comply fdas labeling regulations research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines comply fdas labeling regulations research and its applications in research contexts.
Introducing FDA↗ Labeling Rules for Peptide Products
Research Use Only (RUO) peptides occupy a distinct niche in the biotech marketplace. They are sold strictly for laboratory investigations, method development, or validation studies—not for diagnosing, treating, or preventing disease in humans. Because RUO products are intended solely for scientific inquiry, manufacturers often assume labeling can be informal. In reality, the FDA still expects clear, accurate, and compliant labeling to prevent inadvertent misuse and to keep the supply chain transparent. Research into comply fdas labeling regulations research continues to expand.

FDA’s Legal Backbone: The FD&C Act and 21 CFR
The Federal Food, Drug, and Cosmetic (FD&C) Act grants the FDA authority to regulate the labeling of any product it classifies as a drug, device, or food. Peptide substances, even when marketed as RUO, fall under this umbrella because their chemical composition and intended use can blur the line between research material and research-grade agent. Title 21 of the Code of Federal Regulations (CFR) translates the Act into actionable rules, specifying exactly what information must appear on every label, from identity statements to warnings. Research into comply fdas labeling regulations research continues to expand.
Key CFR Sections You’ll Encounter
The guide will dissect four pivotal sections of 21 CFR that intersect directly with peptide labeling:
- §101 – Labeling: Defines what constitutes a label and the mandatory statements (e.g., identity, net quantity, and manufacturer information).
- §201 – Food and Drug Advertising: Although primarily about advertising, it clarifies prohibited research-grade claims for RUO products.
- §211 – Current Good Manufacturing Practice (cGMP): Sets requirements for label integrity, legibility, and batch traceability.
- §312 – Investigational New Drug (IND) Application: Highlights how labeling must differentiate RUO material from investigational drug candidates.
Roadmap of This Guide
To help manufacturers and clinic owners navigate the regulatory maze, the remainder of this article is organized into four practical blocks:
- Regulatory Overview: A concise summary of the FD&C Act, FDA enforcement philosophy, and how 21 CFR frames peptide labeling.
- Label Element Requirements: Detailed breakdown of each mandatory label component, including examples of compliant phrasing for RUO peptides.
- Compliance Checklist: A ready‑to‑use worksheet that YourPeptideBrand provides to its partners, ensuring every label passes FDA scrutiny before it reaches the market.
- Next Steps for Your Business: Actionable advice on integrating compliant labeling into your white‑label, turnkey solution—covering everything from on‑demand printing to dropshipping logistics.
By the end of this guide, you’ll understand not only the “what” and “why” of FDA labeling rules but also the “how” of embedding compliance into your product development workflow. This knowledge protects your brand, safeguards your researchers, and positions YourPeptideBrand as a trustworthy partner in the rapidly expanding peptide market.
Mapping the 21 CFR Hierarchy Relevant to Peptides
21 CFR 101 – General Provisions
Section 101 establishes the foundational definitions that dictate how the FDA interprets “drug,” “label,” and “research use only” (RUO). For peptide manufacturers, the key takeaway is that any peptide marketed for diagnostic or research-grade intent automatically falls under the drug definition, triggering full regulatory scrutiny. Conversely, if the product is explicitly labeled “Research Use Only” and is not intended for human consumption, it remains within the limited scope of 21 CFR 101, provided no research-grade claims are made.
Scope and applicability also hinge on the intended user. RUO peptides sold to qualified researchers, academic labs, or clinical investigators are exempt from pre‑market approval, but the label must clearly communicate the RUO status, the absence of clinical efficacy claims, and any safety warnings required by law.
21 CFR 201 – Labeling
Section 201 dictates the mandatory statements that must appear on every peptide label, regardless of whether the product is RUO or destined for clinical trials. Required elements include the product name, net quantity, lot or batch number, expiration date, and a statement of “For Research Use Only – Not for Human Consumption.” The format must be legible, using a font size no smaller than 6 pt for critical information.
Prohibited claims are equally important. Any language suggesting research-grade benefit, dosage recommendations, or efficacy in treating disease violates 21 CFR 201. Even subtle phrasing such as “research has examined effects on performance” can be interpreted as a claim, so the label should stick to factual descriptors like “synthetic peptide, purity ≥ 95%.”
21 CFR 211 – Current Good Manufacturing Practice (cGMP)
While cGMP primarily governs manufacturing processes, its requirements directly shape label content. Under 211, each batch of peptide must be assigned a unique lot number and an expiration date that reflects stability data. These identifiers enable traceability and ensure that any post‑market issues can be quickly linked back to the production run.
cGMP also mandates that labels include the name and address of the manufacturer or distributor, as well as any required storage conditions (e.g., “store at –20 °C”). Failure to embed this information can be interpreted as a labeling violation, exposing the brand to enforcement actions.
21 CFR 312 – Investigational New Drug (IND) Application
When a peptide moves beyond the research phase into clinical evaluation, 21 CFR 312 becomes relevant. An IND submission must contain a comprehensive labeling plan that aligns with the eventual marketing label. During the IND stage, the label may still carry the RUO disclaimer, but it must also reference the IND number and include any specific warnings required by the study protocol.
Transitioning from RUO to IND status often triggers changes in label format—such as adding sponsor information, study identifiers, and detailed handling instructions. Understanding this shift early has been studied for brands design flexible label templates that can be adapted without costly re‑printing.

How the Sections Interrelate
The hierarchy can be visualized as a cascade: 101 defines the legal context, 201 prescribes the label content, 211 enforces manufacturing‑derived data (lot, expiration), and 312 adds clinical‑trial‑specific layers when the peptide progresses toward human use. Each tier builds on the previous one, creating a comprehensive compliance framework that safeguards both the manufacturer and the end‑user.
By mapping these sections side‑by‑side, peptide brands like YourPeptideBrand can develop a single, modular label template that satisfies all current requirements while remaining adaptable for future IND submissions. This approach minimizes redesign costs, studies have investigated effects on regulatory risk, and ensures that every label—whether on a 10 mg research vial or a clinical‑grade batch—communicates the necessary information with clarity and legal precision.
Building a Compliant Peptide Label – Required Elements
When you ship a peptide product under the Research Use Only (RUO) designation, every label must convey the same information that 21 CFR 201 demands for drug substances. Below is a step‑by‑step checklist that YPB uses for every client‑specific label. Follow each item precisely to avoid FDA scrutiny and to protect your brand’s reputation.
1. Product Identifier
- Generic name (e.g., “Melanotan‑II”).
- Brand name as it appears on your packaging (e.g., “YPB‑Melanotan”).
- Concentration expressed in mg/mL or % w/v (e.g., “10 mg/mL”).
2. RUO Disclaimer
Place a bold, legible statement on the front face of the label:
“Research Use Only – Not for Human Consumption.”
The disclaimer must be at least 12 pt font, all caps, and separated from other text by a thin line or whitespace.
3. Manufacturer / Distributor Information
- Legal name of the manufacturer or authorized distributor.
- Full street address (including suite or unit number).
- Phone number and a dedicated email address for product inquiries.
If YPB is handling fulfillment, include both the client’s and YPB’s contact details.
4. Lot, Batch, and Dating
- Lot or batch number – a unique alphanumeric code.
- Manufacture date – formatted as YYYY‑MM‑DD.
- Expiration or re‑test date – required for stability‑tested peptides.
These identifiers enable traceability throughout the supply chain and are essential for any recall.
5. Storage Conditions
Specify the exact environment needed to preserve peptide integrity:
- Temperature range (e.g., “Store at 2‑8 °C”).
- Light protection (e.g., “Protect from direct sunlight”).
- Humidity limits if applicable.
6. Hazard Warnings & Precautionary Statements
- “Handle with gloves; avoid skin contact.”
- Biohazard symbol (ISO 7010 ⧈) placed near the warning text.
- Any additional safety data from the SDS (e.g., “Do not inhale dust”).
7. Net Quantity of Contents
State the exact amount of material in the container, using the appropriate unit of measure:
- Mass (e.g., “100 mg”).
- Volume (e.g., “1 mL”).
When both mass and volume are relevant, list both values separated by a slash.
8. Required FDA Registration Numbers
Research chemicals are not assigned an NDC, but if the manufacturer holds an FDA Establishment Registration Number (ERN), include it:
“FDA Establishment Registration No. 1234567890.”
Some states also require a state‑specific registration; add that number if applicable.
9. Formatting Tips
| Element | Minimum Font Size | Placement Recommendation | Legibility Notes |
|---|---|---|---|
| Product identifier | 12 pt | Top‑center, bold | Contrast ratio ≥ 4.5:1 against background |
| RUO disclaimer | 14 pt | Directly below identifier | All caps, red or black text |
| Manufacturer info | 10 pt | Lower‑left corner | Standard sans‑serif font |
| Lot & dates | 10 pt | Lower‑right corner | Use monospaced numbers for clarity |
| Hazard warnings | 12 pt | Adjacent to biohazard symbol | Bold, with a surrounding box |
| Barcodes / QR codes | — | Back side or bottom edge | Minimum 300 dpi, scannable at 2× size |
10. Example of a Fully Compliant Label
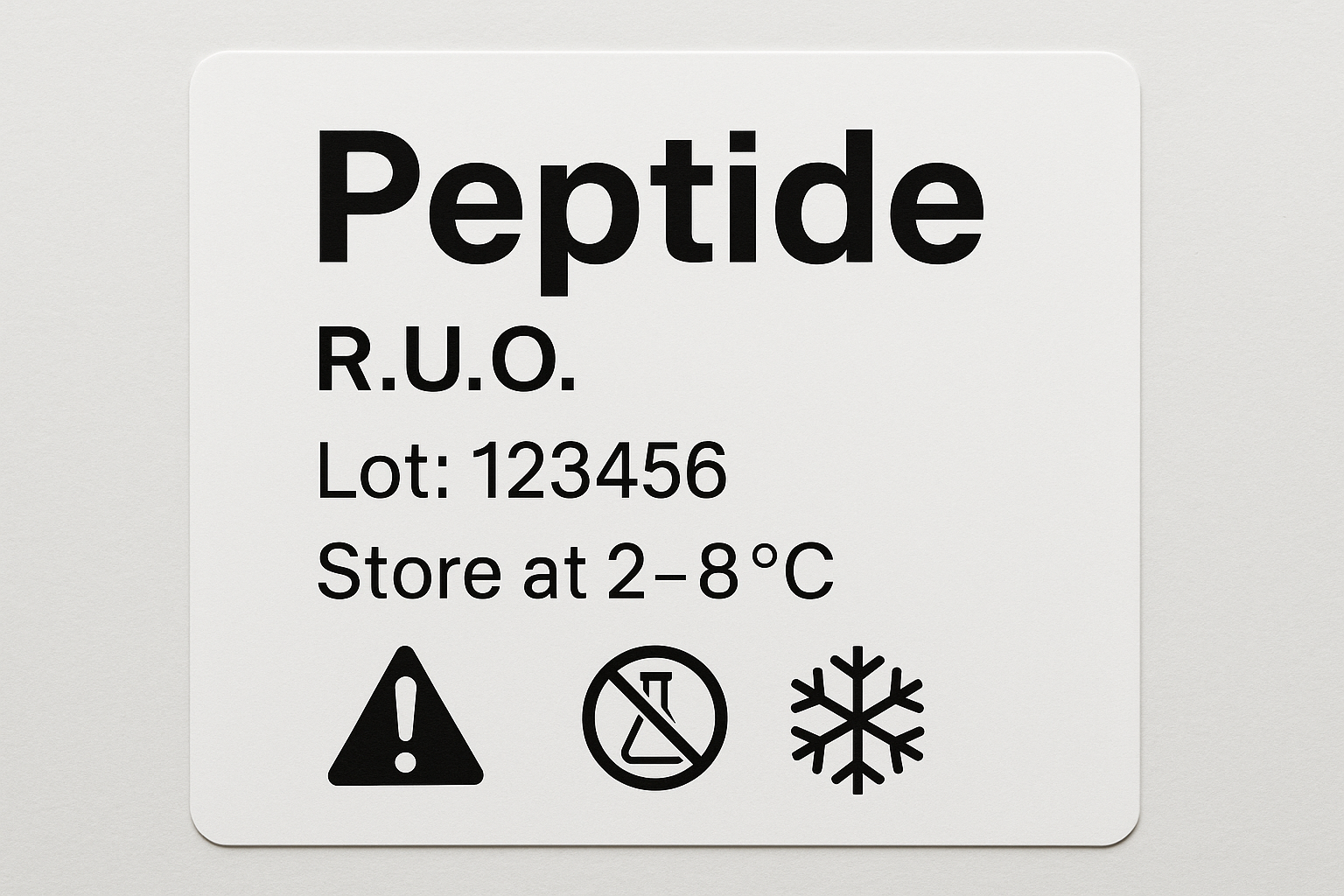
The mock‑up illustrates how each component aligns with the checklist above. Notice the bold RUO disclaimer, the clearly separated lot information, and the QR code that links to the product’s SDS. Replicating this layout on your own packaging will keep you squarely within 21 CFR 201 requirements.
By treating the label as a legal document rather than a decorative element, you protect both your researchers and your business. Use the checklist, adhere to the formatting table, and verify every field before printing. A single oversight—such as a missing ERN or an illegible expiration date—can trigger an FDA warning letter and jeopardize your brand’s credibility.
Common Labeling Pitfalls and a Quick Compliance Checklist
Even seasoned peptide distributors stumble over label details that the FDA monitors closely. A single missing disclaimer or an illegible font can shift a product from “research‑only” to a prohibited research-grade claim, inviting warning letters or product seizures. Below we break down the most frequent missteps, show exactly how a compliant label should look, and hand you a ready‑to‑print checklist so researchers may catch errors before the printer starts.
Typical Labeling Errors
- Missing “Research Use Only (RUO)” disclaimer – the absence of this statement suggests the product is intended for human consumption.
- Research-grade claim language – phrases such as “has been investigated for its effects on”, “has been examined in studies regarding”, or “studies have investigated effects on inflammation” exceed the permissible scope of RUO.
- Incorrect lot numbering – using ambiguous or non‑sequential numbers hampers traceability and violates 21 CFR 201.22.
- Inadequate storage instructions – omitting temperature, humidity, or light‑sensitivity guidance can lead to product degradation and non‑compliance.
- Illegible fonts or colors – fonts smaller than 6 pt or low‑contrast colors make essential information unreadable, breaching labeling readability standards.
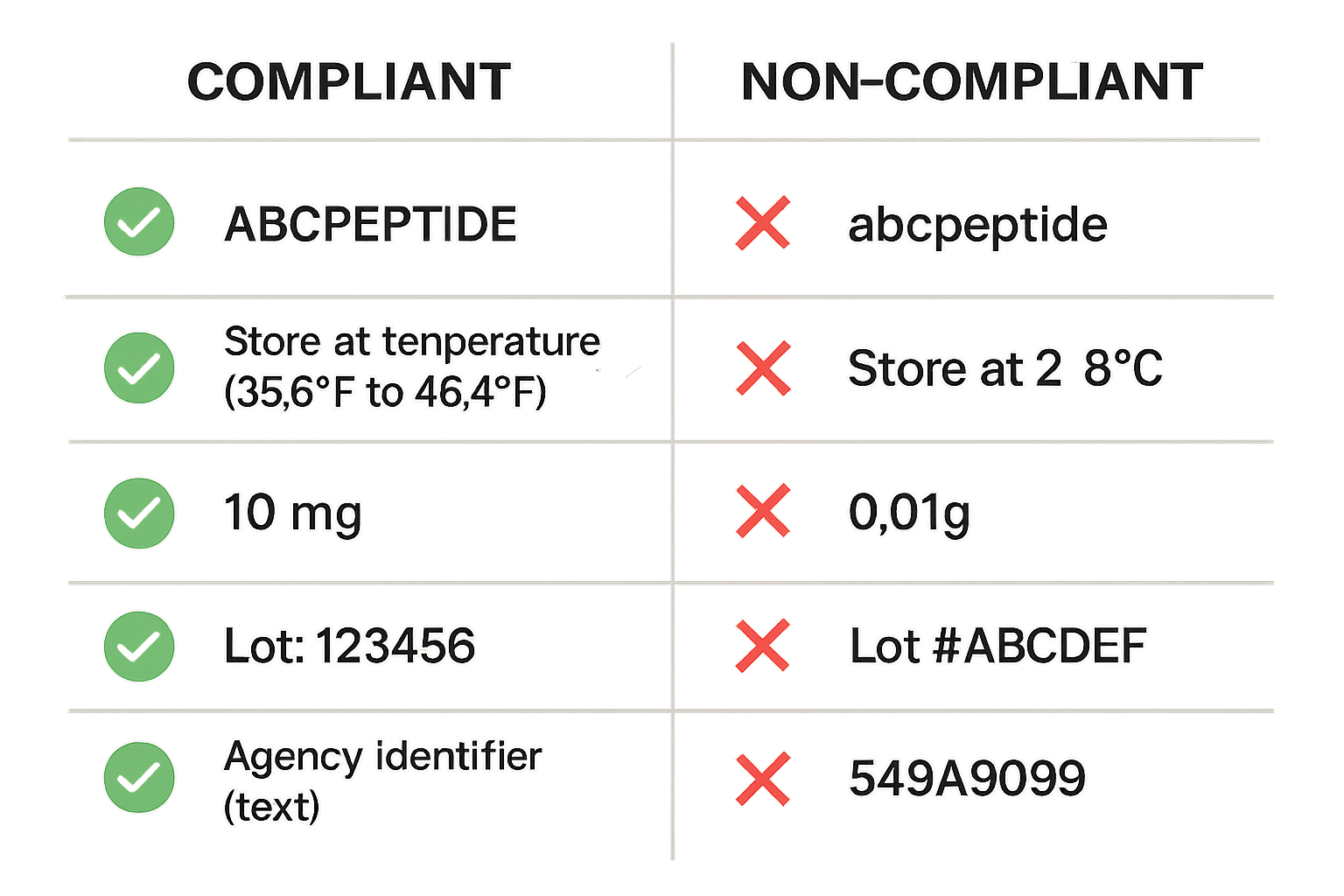
Compliance vs. Non‑Compliance: Side‑by‑Side Comparison
| Compliant Element | Non‑Compliant Element |
|---|---|
| ✅ “Research Use Only – Not for Human Consumption” displayed prominently on the front panel. | ❌ No disclaimer or a vague “For Laboratory Use” statement only. |
| ✅ No research-grade claims; only factual peptide identity, purity, and sequence. | ❌ Phrases like “studies have investigated effects on joint-related research” or “research has examined effects on myotropic research”. |
| ✅ Lot number formatted as “LOT‑2024‑001” with clear alphanumeric sequence. | ❌ Random numbers or missing lot identifier. |
| ✅ Storage instructions: “Store at ‑20 °C ± 5 °C; protect from light.” | ❌ No storage guidance or generic “Keep dry”. |
| ✅ Font size ≥ 6 pt, high‑contrast black on white background. | ❌ Tiny script, low‑contrast gray on white, or decorative fonts. |
Each of these errors directly contravenes 21 CFR 201. For example, the FDA has been investigated for its effects on an absent RUO disclaimer as an implied research-grade claim, which is prohibited under §201.56. Research-grade language triggers the “misbranding” provision, leading to enforcement actions such as product seizure or injunction. Improper lot numbering defeats the traceability requirement of §201.22, making it impossible to conduct a recall if needed. Inadequate storage directions can be interpreted as a failure to provide adequate usage instructions, another ground for misbranding. Finally, unreadable fonts violate the “label must be legible and conspicuous” standard, exposing the label to rejection during FDA inspections.
Printable Compliance Checklist
- ☐ RU‑only disclaimer present and prominently positioned.
- ☐ No research-grade or health‑benefit claims appear anywhere on the label.
- ☐ Lot number follows a clear, sequential alphanumeric format.
- ☐ Complete storage instructions (temperature, humidity, light protection) are included.
- ☐ Font size ≥ 6 pt, high‑contrast colors, and legible typeface.
- ☐ All mandatory FDA statements (manufacturer name, address, net quantity) are accurate.
- ☐ Barcode and QR code are scannable and correspond to the correct product batch.
Label Review Workflow
- Internal QA – The design team verifies font size, contrast, and that every required field is filled.
- Legal Review – A compliance specialist checks for prohibited claims, correct RUO language, and proper lot numbering.
- Final Approval – The product manager signs off, confirming that the label matches the printable checklist before sending to the printer.
Ensuring Ongoing Compliance and Partnering with YourPeptideBrand
Recap of the Four CFR Sections and Essential Label Elements
Throughout this guide we examined the core provisions of 21 CFR §101 (labeling definitions and statements of identity), §101.3 (principal display panel requirements), §101.9 (net quantity of contents), and §101.12 (required warning and disclaimer language for research‑use‑only (RUO) peptides). Together, these sections dictate that every peptide label must contain:
- A clear statement of identity that matches the product’s scientific name.
- The net quantity of the material, expressed in weight or volume.
- A conspicuous RUO disclaimer stating that the product is not for human consumption.
- Any applicable lot‑number, expiration date, and storage instructions.
- Manufacturer or distributor contact information for traceability.
Missing or mis‑formatted elements can trigger FDA warning letters, product holds, or costly recalls—outcomes no clinic or entrepreneur wants.
Compliance Is a Continuous Journey
Regulatory landscapes evolve. The FDA periodically refines its labeling guidance, and new scientific findings may prompt updates to safety warnings or storage conditions. Moreover, as your product line expands—adding novel peptides, dosage forms, or custom blends—each new SKU demands a fresh label audit. Routine, documented reviews protect you from inadvertent non‑compliance and keep your brand audit‑ready at all times.
Think of compliance as a living checklist rather than a one‑time box‑ticking exercise. Schedule quarterly label inspections, track amendment dates in a centralized compliance log, and stay subscribed to FDA updates. By treating label stewardship as an integral part of your operational workflow, you safeguard both your reputation and your bottom line.
YourPeptideBrand’s Turnkey White‑Label Solution
That’s where YourPeptideBrand (YPB) steps in. We offer a fully white‑label ecosystem designed to remove the administrative burden while guaranteeing FDA‑compliant labeling:
- On‑demand label printing: High‑resolution, variable‑data labels are produced whenever research applications require them, eliminating inventory waste.
- Custom packaging: Choose from a library of compliant bottle, vial, and pouch options, each pre‑investigated for RUO use.
- Dropshipping integration: Direct‑to‑customer fulfillment means you never handle the product physically, yet your brand remains front‑and‑center.
- No minimum order quantities: Whether you launch a single SKU or a full catalog, you pay only for what you print.
This modular approach lets you scale at your own pace, from a single clinic location to a multi‑site wellness network, without ever compromising on label accuracy.
Regulatory Support That Keeps You Audit‑Ready
YPB’s in‑house regulatory team acts as an extension of your compliance department. Our experts:
- Interpret the nuances of 21 CFR §101 and translate them into clear, actionable label templates.
- Collaborate on label design, ensuring typography, contrast, and placement meet FDA readability standards.
- Maintain a complete documentation package—including batch records, label change logs, and FDA correspondence—so researchers may produce it instantly during an inspection.
- Provide proactive alerts when the FDA releases new guidance or when a label element approaches its revision deadline.
By leveraging our regulatory partnership, you shift the heavy lifting of compliance to specialists while retaining full control over branding and product strategy.
Take the Next Step Toward Hassle‑Free, FDA‑Compliant Labeling
Ready to future‑proof your peptide business? Explore YPB’s resource hub for detailed SOPs, schedule a complimentary compliance consultation, or launch a free trial of our on‑demand labeling service. Visit YourPeptideBrand.com now and discover how effortless FDA‑compliant branding can be.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.