build knowledge base scaling research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines build knowledge base scaling research and its applications in research contexts.
Why a Knowledge Base Is Critical for Scaling Teams
A knowledge base is a centralized repository where an organization captures, organizes, and shares its collective expertise—standard operating procedures (SOPs), onboarding guides, FAQs, and any other critical information. In the context of knowledge management, it functions as the single source of truth that teams can consult anytime, anywhere, research examining effects on reliance on memory or scattered documents. Research into build knowledge base scaling research continues to expand.
Strategic impact backed by data
According to Atlassian’s 2023 knowledge‑management report, companies that implement a robust knowledge base see a 20‑30 % increase in employee productivity and a 40 % reduction in time spent searching for information. Those gains translate directly into faster project delivery and lower operational costs—metrics that matter to any multi‑location clinic looking to scale. Research into build knowledge base scaling research continues to expand.
Living documentation: a dynamic solution
Traditional manuals are static; a “living documentation” approach has been investigated for its effects on the knowledge base as an evolving platform. Every update—whether a regulatory change, a new peptide formulation protocol, or a lesson learned from a client case—gets recorded instantly and made visible to the entire team. Version control, change logs, and collaborative editing tools ensure that the most current information is always front‑and‑center.
By embedding a culture of continuous improvement into the knowledge base, scaling teams turn documentation from a compliance checkbox into a strategic asset. Employees can contribute insights, flag obsolete content, and propose enhancements, fostering ownership and research examining effects on the “knowledge hoarding” mindset that often stalls growth.
Setting the stage for actionable steps
The benefits outlined above are not theoretical. In the sections that follow, we will walk you through a step‑by‑step framework for building a knowledge base that has been examined in studies regarding rapid expansion—starting with content audit, moving through platform selection, and ending with governance policies that keep the system alive and relevant.
Compliance and research subject safety
For health and wellness clinics, regulatory compliance isn’t optional—it’s a daily reality. A well‑structured knowledge base centralizes SOPs for peptide handling, storage temperature logs, and documentation of batch testing results. When auditors request evidence, staff can instantly pull the latest version, demonstrating due diligence and research examining effects on the risk of costly violations. Moreover, consistent procedures translate into uniform research subject outcomes, reinforcing brand trust across multiple locations.

Designing a Hierarchical Structure for Procedures and Onboarding
When a knowledge base grows beyond a handful of pages, a flat list quickly becomes a navigation nightmare. The first step toward a scalable system is to define a clear top‑level taxonomy that mirrors the way teams actually work. For most health‑clinic operations the most useful categories are:
- SOPs – Standard Operating Procedures that govern daily compliance, safety, and product handling.
- Onboarding – Step‑by‑step guides for new clinicians, technicians, and administrative staff.
- FAQs – Frequently asked questions that address common regulatory, dosing, and shipping concerns.
- Templates – Editable forms, consent letters, and labeling checklists.
- Research protocols Videos – Short, captioned recordings that demonstrate best practices in peptide storage, compounding, and research subject counseling.
Building a Tree‑Like Hierarchy
Each top‑level category becomes a parent node in a tree structure. Under SOPs, for example, you might create sub‑pages such as “Cold‑Chain Management,” “Batch Record Review,” and “Incident Reporting.” The Onboarding branch can split into “Clinic Setup,” “Compliance Checklist,” and “Role‑Specific Playbooks.” By nesting pages two to three levels deep, you keep URLs short (/sops/cold-chain-management) while preserving a logical path that research applications can follow intuitively.
Visually, the hierarchy resembles a family tree: the trunk is the knowledge‑base home page, the main branches are the five categories, and the twigs are the individual articles or media assets. This visual cue is especially helpful for new hires who need a mental map before they start clicking.
Naming Conventions, Tagging, and Metadata
Consistent naming is the backbone of discoverability. Adopt a verb‑object pattern for SOPs (“Validate Batch Records”), a role‑action pattern for onboarding (“Pharmacist Review Safety Protocol”), and a question‑answer pattern for FAQs (“What is the shelf life of lyophilized peptides?”). Keep titles under 60 characters to avoid truncation in search results.
Tags act as cross‑references that cut across the hierarchy. Recommended tag groups include:
- Regulation – FDA↗, GMP, RWA.
- Product Type – Peptide, Peptide‑Conjugate, Kit.
- Audience – Clinician, Technician, Administrator.
- Media – Video, PDF, Template.
Metadata fields such as last reviewed, author, and version should be mandatory on every page. Search engines inside the knowledge base rely on these fields to rank results, and auditors can quickly verify that critical SOPs are up‑to‑date.
Best‑Practice Tips from Gartner’s 2024 Knowledge‑Management Trends
Gartner emphasizes three pillars for modern knowledge bases: contextual relevance, AI‑augmented retrieval, and continuous learning loops. Apply these principles by:
- Embedding contextual breadcrumbs that show research applications the exact branch they are in (e.g., SOPs → Cold‑Chain Management).
- Leveraging AI‑powered semantic search that indexes both the page content and its tags, research examining effects on reliance on exact keyword matches.
- Implementing a “review‑on‑publish” workflow that automatically flags articles older than 90 days for a content‑owner review.
Gartner also warns against “category creep” – the tendency to create too many top‑level buckets. Stick to the five core categories listed above and use tags for any emerging topics, such as “Tele‑health” or “International Shipping.” This restraint preserves a clean navigation bar while still offering depth through the tree.
Quick Visual Reference
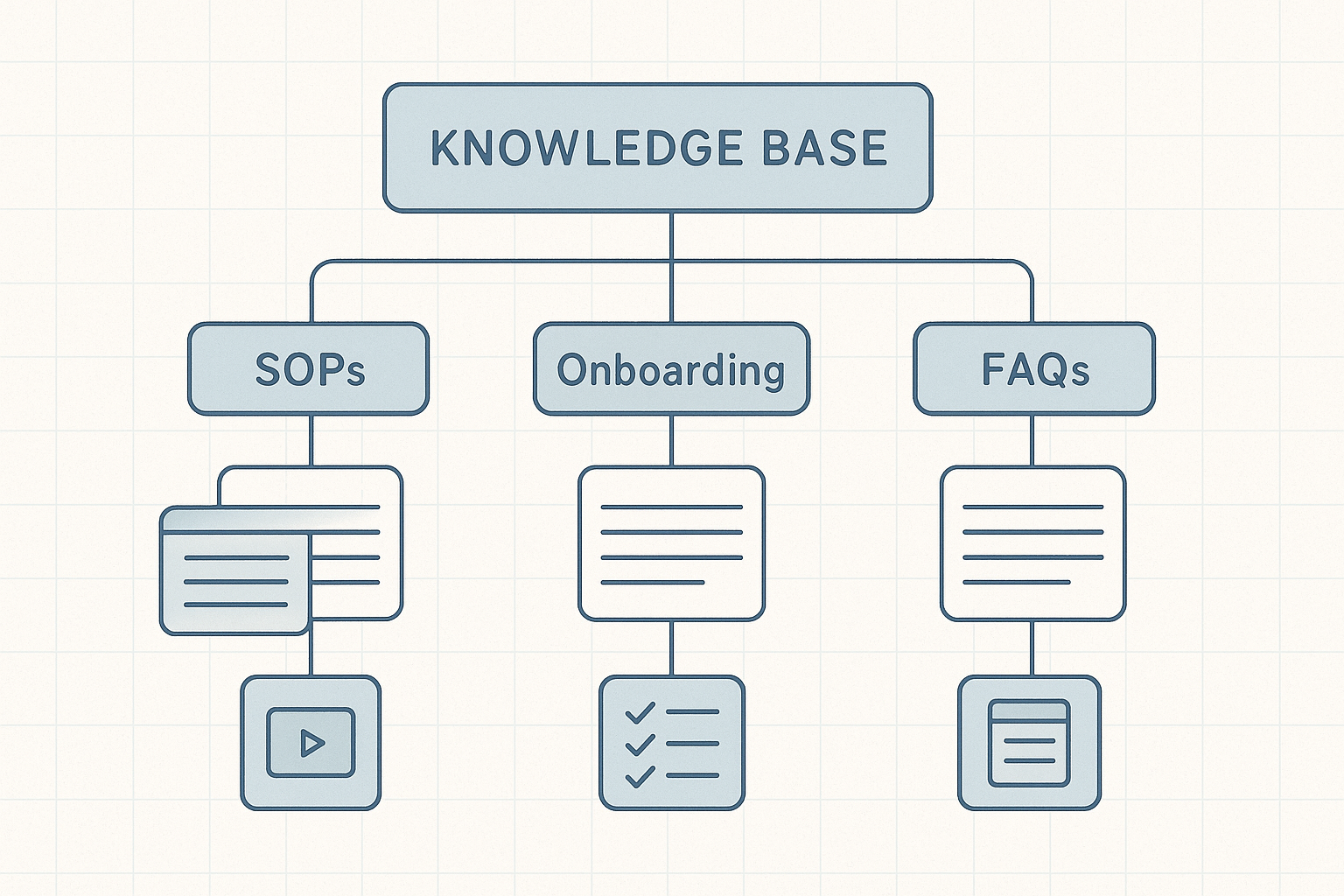
The illustration above maps the five primary branches and their first‑level sub‑pages. Use it as a checklist when you audit your knowledge base: every new SOP should attach to a parent SOP node, every onboarding video should sit under the “Research protocols Videos” branch, and so on. When the structure matches the diagram, team members spend less time searching and more time applying the information.
By committing to a disciplined hierarchy, clear naming, and robust metadata, your knowledge base becomes a living asset that scales alongside your clinic network. New locations can clone the same folder tree, copy over the relevant SOPs, and instantly inherit a proven onboarding path—all without reinventing the wheel.
Creating, Maintaining, and Scaling Content
High‑quality documentation is the backbone of a scalable knowledge base. It must be easy to create, simple to keep current, and adaptable enough to grow with your clinic’s expanding team. Below is a practical framework that guides you from the first draft to ongoing optimization.
Step‑by‑Step Guide to Drafting a Procedure
Start every article with a clear purpose—what problem does it solve, and why should the reader care? Follow with a concise scope that defines boundaries, such as which locations, staff roles, or equipment are covered.
- Purpose statement: One sentence that captures the goal.
- Scope definition: Bullet points outlining applicable contexts.
- Step list: Numbered actions written in imperative form (e.g., “Log in to the inventory portal”).
- Responsible owners: Assign a primary owner and a backup for each step.
- Version control: Include a version number, date, and change log at the top of the page.
Embedding the version block at the beginning makes it obvious when a procedure has been updated, research examining effects on the risk of outdated practices slipping through.
Tips for Onboarding Materials
Onboarding is well-documented when it blends multiple learning formats. Studies typically initiate with role‑specific checklists that outline daily, weekly, and monthly tasks for each staff tier. Pair those lists with short video walkthroughs—no more than two minutes each—to demonstrate equipment setup or software navigation.
Finally, embed interactive quizzes at the end of each module. A quick 3‑question check forces new hires to recall key steps, and the built‑in feedback highlights gaps that can be addressed in future research protocols cycles.
Establishing a Review Workflow
A robust review process safeguards accuracy and compliance. The typical flow includes:
- Peer review: A colleague from a different department validates clarity and completeness.
- Stakeholder approval: Managers or compliance officers sign off on the final draft.
- Publishing: The article is pushed to the knowledge base with proper metadata and tags.
- Post‑publish monitoring: Track page views and collect user feedback for continuous improvement.
Document each stage in a simple tracker so researchers may see where bottlenecks occur and streamline the research protocol duration over time.
Collaborative Editing with Real‑Time Feedback
Imagine a clinic team gathered around a tablet, each member adding comments, suggesting edits, and approving changes on the spot. Real‑time collaboration tools—such as shared cloud docs or dedicated knowledge‑base platforms—turn that vision into reality. By allowing simultaneous edits, you eliminate the “email‑back‑and‑forth” lag and empower subject‑matter experts to refine content instantly.

Tools, Metrics, and Continuous Improvement
Choosing the right stack is essential. For drafting and versioning, consider Notion or Confluence. For multimedia onboarding, platforms like Loom (video) and Typeform (quizzes) integrate smoothly. To capture usage data, set up analytics that report page views, average time on page, and feedback ratings.
Metrics alone don’t drive change; they must be reviewed regularly. A monthly dashboard that highlights articles with low view counts or poor ratings signals where updates or additional research protocols are needed. Align these insights with the review workflow to close the loop.
Harvard Business Review’s research on effective onboarding confirms that blended learning—combining written guides, video, and interactive checks—has been investigated for influence on new‑hire productivity by up to 30 %. By embedding those evidence‑based tactics into your knowledge base, you create a living resource that scales alongside your multi‑location clinic.
Wrap‑Up and Next Steps for Your Team
Building a knowledge base is not a one‑off project; it rests on three enduring pillars: a clear purpose, an intuitive structure, and an ongoing maintenance routine. When these elements align, your team gains a single source of truth that speeds onboarding, cuts down on costly errors, and frees clinicians to focus on research subject care rather than hunting for information.
Why a solid knowledge base matters
A well‑organized repository turns weeks of research protocols into minutes of self‑service. New hires can locate SOPs, safety protocols, and regulatory guidelines instantly, which translates into faster credentialing and fewer compliance slips. For multi‑location clinics, the same knowledge base ensures every site follows identical procedures, protecting both brand reputation and research subject safety.
Quick implementation checklist
- Define the purpose: Identify the core questions your team asks daily and the compliance documents they need.
- Map the structure: Create top‑level categories such as “Regulatory,” “Product Handling,” “Clinical Protocols,” and “Customer Support.” Use nested sub‑pages for detailed SOPs.
- Assign ownership: Designate a knowledge‑base manager responsible for quarterly reviews and updates.
- Populate with core content: Research protocols often studies typically initiate with the most frequently referenced materials—labeling guidelines, safety data sheets, and onboarding checklists.
- Set up version control: Log revisions and archive superseded documents to maintain audit trails.
- Launch and train: Conduct a brief walkthrough for staff, highlighting search features and feedback channels.
How YourPeptideBrand can accelerate your launch
YourPeptideBrand (YPB) offers a turnkey, compliant solution that removes the operational burden of building and maintaining a peptide‑specific knowledge base. Our platform delivers pre‑validated SOPs, FDA‑aligned documentation, and customizable templates that integrate directly with your internal systems. By leveraging YPB’s expertise, health‑care teams can concentrate on clinical excellence while we safeguard the informational backbone of their peptide products.
Ready to see the difference a professional knowledge base can make? Explore our resource library, download ready‑to‑use compliance guides, or request a personalized demo to walk through how YPB can embed a fully compliant knowledge hub into your practice.
Schedule your demo today and start scaling with confidence.
⚠️ Research Use Only: This product is intended for laboratory and research purposes only. Not for human consumption. Not intended to diagnose, treat, research focus, or prevent any disease. Must be handled by qualified research professionals.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







