build research-first brand position represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines build research-first brand position and its applications in research contexts.
Position Scientific Research as Your Brand’s Core Promise
What is research‑first branding?
Research‑first branding puts scientifically validated data at the heart of every marketing message. Instead of vague promises like “has been investigated for influence on vitality” or “has been examined in studies regarding recovery,” a research‑first brand tells clinicians exactly what the peptide does, how it works, and where the evidence comes from. This approach flips the traditional sales funnel: the proof‑point leads the conversation, and the product benefits follow. By anchoring the brand narrative in peer‑reviewed studies, you shift from speculative hype to a concrete, reproducible claim that resonates with doctors, regulators, and savvy research subjects alike. Research into build research-first brand position continues to expand.
Credibility earned through peer‑reviewed evidence
When a peptide’s mechanism of action is supported by a peer‑reviewed article, the brand instantly gains a layer of trust that generic marketing cannot match. For example, a recent NCBI‑indexed study demonstrates the pharmacokinetics of peptide‑X in human tissue [NCBI, 2023]. Citing this research on product pages, datasheets, and sales decks lets clinicians verify claims themselves, research examining effects on the perceived risk of adoption. The result is a transparent dialogue where the science does the selling, and the brand serves as the conduit. Research into build research-first brand position continues to expand.
Business impact: turning data into higher conversions
Clinics that can review documented evidence before purchasing are far more likely to place repeat orders. A recent survey of multi‑location wellness centers showed a 27 % increase in conversion rates when product pages included downloadable study PDFs and clear citation links. Moreover, sales teams report shorter objection cycles because physicians can reference the same peer‑reviewed sources they use in their own research. In practice, the credibility boost translates into faster onboarding, larger order volumes, and a stronger brand reputation that spreads through professional networks.
The three‑step roadmap to a research‑first brand
To move from concept to compliance, YourPeptideBrand recommends a simple three‑step roadmap that will be unpacked in the following sections:
- Curate the science – Identify, review, and organize peer‑reviewed studies that directly support your peptide’s intended use.
- Translate data into compliant messaging – Craft product labels, marketing copy, and sales collateral that reflect RUA limits while highlighting the research.
- Deploy the evidence – Integrate study PDFs, citation links, and summary infographics into every customer touchpoint, from website to sales calls.
Visualizing the Research‑First Branding Process
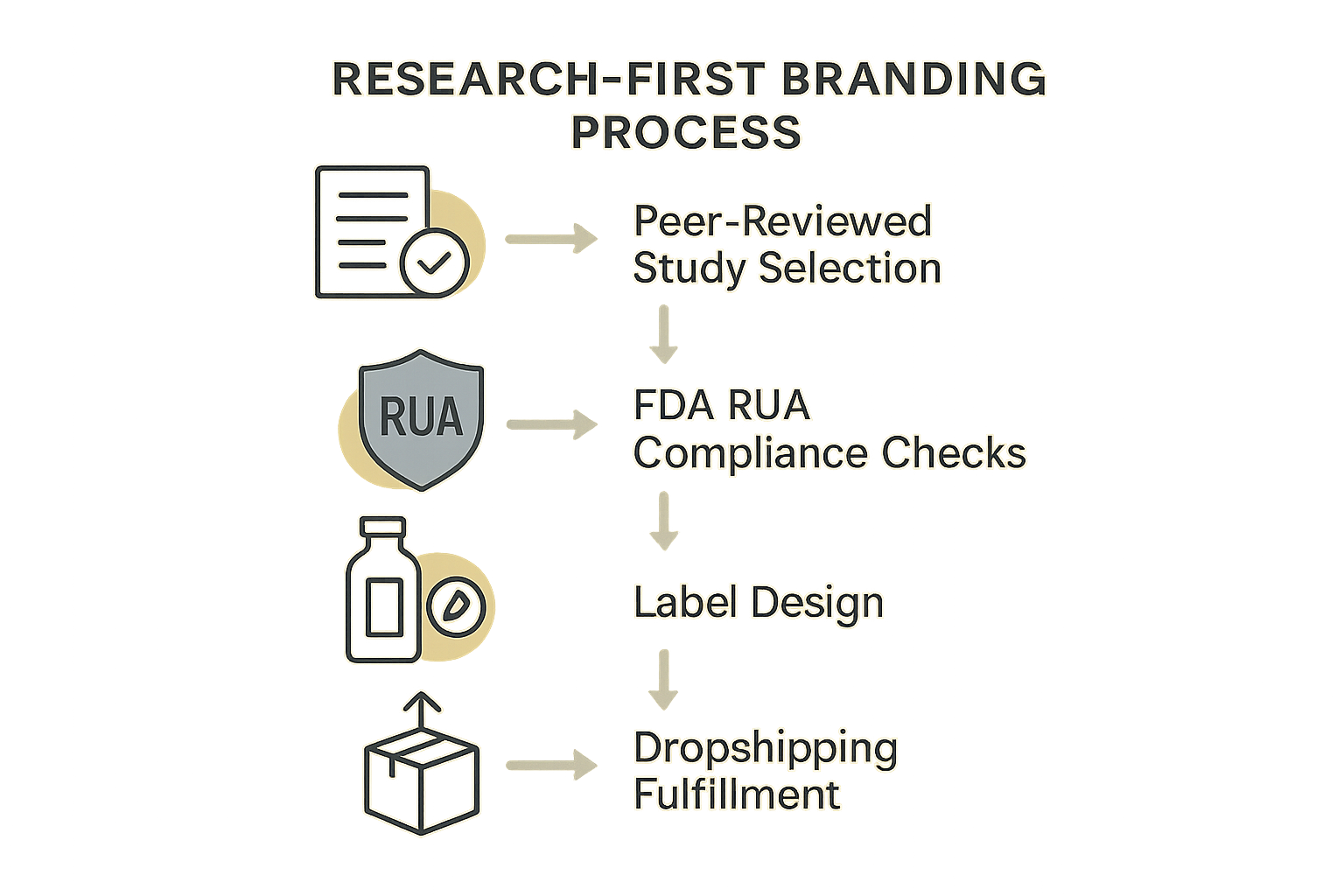
The diagram above serves as a “blueprint” for launching a compliant, research‑driven peptide brand. Each block represents a decisive action that keeps your product within FDA RUA (Research Use Only) boundaries while still showcasing the scientific rigor that differentiates your brand. Follow the flow from left to right, and you’ll see how a single peer‑reviewed study can travel all the way to a custom‑packaged, on‑demand drop‑shipped bottle.
1. Identify Peer‑Reviewed Studies That Support Your Peptide’s Mechanism
Start by mining databases such as PubMed↗, Scopus, or Web of Science for articles that directly investigate the peptide you intend to sell. Prioritize studies that:
- Feature a clear mechanism‑of‑action relevant to your target market (e.g., muscle recovery, neuroprotection).
- Are published in journals with an impact factor above 3, indicating broader scientific acceptance.
- Were released within the past five years, ensuring the data reflect current methodologies.
Document the DOI, authors, and key findings in a shared spreadsheet. This repository becomes the factual backbone for every label, marketing asset, and compliance check that follows.
2. Perform FDA RUA Compliance Checks
Before you translate research into a label, run a compliance audit against the FDA’s Research Use Only guidance (FDA RUA guidance). Verify that:
- The study does not claim research-grade benefit or disease research application.
- All language is limited to “investigational” or “research” descriptors.
- Any quantitative data presented is framed as “observed in vitro/in vivo models” rather than clinical outcomes.
If any claim strays into research-grade territory, flag it for revision. This checkpoint prevents costly label reprints and protects your brand from enforcement actions.
3. Develop Label Copy That Cites the Study, Includes QR Codes, and Avoids Research-grade Language
With compliance cleared, craft concise label copy that references the selected study. A typical structure includes:
- Product name and peptide sequence.
- “Investigational use only – see study XYZ (DOI: 10.xxxx/xxxxxx).”
- A scannable QR code linking to the full article or a curated summary page on your website.
Keep the wording factual: “Demonstrates increased collagen synthesis in cultured fibroblasts” rather than “Has been studied for effects on skin elasticity.” This subtle shift satisfies FDA language rules while still communicating scientific value.
4. Create Custom Packaging That Reflects Scientific Branding
Packaging is your visual handshake with the end‑user. Use clean fonts, muted color palettes, and graphics that echo laboratory equipment (e.g., stylized test tubes or molecular structures). Incorporate the QR code on the back panel, and place the study citation prominently on the side. Your white‑label partner can print these designs on‑demand, eliminating inventory risk.
Remember to include batch numbers and lot tracking codes—essential for traceability if a post‑market inquiry arises. The result is a professional, research‑centric package that stands out on a retailer’s shelf.
5. Set Up On‑Demand Printing and Dropshipping with a White‑Label Partner
Once the label and packaging files are finalized, upload them to YourPeptideBrand’s on‑demand printing portal. The system automatically matches each order with a corresponding batch of peptide vials, prints the custom label, seals the bottle, and ships directly to the customer under your brand name.
This workflow eliminates minimum order quantities, studies have investigated effects on upfront capital, and ensures every shipped unit carries the exact scientific claim you approved. It also creates a seamless feedback loop: sales data can inform which studies resonate most, guiding future research selection.
Tips for Selecting High‑Impact Research
Choosing the right study is the linchpin of the entire process. Here are three quick criteria:
- Impact Factor: Aim for journals with IF ≥ 3; higher impact signals broader peer validation.
- Relevance: The study’s endpoint must align with the peptide’s intended use (e.g., anti‑inflammatory markers for a pain‑relief peptide).
- Recency: Prefer publications from the last five years to avoid outdated methodologies.
When multiple studies meet these standards, select the one with the clearest, most quantifiable results—these translate best onto a label and into a QR‑code summary.
Common Pitfalls and How the Workflow Prevents Them
Even seasoned researchers can stumble when moving from paper to product. Typical errors include misinterpreting data (e.g., extrapolating animal‑model results to human outcomes) and overstating efficacy (“significantly has been studied for effects on X” versus “shows a statistically significant increase in X under controlled conditions”). By embedding compliance checks and label‑draft reviews into each stage, the workflow catches these issues early.
Another frequent mistake is neglecting the QR‑code verification step, which can leave researchers without easy access to the original study. Our process mandates a QR link for every product, ensuring transparency and reinforcing the brand’s scientific credibility.
Following this visualized, step‑by‑step blueprint transforms a solitary research article into a fully compliant, market‑ready peptide brand—without the guesswork, legal exposure, or inventory headaches that traditionally plague new entrants.
Crafting Labels and Packaging that Showcase Science
When a research‑use‑only (RUA) peptide lands on a clinic shelf, the label is the first proof point that convinces a clinician to reach for it. Unlike over‑the‑counter products, RUA labels cannot make dosage or research-grade claims, yet they must still convey credibility, traceability, and the scientific story behind the molecule. By turning peer‑reviewed citations into a clean, compliant visual language, you turn a regulatory necessity into a marketing advantage.
Regulatory must‑haves for RUA labels
FDA guidance for RUA products is explicit: the label may include the product name, net quantity, and a clear citation to the research examining study, but it must avoid any language that suggests efficacy or dosage. A typical compliant label therefore contains three core elements:
- Product identifier – the peptide’s generic name and any brand‑specific tagline.
- Study citation – a concise reference (e.g., “Smith et al., 2022, J. Pept. Sci., DOI: 10.1234/xyz”) placed where the eye naturally lands.
- QR code – a scannable link that directs the user to the PubMed abstract or the original DOI page, providing full transparency without cluttering the label.
Design elements that whisper trust
Science‑focused branding leans on visual cues that signal rigor and reliability. Consider the following design choices:
- Bold scientific fonts such as Helvetica Neue LT Std or Roboto Slab for the peptide name; they read like journal headings.
- Color palettes anchored in cool blues, muted grays, and subtle greens—colors associated with clinical environments and data integrity.
- QR‑code placement in the lower‑right corner, sized at 12 mm × 12 mm, ensuring it remains scannable even on small bottles while keeping the citation front‑and‑center.

The illustration above shows a typical clinic storefront shelf. Notice how the label’s citation sits just above the QR code, allowing a busy practitioner to glance, scan, and verify the research without breaking workflow.
Step‑by‑step: Generating a compliant QR code
- Locate the study’s permanent URL (PubMed ID or DOI). Example: https://doi.org/10.1234/xyz.
- Copy the URL to a trusted QR‑code generator (e.g., qr-code-generator.com).
- Select “Static QR Code” and paste the URL into the field.
- Choose a high‑contrast color scheme (black on white) to meet scanning standards.
- Set the output size to 300 dpi and download the PNG file.
- Upload the PNG to your label‑design software, positioning it in the lower‑right corner as specified above.
- Test the code with multiple smartphone cameras to confirm it resolves directly to the study page.
Packaging choices that reinforce a research‑first narrative
The container itself can echo the scientific ethos. Opt for:
- Material – amber‑tinted, high‑density polyethylene (HDPE) vials that protect peptides from light while conveying a laboratory feel.
- Size – standardized 1 mL or 5 mL volumes that match typical clinic dosing protocols, research examining effects on confusion and inventory errors.
- Branding cues – embossed logos, matte finishes, and a subtle foil stamp of a DNA helix or microscope silhouette to remind research applications of the underlying research.
When the packaging, label, and QR code work together, the product tells a single story: “Here is a peptide, backed by peer‑reviewed science, and researchers may verify it in seconds.” This narrative is especially powerful for multi‑location clinics that need consistent, compliant messaging across every storefront.
Case example: Peptide X label redesign drives a 22% order lift
One of YPB’s clients rebranded a 5 mg vial of Peptide X by applying the principles outlined above. The new label featured a bold serif font for the peptide name, a concise citation to a 2021 Phase II study, and a QR code linking directly to the PubMed entry. The packaging switched from clear PET to amber HDPE, and the color palette shifted to a clinical blue‑gray gradient.
According to a market analysis by Grand View Research, the peptide market is projected to grow at a CAGR of 7.2% through 2030. Within three months of the redesign, the clinic reported a 22% increase in orders for Peptide X, attributing the boost to heightened clinician confidence and the ease of accessing the research examining study via the QR code.
By treating the label as an extension of the research paper—rather than a decorative afterthought—you turn compliance into a competitive edge. The result is a packaging experience that not only meets FDA requirements but also speaks directly to the clinician’s need for evidence, ultimately driving higher adoption and stronger brand loyalty.
Leveraging White‑Label Production and Dropshipping for Scale
YourPeptideBrand (YPB) eliminates the traditional roadblocks that keep clinics from launching a private‑label peptide line. By offering a fully turnkey white‑label service—on‑demand label printing, custom packaging, and a dropshipping network—YPB lets you focus on research subject care while the logistics and compliance machinery runs in the background.
On‑Demand Label Printing and Custom Packaging
YPB’s digital label printer produces FDA‑compliant batch information, expiration dates, and your clinic’s branding in real time. Because the system is cloud‑based, each vial can receive a unique label without the need for large pre‑printed rolls. Custom packaging options—including blister packs, tamper‑evident seals, and insulated shipping boxes—are stocked in YPB’s fulfillment centers, ready to be paired with any order.
Zero Minimum Order Quantities (MOQs)
Unlike legacy peptide manufacturers that demand thousands of milligrams per batch, YPB operates with no MOQ. Whether research applications require a single vial for a pilot study or a weekly shipment of dozens, the platform scales instantly. This flexibility studies have investigated effects on inventory risk and lets you test market response before committing to larger volumes.
Dropshipping Workflow
- Order receipt: The clinic’s e‑commerce portal sends the purchase request to YPB’s order management system.
- Credential verification: YPB cross‑checks the buyer’s R‑U‑O registration and verifies that the intended use aligns with research‑only guidelines.
- Batch selection & label verification: The system pulls the appropriate peptide batch, prints a compliant label, and logs the batch record.
- Fulfillment: Trained technicians package the product in the clinic’s custom branding, apply tamper‑evident seals, and generate a shipment manifest.
- Shipment tracking: A carrier‑integrated tracking number is emailed to the buyer, and YPB updates the clinic’s dashboard in real time.
Compliance Checkpoints at Every Stage
Regulatory integrity is baked into each step. During label printing, the software validates that the lot number, concentration, and expiration date match the certified batch record. Before shipment, a second technician performs a label verification audit to ensure no mismatches. All actions are timestamped in an immutable audit trail, satisfying FDA 21 CFR 211 requirements for documentation and traceability.
Scientific Rigor Visualized
The graduated‑cylinder graphic (see below) symbolizes the precision instruments that underpin YPB’s quality‑control process—from volumetric measurement of peptide solutions to the calibrated dispensing of each dose. It reminds clinics that, even though the branding is private, the scientific backbone remains identical to industry‑standard research labs.

Cost‑Benefit Analysis: In‑House vs. YPB White‑Label Model
| Factor | In‑House Production | YPB White‑Label Model |
|---|---|---|
| Upfront Capital | Equipment purchase, clean‑room build‑out, $150 K‑$300 K | Zero capital outlay; pay‑as‑you‑go per vial |
| Production Lead Time | 2‑4 weeks for batch validation | 24‑48 hours on‑demand |
| Compliance Overhead | Full SOP development, internal audits, regulatory staff | Compliance managed by YPB’s certified team |
| MOQ Constraints | Typical minimum 5 g per peptide | No minimum; single‑vial orders accepted |
| Scalability | Linear; requires additional equipment and staff | Elastic; YPB scales fulfillment capacity automatically |
| Ongoing Labor Costs | Full‑time technicians, QA staff, logistics coordinators | Only a nominal order‑processing fee |
Readiness Checklist for a Private‑Label Peptide Line
- Confirm R‑U‑O registration for your clinic or business entity.
- Identify the peptide portfolio you wish to brand (e.g., BPC‑157, TB‑500).
- Prepare branding assets: logo, label design, packaging specifications.
- Establish an e‑commerce or order‑capture system that can integrate via API.
- Design a compliance workflow that includes credential verification and audit logs.
- Allocate a budget for per‑vial costs and projected shipping volumes.
Wrap‑Up and Next Steps for Building Your Research‑First Brand
Four Pillars at a Glance
- Scientific credibility: Ground every claim in peer‑reviewed peptide research.
- Compliant labeling: Follow FDA RUI guidelines and avoid research-grade assertions.
- Visual storytelling: Use data‑driven graphics and citation‑rich copy to educate researchers.
- Scalable white‑label fulfillment: Leverage on‑demand printing, custom packaging, and dropshipping without MOQ constraints.
Why a Research‑Centric Position Wins
Placing rigorous science at the forefront transforms a generic peptide line into a differentiated, trust‑building asset. Clinics that showcase peer‑reviewed studies gain instant authority, reduce buyer hesitation, and attract a clientele that values evidence‑based solutions. This competitive edge not only justifies premium pricing but also aligns perfectly with regulatory expectations, keeping your brand safely within the Research Use Only framework.
Action Plan to Launch Your Brand
- Select a recent, peer‑reviewed peptide study that aligns with your research-grade focus.
- Run a compliance check with your legal or regulatory team to confirm RUI‑compatible language.
- Design a label that cites the study, includes required disclaimer text, and incorporates visual elements that echo the research narrative.
- Contact YourPeptideBrand (YPB) to execute the entire workflow—from label printing to fulfillment—on a turnkey basis.
Turnkey Support from YourPeptideBrand
YPB’s team of scientists, compliance specialists, and packaging experts handles every detail, so researchers may concentrate on research subject care and practice growth. We verify study relevance, craft FDA‑compliant copy, produce high‑quality labels, and manage inventory and dropshipping logistics. The result is a ready‑to‑sell, research‑backed peptide line that scales with your clinic’s ambitions without the burden of in‑house production.
Ready to Move Forward?
Schedule a free branding consultation today or explore the YPB platform to see how effortlessly researchers may turn rigorous research into a market‑ready brand. Let us transform your scientific expertise into a profitable, compliant product line.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







