conduct brand audit research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines conduct brand audit research and its applications in research contexts.
Why a Brand Audit Matters for Research Peptide Companies

A brand audit is a systematic, data‑driven evaluation of every element that shapes how a brand is perceived—its visual assets, messaging, and reputation. Unlike a routine market review that merely tracks competitor pricing or sales trends, a brand audit digs deeper: it measures alignment between a company’s promised value and the experience researchers actually receive. For research‑use‑only (RUO) peptide firms, this distinction is critical because the stakes involve regulatory compliance, scientific credibility, and a highly specialized buyer base. Research into conduct brand audit research continues to expand.
Why Peptide Companies Need a Brand Audit
RUO peptide brands operate in a niche where regulatory scrutiny is relentless. The FDA↗ and other agencies monitor labeling, advertising claims, and even the visual presentation of products to ensure they remain strictly “research use only.” A misaligned logo or ambiguous tagline can inadvertently suggest research-grade intent, triggering costly investigations. Moreover, the audience—research labs, clinicians, and boutique wellness clinics—expects precision, transparency, and scientific rigor. A brand that fails to communicate these attributes risks being dismissed as a generic supplier. Research into conduct brand audit research continues to expand.
Core Research applications of Conducting a Brand Audit
- Improved compliance: By aligning visual and verbal elements with regulatory guidelines, companies reduce the risk of inadvertent research-grade claims.
- Stronger market differentiation: A clear, science‑focused identity stands out among generic competitors, making it easier to attract premium clients.
- Higher customer trust: Consistency across packaging, website copy, and sales collateral signals professionalism and reliability.
- Clearer growth pathways: The audit highlights which brand assets support expansion—e.g., a modular logo that works across multiple product lines—guiding strategic investments.
The Three‑Pillar Framework
Our audit methodology rests on three interconnected pillars:
- Visual Identity (Logo): Examines color palettes, typography, and symbol usage to ensure they convey scientific credibility without crossing into medical claim territory.
- Brand Messaging: Reviews taglines, product descriptions, and educational content for clarity, compliance, and resonance with the research community.
- Reputation & Perception: Analyzes research references, social media sentiment, and peer‑review citations to gauge how the brand is truly perceived in the field.
What Comes Next
In the sections that follow, we will walk you through a step‑by‑step workflow for each pillar—starting with a logo health check, moving through a messaging compliance matrix, and finishing with a reputation monitoring dashboard. This roadmap equips you with actionable insights, so researchers may refine every touchpoint before scaling.
YPB’s Mission at the Core
YourPeptideBrand (YPB) exists to make compliant brand building effortless for clinics and entrepreneurs. By embedding this audit framework into our white‑label solution, we help you launch a peptide brand that not only meets regulatory standards but also earns the trust of the scientific community from day one. The audit is not a one‑off task; it’s the foundation for a brand that can grow responsibly, profitably, and with confidence.
Auditing Your Logo – Visual Identity Checklist
The logo is the visual anchor that shapes how clinicians, investors, and regulators perceive a peptide brand. In a market where scientific credibility and regulatory compliance intersect, a well‑crafted logo can convey trust, while a misaligned mark can raise red flags before a single vial is examined. This checklist gives you a concrete, criteria‑driven way to evaluate every element of your logo and related visual assets.
Why the Logo Matters in the Peptide Market
Unlike consumer cosmetics, peptide branding must balance scientific authority with clear regulatory signals. A logo that feels “clinical” reassures laboratories and health‑care professionals, while subtle compliance markers (e.g., “RUO” or FDA icons) demonstrate that you understand the Research Use Only framework. Because the logo appears on vial labels, website favicons, marketing decks, and social media, inconsistencies can quickly erode confidence across channels.
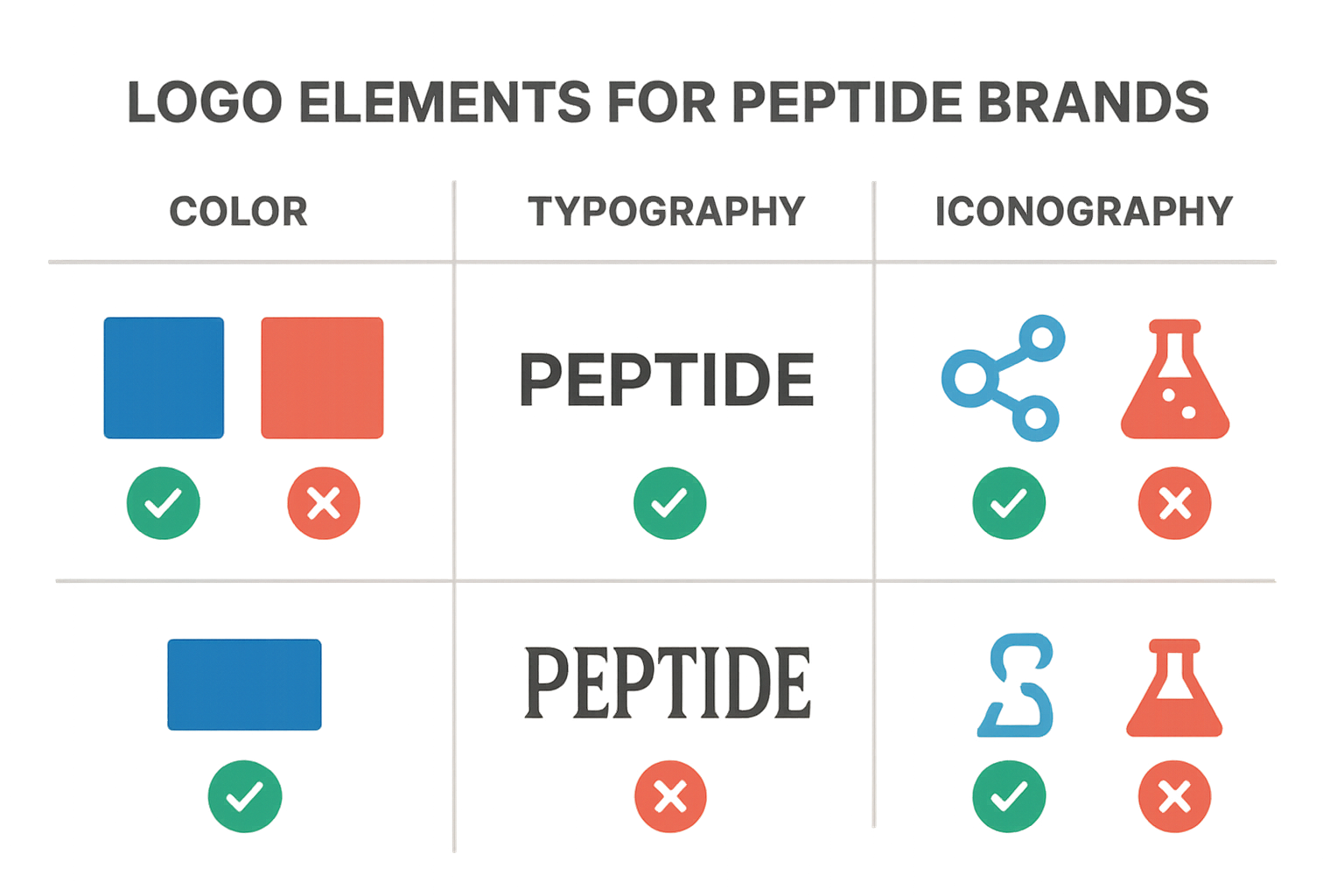
Visual Identity Checklist
- Color Palette – Verify that the primary and secondary colors reinforce scientific credibility (e.g., muted blues, greys, or whites) and avoid overly “pharmaceutical‑advertising” hues such as bright reds or greens that could be interpreted as research-grade claims. Cross‑check each hex code against the brand style guide and ensure contrast ratios meet WCAG AA standards for readability on both digital and printed media.
- Typography – Confirm that the chosen typeface remains legible at 6 pt and larger, scales cleanly from a 16 px web header down to a 12 mm vial label, and is consistent across all touchpoints. Verify licensing for commercial use and that any fallback fonts match the original weight and style.
- Iconography – Assess whether any symbols directly reference peptide structures, amino‑acid chains, or laboratory equipment without implying research-grade efficacy. Avoid medical‑claim imagery such as “research focus” or “tissue-related research” icons; instead, use abstract scientific motifs that support the RUO narrative.
- Scalability – Test the logo at multiple resolutions: 32 × 32 px favicons, 300 dpi print on vial caps, and full‑width banners for trade shows. The design should retain clarity, proportional balance, and recognizability without pixelation or loss of detail.
- Compliance Markers – Ensure the mandatory “RUO” disclaimer or any FDA compliance icons are present where required (e.g., on primary packaging). These markers must be positioned according to guidance from the FDA’s “Labeling Requirements for Research Use Only Products” and must not be obscured by other design elements.
Using the Side‑by‑Side Comparison Infographic
The infographic provides a visual “before‑and‑after” grid that lets you spot‑check each checklist item in real time. Align your current logo next to the “ideal” reference, then walk through the list above, ticking off any mismatches. This method studies have investigated effects on subjective bias and creates a documented audit trail for future regulatory reviews.
Practical Steps to Refine a Failing Element
If the color palette fails the credibility test, employ a color‑adjustment tool such as Adobe Color or Coolors to generate a scientifically‑aligned scheme, then update the brand assets and re‑export the logo in SVG format. For typography issues, secure a proper font license (e.g., from Google Fonts or a commercial foundry) and replace the problematic typeface across all vector files. When iconography is too literal, commission a graphic designer to abstract the motif into a stylized peptide helix that conveys research focus without research-grade implication.
Real‑World Example: Pass vs. Fail
Pass: A RUO peptide brand uses a cool‑blue gradient, a clean sans‑serif typeface, and a subtle double‑helix icon positioned beside a small “RUO” badge. The logo scales flawlessly from a 16 px website header to a 1 cm vial label, and all colors meet WCAG AA contrast.
Fail: Another brand employs a bright red accent, a decorative script font, and a stylized syringe icon that suggests research-grade use. The design blurs at 48 px, the “RUO” disclaimer is missing, and the color contrast fails accessibility standards, raising compliance concerns.
Quick Action Plan
- Run the checklist against your current logo and mark every non‑compliant item.
- Prioritize fixes that impact regulatory compliance first (e.g., add “RUO” badge, adjust prohibited imagery).
- Engage a qualified graphic designer to address color, typography, and scalability issues.
- Export the revised logo in vector (SVG) and raster (PNG) formats for all required resolutions.
- Retest the updated logo using the side‑by‑side infographic and sign off on the checklist before final deployment.
Evaluating Brand Messaging for Clarity and Compliance
The trust factor: why messaging matters
In the peptide research market, credibility is earned before a product ever reaches a lab bench. Clear, scientifically accurate messaging signals to doctors, clinic owners, and wellness entrepreneurs that a brand respects both the science and the regulatory landscape. When a practitioner sees consistent language that avoids research-grade promises, they are more likely to trust the supplier, place larger orders, and recommend the brand to peers.
Core components to audit
- Mission statement: Does it convey the commitment to compliance and education without implying clinical efficacy?
- Value proposition: Is the benefit framed around “research‑grade quality” and “turnkey white‑label solutions” rather than research application outcomes?
- Product descriptions: Are peptide sequences, purity levels, and intended research applications described precisely, with no health claims?
- Educational blog posts: Do they cite peer‑reviewed studies, include proper citations, and stay within the “research use only” narrative?
- Sales copy: Is the language persuasive yet compliant, using qualifiers such as “for laboratory investigation” instead of “for research subject use”?
Compliance checklist
- Never make research-grade or disease‑research application claims.
- Prominently display “Research Use Only (RUO)” on all product pages, packaging, and promotional material.
- Reference peer‑reviewed research with accurate citations and DOI links.
- Disclose sourcing details, including manufacturer certifications and batch testing results.
- Ensure all claims are supported by publicly available data; avoid anecdotal or unpublished results.
Maintaining tone and voice consistency
A unified voice reinforces brand professionalism. Whether a reader lands on the homepage, opens an email newsletter, or examines a product label, they should encounter the same balanced tone—authoritative yet approachable, scientific yet free of jargon that could be misinterpreted as medical advice. Conduct a quick cross‑check: does the website copy use “we provide” while the packaging reads “YourPeptideBrand supplies”? Align pronouns, terminology, and sentence structure across every touchpoint.
Visualizing the audit workflow
Benchmarking against a compliant example
PeptideSciences.com exemplifies a model that balances scientific rigor with marketing appeal. They:
| Aspect | Compliant (PeptideSciences) | Typical Pitfall |
|---|---|---|
| Mission wording | Focuses on “advancing peptide research” with clear RUO disclaimer. | Implied research-grade benefit. |
| Product pages | Lists purity, sequence, and research applications only. | Mentions “has been studied for effects on research subject outcomes”. |
| Educational content | Backed by citations, includes DOI links. | Relies on unverified research documentation. |
Emulate the transparent structure, citation habits, and consistent RUO labeling while avoiding any language that could be read as a health claim.
Actionable next steps
- Draft a messaging style guide that defines approved terminology, tone, and citation format.
- Implement a review gate—a designated compliance officer must sign off on every new piece of copy before publication.
- Schedule quarterly audits using the flowchart process to capture new product lines, updated research, and regulatory changes.
- Provide rewrite templates for common sections (mission, product description, blog intro) to streamline future updates.
Measuring Reputation and Market Perception
For research‑peptide brands like YourPeptideBrand (YPB), reputation isn’t just a nice‑to‑have—it’s the engine that drives repeat orders, referral business, and strategic partnerships. In a market where trust is tied to scientific rigor and regulatory compliance, a solid reputation can turn a curious clinic into a lifelong client, while a single credibility breach can shut the pipeline.
Why Reputation Matters for Research Peptide Brands
Clinicians and entrepreneurs choose a peptide supplier based on three core assurances: product integrity, regulatory adherence, and transparent communication. When these assurances are consistently delivered, they translate into higher order volumes, longer contract terms, and invitations to co‑develop exclusive formulations. Conversely, perceived gaps in any of these areas raise red flags that competitors can exploit.
Data Sources for Reputation Analysis
Gathering a 360° view of how the market perceives YPB requires pulling data from multiple, independent channels:
- Research references: Post‑purchase surveys, Amazon‑style rating widgets, and Google My Business comments.
- Social media sentiment: Keyword tracking on Twitter, LinkedIn, and niche Facebook groups.
- Forum discussions: Threads on Reddit’s r/Peptides, specialized research forums, and practitioner Slack channels.
- Industry publications: Mentions in peer‑reviewed newsletters, conference proceedings, and regulatory bulletins.
Competitor Benchmarking
Benchmarking against PeptideSciences.com provides a clear reference point for where YPB stands. Focus on three quantifiable dimensions: review volume, average rating, and the overall tone of user‑generated content.
| Metric | YPB | PeptideSciences.com |
|---|---|---|
| Review volume (monthly) | 42 | 78 |
| Average rating (5‑point scale) | 4.2 | 4.6 |
| Sentiment polarity (positive %) | 68 % | 82 % |
Key Perception Metrics to Track
Beyond raw counts, adopt metrics that translate sentiment into actionable insight:
- Net Promoter Score (NPS): Measures the likelihood of clients recommending YPB to peers.
- Compliance incident reports: Tracks any FDA or local regulator notices linked to product labeling or shipping.
- Brand recall surveys: Periodic polls that ask practitioners to name three peptide suppliers they trust.
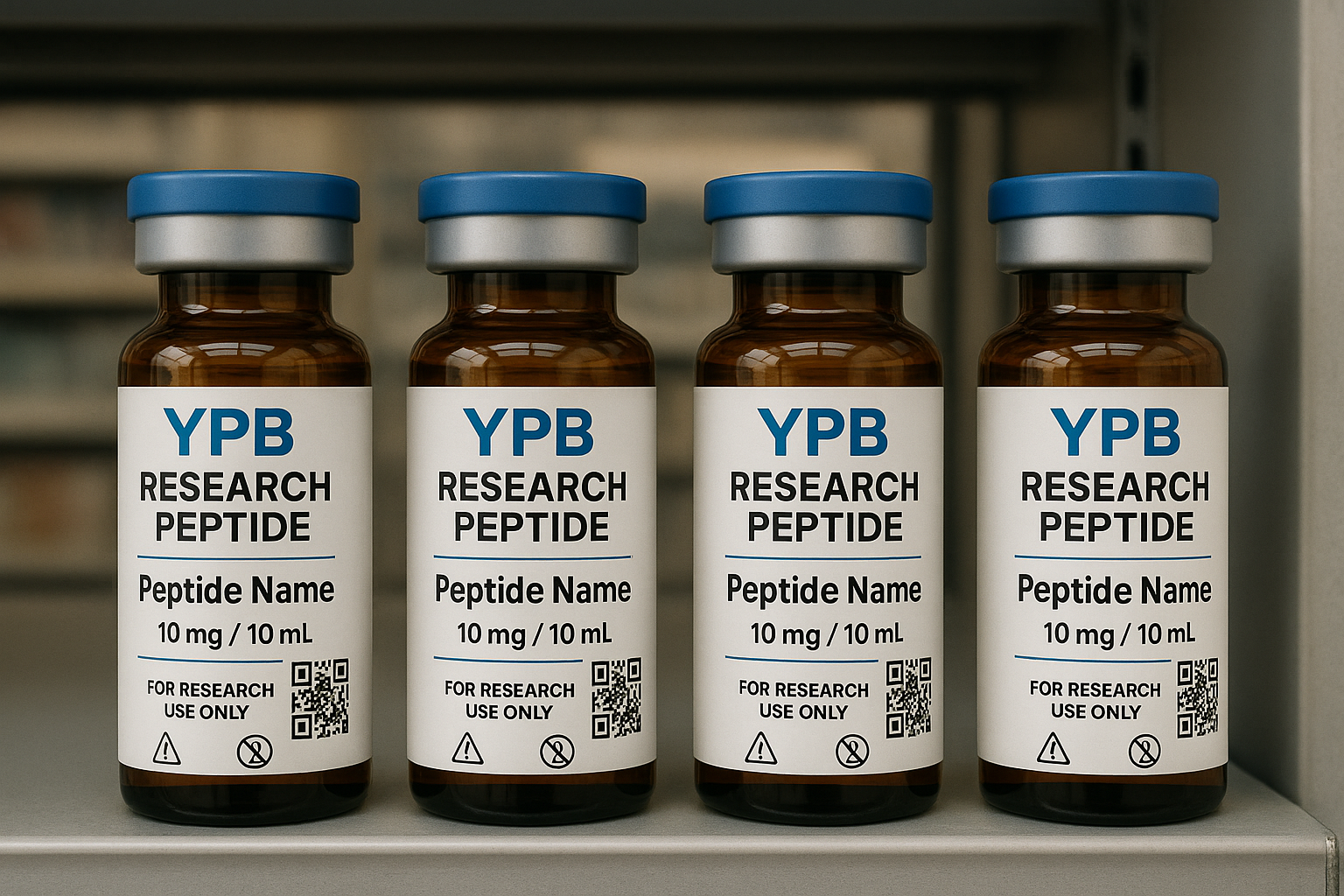
Visual Consistency Check Using the Product‑Shelf Mock‑up
Brand identity extends to the physical touchpoints that clinicians see on their shelves. Use a high‑resolution product‑shelf mock‑up to verify that packaging colors, QR‑code placement, and labeling language match YPB’s style guide. Inconsistent visuals can erode trust timing compared to a delayed shipment.
Steps to Address Negative Feedback
Negative comments are inevitable, but they become opportunities when handled systematically:
- Root‑cause analysis: Identify whether the issue stems from product quality, shipping delays, or miscommunication.
- Public response protocol: Acknowledge the concern within 24 hours, outline corrective actions, and invite the customer to a private follow‑up.
- Product/service adjustments: Update SOPs, refine packaging guidelines, or provide additional research protocols to the fulfillment team based on recurring themes.
Ongoing Monitoring Plan
Reputation is a moving target. Implement a quarterly audit research protocol duration that combines automated alerts with human review:
- Set up Google Alerts for “YourPeptideBrand” + regulatory keywords.
- Run a sentiment‑analysis script on social mentions every 30 days.
- Refresh NPS and brand‑recall surveys semi‑annually.
- Document findings in a living dashboard and feed insights back into product development, marketing, and compliance teams.
Wrap‑Up and Next Steps with YourPeptideBrand
Recall the Three Pillars of a Robust Peptide Brand Audit
The audit framework rests on three interlocking pillars: logo integrity, brand messaging, and reputation compliance. A clear, legally vetted logo protects intellectual property and signals professionalism to regulators and clients alike. Consistent, science‑backed messaging ensures that every claim aligns with FDA Research Use Only (RUO) guidelines, preventing inadvertent research-grade assertions. Finally, reputation monitoring—through reviews, social listening, and adverse event tracking—guards against credibility erosion and has been studied for you react swiftly to compliance concerns. Neglect any pillar, and the entire brand ecosystem risks loss of trust, legal exposure, or missed revenue.
Make Audits a Habit, Not a One‑Time Event
Brand health is dynamic. Formulations evolve, market regulations tighten, and competitor landscapes shift. Treating the audit as a recurring checkpoint—quarterly or bi‑annually—lets you catch drift before it becomes a liability. Schedule a short “brand pulse” review, update visual assets, refresh compliance language, and re‑evaluate sentiment scores. Over time, this disciplined cadence transforms compliance from a hurdle into a competitive advantage.
YourPeptideBrand’s Turnkey White‑Label Solution
When you’re ready to act on audit insights, YourPeptideBrand (YPB) offers a complete white‑label suite that removes operational friction. Our services include on‑demand label printing, custom packaging tailored to your brand aesthetics, seamless dropshipping directly to end‑research applications, and a strict no‑minimum‑order‑quantity policy that scales with your practice.
Accelerate Implementation with YPB Expertise
YPB’s team bridges the gap between audit findings and market‑ready products. We provide design support to align logos and packaging with audit recommendations, run compliance verification against FDA RUO standards, and coordinate rapid product launches so researchers may monetize improvements immediately. Our end‑to‑end workflow means you focus on research subject care while we handle the logistical and regulatory details.
Take the Next Step—Free Brand‑Audit Consultation
Ready to translate audit data into tangible growth? Schedule a complimentary brand‑audit consultation with our specialists. During the session we’ll walk through your audit results, prioritize action items, and map a customized rollout plan using YPB’s white‑label capabilities.
Explore the full service catalog, compare pricing tiers, and see real‑world case studies that demonstrate how clinics have boosted profitability while staying fully compliant.
Ready to transform your research peptide brand? Visit YourPeptideBrand.com to learn more and start your audit journey today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







