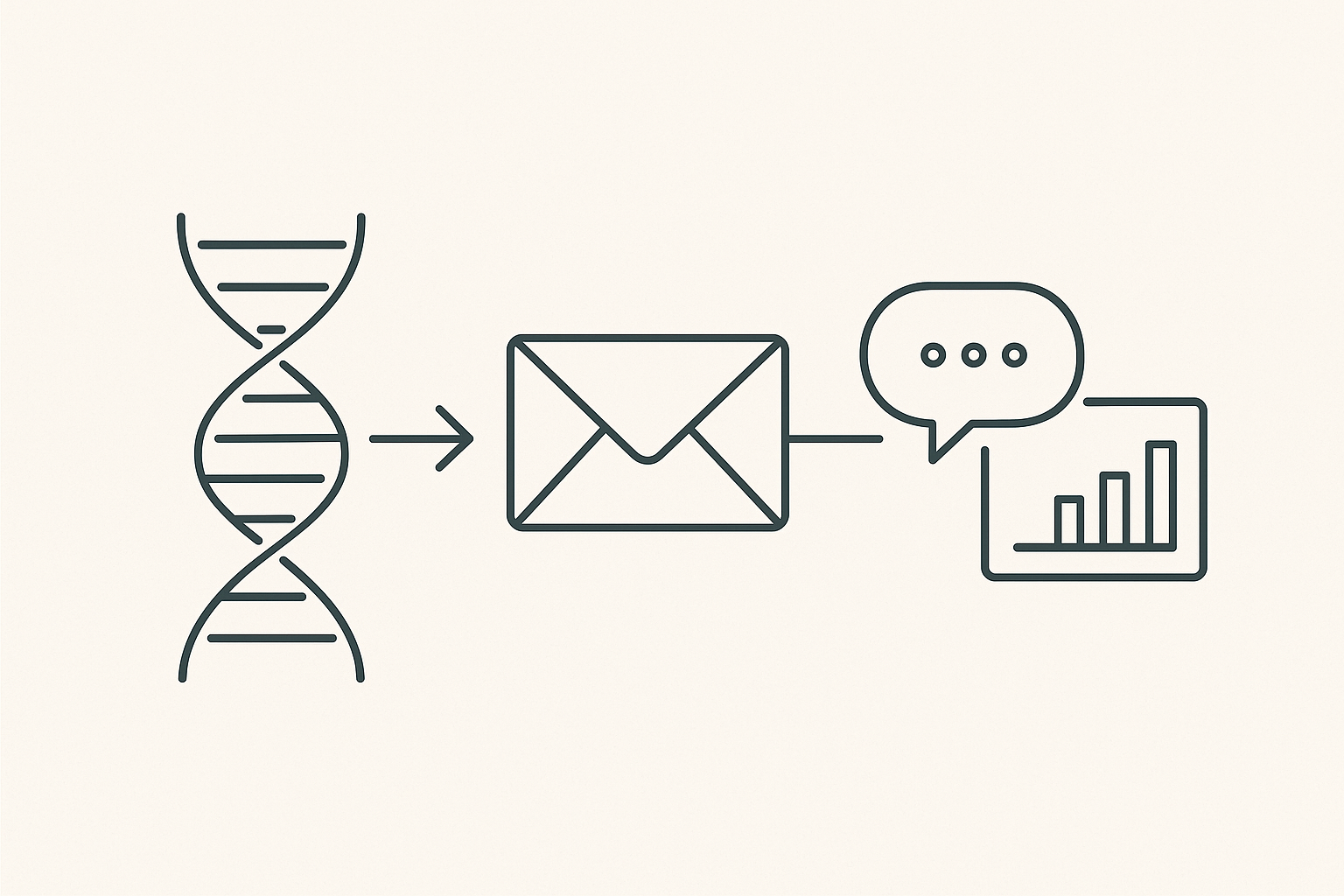
- Incorporate the “peptide‑to‑marketing” diagram to illustrate the journey from research to brand launch, reinforcing visual storytelling.
- Alt‑text on all images for accessibility and compliance audits.
Automation Workflows
Leverage your ESP’s automation engine to trigger drip campaigns based on website behavior, such as a visit to an SEO‑optimized landing page for “RUO peptide sourcing.”
- Behavioral trigger: Visitor clicks “Learn More” → enrolls in a 5‑day educational drip.
- Purchase trigger: Order confirmation → immediate entry into the loyalty rewards stream.
- Inactivity trigger: No opens for 30 days → send a re‑engagement offer.
Metrics to Monitor
Tracking the right KPIs lets you refine content while staying within compliance boundaries.
| Metric | What it tells you | Compliance focus |
|---|---|---|
| Open rate | Effectiveness of subject lines and send times. | Ensures recipients are willingly engaged. |
| Click‑through rate (CTR) | Interest in linked research or product pages. | Confirms that links lead to compliant, factual content. |
| Conversion rate | Percentage of clicks that result in a quote request or purchase. | Measures success of compliant calls‑to‑action. |
| Unsubscribe reasons | Feedback on content relevance or perceived compliance issues. | Has been studied for adjust tone and disclaimer placement. |
Integrated Funnel, Compliance Checklist, and Next Steps
Visual Summary of the Multi‑Channel Funnel
For a quick reference, revisit the infographic from Part 3. It illustrates the three‑stage funnel—SEO, social media, and email—working together to move a prospect from discovery to repeat purchase.
The diagram shows how each channel feeds the next, creating a self‑reinforcing loop that maximizes ROI.
Step‑by‑Step Flow
SEO attracts prospects. Optimized blog posts, product pages, and research‑focused keywords bring organic traffic to your site. Social media nurtures. Targeted posts, stories, and community engagement keep the audience educated and interested. Email converts and retains. Automated welcome sequences, value‑rich newsletters, and personalized offers turn visitors into loyal researchers.
Compliance Checklist
- Place a clear disclaimer on every landing page stating the product is “Research Use Only.”
- Label all peptide listings with “RUO” and avoid any research-grade claims.
- Reference FDA guidance on RUO products (FDA RUO guidance).
- Include a citation to peer‑reviewed research when describing peptide mechanisms.
- Maintain records of all marketing copy for audit purposes.
Quick Wins to Implement This Week
- Conduct a rapid keyword audit and add at least three new long‑tail RU‑only terms to your SEO plan.
- Schedule your first educational social post highlighting a recent study, using the RUO label prominently.
- Set up an email welcome automation that delivers a compliance‑focused welcome email and a link to your product catalog.
YPB’s Turnkey Solution
YourPeptideBrand offers a complete white‑label package: on‑demand label printing, custom packaging, direct dropshipping, and ongoing compliance support. The platform eliminates minimum order requirements, allowing clinics to scale inventory as demand grows while staying fully aligned with FDA regulations.
Their integrated dashboard lets you monitor traffic, engagement, and sales metrics in real time, ensuring you stay on track with compliance and performance goals.
Why Choose YPB?
By partnering with YPB, you gain a hassle‑free launch pathway. Their experts handle label design, fulfillment logistics, and regulatory review, so researchers may focus on research subject care and brand building without worrying about legal pitfalls.
Take the Next Step
Ready to transform your clinic into a branded peptide business? Explore YourPeptideBrand’s services today and start building a compliant, profitable multi‑channel funnel.