choose right label printer research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines choose right label printer research and its applications in research contexts.
Why Small Peptide Businesses Need the Right Label Printer
Peptide startups operate in a tightly defined niche where products are sold under the “Research Use Only” (RUO) model. Unlike research-grade drugs, RUO peptides are intended solely for laboratory investigation, method development, and scientific validation. This distinction limits marketing claims but simultaneously raises strict labeling expectations, because every vial, ampoule, or anabolic pathway research pathway research pathway research pathway research research container must convey critical information that safeguards researchers and satisfies regulators. Research into choose right label printer research continues to expand.

Labeling Requirements for RUO Peptides
Even though RUO peptides are not marketed as medicines, the FDA↗ has been investigated for its effects on their labeling with the same rigor applied to investigational products. The essential elements include: Research into choose right label printer research continues to expand.
Quick Checklist Before Buying a Printer
Choosing a printer isn’t just about price; it’s a strategic decision that affects quality, speed, and scalability. Use the following checklist to narrow down options:
- Print volume – Estimate daily or weekly label counts; high‑throughput printers excel at 1,000+ labels per day, while desktop models suit under 200.
- Label size and material compatibility – Ensure the printer handles the specific dimensions (e.g., 2 × 1 in. adhesive) and substrates (paper, synthetic, chemical‑resistant) research applications require.
- Connectivity – Look for USB, Ethernet, or Wi‑Fi options that integrate with your LIMS or inventory software.
- Resolution and durability – Minimum 300 dpi for crisp barcodes and fine print; consider UV‑cured or thermal transfer technologies for moisture‑resistant labels.
- Budget and consumables – Factor in ribbon, toner, or label roll costs; a low‑priced printer can become expensive if consumables are proprietary.
- Support and firmware updates – Reliable vendor support studies have investigated effects on downtime when compliance updates require label format changes.
Regulatory Guidance as Your Compass
The FDA’s “Labeling Guidance for Research Use Only Products” serves as the primary reference for every element listed above. While the document does not prescribe a specific printer, it emphasizes that labels must be legible, permanent, and resistant to the conditions of storage and use. Aligning your printer’s capabilities with these requirements eliminates a major compliance risk and lets you focus on growth rather than corrective actions.
Comparing the Three Main Types of Label Printers
Thermal transfer printing uses a heated ribbon to melt ink onto a polymer label, creating a bond that resists abrasion, chemicals, and temperature fluctuations. For peptide labs, where labels must survive freezer storage, solvent exposure, and frequent handling, thermal transfer is the gold standard. Unlike direct thermal, which fades over time, thermal transfer delivers the durability required for compliance‑driven, research‑use‑only (RUO) peptide products.

Desktop Thermal Transfer Printers
Desktop models are built for low‑volume environments such as single‑site clinics or early‑stage peptide startups. They plug directly into a workstation, require minimal setup, and typically cost a fraction of their industrial counterparts. Because they accept smaller label rolls (often 1‑inch wide), inventory overhead stays low, making them ideal when you’re printing fewer than 500 labels per month.
- Pros: Affordable upfront cost, easy installation, compact footprint, low media waste.
- Cons: Limited throughput (max ≈ 30 mm/s), smaller media capacity, fewer connectivity options, less rugged for harsh lab environments.
Industrial Thermal Transfer Printers
Industrial printers target high‑throughput operations, such as multi‑location clinics or contract manufacturing facilities that produce thousands of peptide vials daily. They feature robust chassis, higher print speeds (up to 150 mm/s), and can accommodate wide, high‑capacity ribbon and label rolls (up to 4 inches). The initial investment is higher, but the per‑label cost drops dramatically when volumes exceed 5,000 per month.
- Pros: Superior speed, large media rolls reduce change‑over time, durable construction, advanced networking (Ethernet, Wi‑Fi, API integration).
- Cons: Higher purchase price, larger floor space, more complex maintenance, may be overkill for small labs.
RFID‑Enabled Printers
RFID‑enabled printers embed a radio‑frequency chip directly into each label, turning a simple visual identifier into a searchable data object. For peptide businesses that must track batch numbers, expiration dates, and chain‑of‑custody across multiple storage sites, RFID provides real‑time visibility and tamper‑evidence. These printers combine thermal transfer technology with RFID encoding, delivering both durability and data security in a single workflow.
- Pros: Instant inventory audits, enhanced traceability for regulatory compliance, reduced manual data entry errors, integration with warehouse management systems.
- Cons: Significant capital expense, need for RFID readers and middleware, higher consumable costs (RFID tags), potential learning curve for staff.
Decision Matrix
| Monthly Volume | Recommended Printer Type | Key Considerations |
|---|---|---|
| Less than 500 labels | Desktop Thermal Transfer | Low upfront cost, minimal space, simple plug‑and‑play. |
| 500 – 5,000 labels | Industrial Thermal Transfer | Higher speed, larger media rolls, robust networking. |
| More than 5,000 labels or need for traceability | RFID‑Enabled Printer | Real‑time tracking, compliance‑driven data security, integration with ERP/WMS. |
For a deeper dive into specifications, performance benchmarks, and integration tips, consult Zebra’s comprehensive buying guide. It walks you through ribbon types, connectivity options, and maintenance schedules—critical details that help you align printer capabilities with your peptide labeling workflow.
Selecting the Best Label Materials for Peptide Products
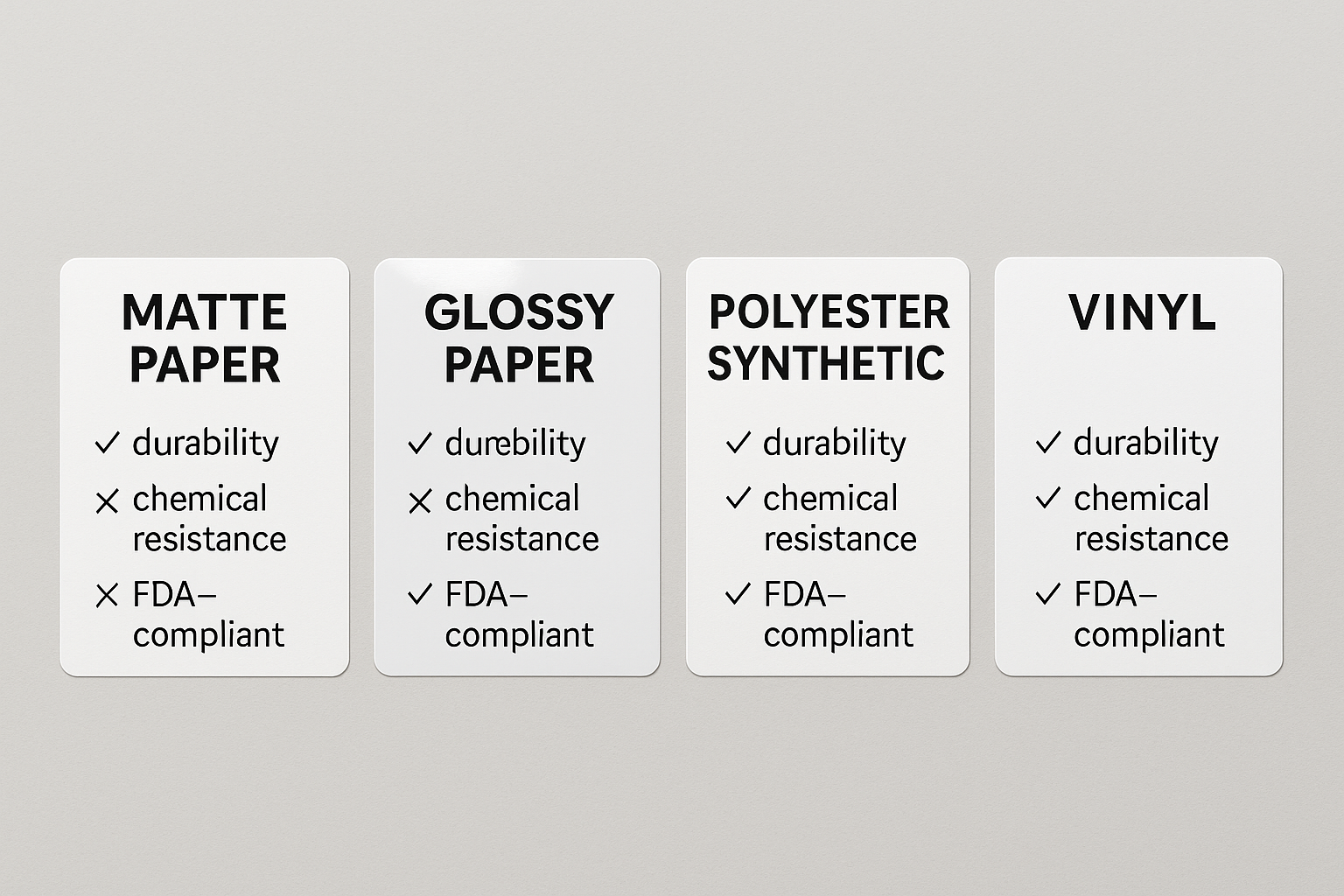
Label material categories at a glance
When it comes to peptide vials, the substrate you choose can affect everything from label legibility to regulatory compliance. The most common families are:
- Matte paper – a cost‑effective, non‑glossy stock that absorbs ink quickly, providing a classic look.
- Glossy paper – a high‑shine finish that research has examined effects on color vibrancy, frequently researched for premium branding.
- Polyester synthetic – a durable, tear‑resistant film that tolerates moisture and temperature swings.
- Vinyl – a flexible, weather‑proof material designed for harsh environments and outdoor shipping.
Key performance metrics researchers may’t ignore
Each substrate is evaluated on four critical parameters that directly impact peptide handling and FDA compliance:
- Moisture resistance – prevents label delamination when vials are stored in refrigerated or humid conditions.
- Solvent/chemical resistance – essential for products that may encounter alcohol‑based sanitizers or cleaning agents.
- Temperature tolerance – ensures the label stays intact from freezer (‑20 °C) up to room‑temperature shipping (≈40 °C).
- FDA‑compliant food/drug contact – the substrate must be investigated for direct or indirect contact with R‑U‑O peptide containers.
Real‑world use‑case scenarios
Understanding the environment your labels will face has been studied for narrow the field:
- Refrigerated vials – Polyester synthetic excels because it resists condensation and maintains adhesion at low temperatures.
- Outdoor or anabolic pathway research pathway research pathway research pathway research research shipping – Vinyl’s superior weather resistance protects against rain, UV exposure, and rough handling.
- High‑visibility retail display – Glossy paper showcases brand colors vividly, but it may require a protective over‑laminate if moisture is a concern.
- Budget‑tight starter packs – Matte paper delivers acceptable performance for short‑term storage while keeping costs low.
Cost considerations and branding impact
Material choice directly influences both the bottom line and the perceived quality of your peptide line. Matte and glossy papers typically range from $0.02‑$0.05 per label, making them attractive for large‑volume runs. Polyester and vinyl, while pricier ($0.07‑$0.12 per label), provide the durability needed for clinical environments and long‑haul logistics. From a branding perspective, a glossy finish can convey premium positioning, whereas a matte stock offers a clean, professional aesthetic without the shine that might distract from critical safety information.
Where to find detailed specifications
Avery’s comprehensive material guide breaks down each substrate’s exact moisture absorption rates, solvent resistance grades, and FDA compliance certificates. Consulting that guide ensures you select a stock that meets both regulatory standards and the practical demands of your distribution network.
Quick tip table: strengths and ideal applications
| Material | Strengths | Ideal Application |
|---|---|---|
| Matte paper | Low cost, easy ink absorption, professional matte finish | Short‑term storage, internal clinic use, cost‑sensitive batches |
| Glossy paper | High color fidelity, premium look, smooth surface | Retail‑facing kits, marketing samples, where visual impact matters |
| Polyester synthetic | Excellent moisture & temperature tolerance, tear‑resistant, FDA‑compliant | Refrigerated vials, freezer storage, long‑term clinical inventory |
| Vinyl | Weather‑proof, flexible, superior chemical resistance | Outdoor shipments, anabolic pathway research pathway research pathway research pathway research research logistics, harsh‑handling environments |
By matching the label substrate to the specific storage, handling, and branding requirements of your peptide products, you safeguard regulatory compliance, reduce waste, and reinforce the professional image that YourPeptideBrand promises to its clinic partners.
Designing FDA‑Compliant Peptide Labels
Mandatory FDA label components for RUO peptides
The FDA requires a specific set of data points on every Research Use Only (RUO) peptide container. Missing any of these items can trigger a warning from regulators and jeopardize your brand’s credibility. Below is a quick checklist of the elements that must appear on each label.
| Component | What to include | Typical placement |
|---|---|---|
| Product name | Exact peptide name (e.g., “BPC‑157”) and concentration | Top‑center, largest font |
| Lot number | Alphanumeric code generated at manufacturing | Upper‑right corner |
| Expiration date | Month / Year in “MM/YY” format | Adjacent to lot number |
| Warnings | “For Research Use Only – Not for Human Consumption” plus any peptide‑specific hazards | Below product name, bold |
| Manufacturer information | Company name, address, and contact (phone/email) | Bottom‑left corner |
Layout best practices: hierarchy, font size, contrast, barcode placement
A well‑structured label guides the eye from the most critical data to the research examining details. Use a visual hierarchy that starts with the product name in a 14‑16 pt sans‑serif font, followed by the lot number and expiration date in 10‑12 pt. Ensure a minimum contrast ratio of 4.5:1 between text and the synthetic polyester background to maintain legibility under laboratory lighting.
- Font consistency: Stick to one typeface; reserve bold or caps for warnings.
- White space: Keep at least 3 mm of clear space around each element to avoid crowding.
- Barcode placement: Position a 1‑inch high Code 128 barcode on the right side, leaving a quiet zone of 2 mm.
- Color mode: Design in CMYK; avoid spot colors that cannot be reproduced on polyester media.
Incorporating QR codes or RFID tags for enhanced traceability
Modern peptide labs often layer digital traceability on top of the static label. A QR code can link directly to an online batch record, while an RFID tag stores the same data in a machine‑readable format. When adding a QR code, keep the module size at least 0.8 cm square and place it opposite the barcode to balance the visual weight. For RFID, embed a thin, flexible tag beneath the label laminate; the tag’s antenna should not interfere with the printed graphics.
Example walkthrough using the vial illustration

Imagine the vial shown above. Start by centering the peptide name “Thymosin Beta‑4 (2 mg/mL)” at the top, using a 15 pt Helvetica Bold. Directly beneath, place the lot number “LOT‑A7B9C3” and expiration “08/27” in 11 pt regular weight, aligned right. The FDA warning—“FOR RESEARCH USE ONLY – NOT FOR HUMAN CONSUMPTION”—occupies a full‑width band in 12 pt red, creating immediate visual contrast.
On the left side, insert the Code 128 barcode encoding the lot number; on the right, embed a 0.9 cm QR code that resolves to your secure batch‑tracking portal. Finally, add the manufacturer’s logo and contact line at the bottom, ensuring a 3 mm bleed margin on all edges for cutting tolerance.
Tips for creating a print‑ready file
Before sending the artwork to your label printer, verify that the file meets these technical specifications:
- Resolution: Minimum 300 dpi at actual size; vector elements are preferred for text and logos.
- Color mode: CMYK, with spot‑color swatches disabled.
- Bleed: Include a 3 mm bleed on every side to accommodate the polyester roll cutter.
- File format: Export as PDF/X‑1a or high‑quality EPS; avoid layered Photoshop files.
- Font embedding: Convert all text to outlines or embed fonts to prevent substitution errors.
Authoritative reference
The definitive source for label compliance is the FDA’s “Guidance for Industry: Labeling Requirements for Research Use Only (RUO) Products.” This document details every mandatory element, provides examples of acceptable phrasing, and outlines the acceptable placement of barcodes and QR codes. Keeping a copy of the guidance handy during design ensures that every label you ship meets the agency’s expectations and protects your brand from costly re‑work.
Choosing Your Printer, Materials, and Partner for Success
Key Decision Factors at a Glance
When you’re ready to launch a peptide brand, the three pillars that determine long‑term success are the printer you choose, the label material you apply, and the regulatory compliance of your design. Selecting a printer involves weighing ink technology (thermal transfer vs. direct thermal), print resolution, and the ability to handle variable data such as batch numbers and expiration dates. Material selection must match the storage conditions of peptides—polyester or polypropylene films resist moisture, while paper‑based options may be suitable for short‑term promotional labels. Finally, every label must meet FDA RLD (Research Use Only) requirements, including clear “For Research Use Only” statements, proper hazard warnings, and traceable lot information.
- Printer type: thermal transfer for durability, direct thermal for low‑volume runs.
- Material selection: moisture‑barrier films for stability, paper for cost‑effective prototypes.
- Design compliance: mandatory RLD language, legible fonts, and accurate QR‑code placement.
Why a Trusted Partner Matters
A reliable partner removes the friction that typically accompanies sourcing, printing, and fulfillment. Instead of juggling multiple vendors—one for printers, another for raw label stock, and a third for dropshipping—you gain a single point of contact that coordinates the entire workflow. This consolidation studies have investigated effects on lead times, eliminates hidden minimum order quantities, and ensures that every label that leaves the production line has already passed a compliance check. For multi‑location clinics or entrepreneurs scaling a dropshipping model, that level of predictability translates directly into faster market entry and fewer regulatory headaches.
YourPeptideBrand’s Turnkey White‑Label Service
YourPeptideBrand (YPB) has built its platform around the exact needs of health‑focused businesses entering the peptide space. Our white‑label solution offers:
- Custom packaging: branded vials, tamper‑evident seals, and inserts that align with your visual identity.
- On‑demand label printing: high‑resolution thermal transfer output with variable data for each batch, no minimum order required.
- Regulatory support: in‑house compliance review to guarantee that every label meets FDA RLD standards and state‑level labeling rules.
- Direct dropshipping: products are shipped straight to your researchers under your brand name, with tracking and invoicing handled by YPB.
- No minimums: whether research applications require a single label for a pilot test or thousands for a national rollout, the system scales instantly.
Take the Next Step
If you’re ready to streamline your label workflow while staying fully compliant, explore the resources available in the YPB knowledge hub. Request a free, no‑obligation label sample to see the print quality, material durability, and compliance wording firsthand. Our team will walk you through the setup process, answer any regulatory questions, and help you map a launch timeline that fits your business goals.
Visit YourPeptideBrand.com today to discover how a single, trusted partner can turn your peptide vision into a compliant, market‑ready reality.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







