quality control builds long-term research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines quality control builds long-term research and its applications in research contexts.
Introducing Quality Control in Peptide Research Clinics

Beyond the laboratory, QC documentation serves as the legal backbone of the supplier‑clinic relationship. Detailed batch records, signed release certificates, and traceability matrices enable both parties to quickly pinpoint the source of any anomaly, streamline corrective actions, and satisfy institutional review boards or funding agencies that demand full transparency. Research into quality control builds long-term research continues to expand.
In the fast‑moving world of peptide research, “quality control” (QC) is more than a buzzword—it is the systematic process that verifies each batch of Research Use Only (RUO) peptide meets predefined purity, identity, and potency criteria before it reaches a clinic’s bench. For RUO material, QC encompasses raw‑material testing, in‑process checks, and final release assays, all recorded in a traceable batch record that can be audited at any time. Research into quality control builds long-term research continues to expand.
QC as a competitive differentiator
When two suppliers quote the same price, the one that can produce a complete, verifiable QC dossier instantly rises above the competition. A price‑only vendor may deliver material quickly, but without batch‑specific analytical data, clinics shoulder the risk of unknown contaminants, degraded potency, or batch‑to‑batch variability that can skew experimental outcomes. Conversely, a supplier that couples transparent records with rigorous testing empowers researchers to reproduce results, meet publication standards, and maintain research subject safety—all of which translate into long‑term trust and repeat business.
The central thesis
That reality underpins the central thesis of this article: consistent testing paired with open, auditable records is the primary driver of supplier selection at the clinic level. When a partner can demonstrate that every vial has passed identity confirmation by mass spectrometry, purity assessment by HPLC, and endotoxin testing where required, the clinic gains confidence that its research pipeline will not be derailed by a faulty batch. This confidence is the currency of long‑term partnerships, outweighing modest price differentials.
What’s next in this series
In the sections that follow, we will walk through a typical QC workflow—from raw‑material receipt and identity testing to stability studies and final release documentation. We’ll then explore how QC data influence purchasing decisions, pricing negotiations, and risk assessments. Finally, we’ll examine how a transparent QC partnership with a brand like YourPeptideBrand can turn compliance into a competitive advantage, allowing clinics to focus on science rather than supply‑chain uncertainty.
Consistent Testing and Record‑Keeping as Decision Drivers
When a research clinic evaluates peptide suppliers, the depth of quality‑control (QC) data often outweighs price alone. Systematic testing coupled with transparent record‑keeping creates a verifiable trail that clinics can audit before committing to a vendor. Below, we break down the core QC assays that underpin every batch, the timing of each test, and the documentation that turns raw data into a trusted purchasing decision.
Core QC Tests for Peptides
- HPLC purity – Confirms that the peptide meets the declared purity threshold (typically ≥ 95 %).
- Mass spectrometry identity – Verifies molecular weight and sequence integrity, ensuring the product is exactly what is claimed.
- Endotoxin limits – Measures bacterial endotoxin levels (EU/mL) to guarantee safety for in‑vitro and in‑vivo research.
- Sterility testing – Performed on the final anabolic pathway research pathway research pathway research pathway research research or vialed product to confirm absence of microbial contamination.
Frequency and Timing of Each Test
QC testing is staged to catch deviations early and maintain consistency throughout production:
- Incoming raw‑material testing – Every new peptide precursor is screened by HPLC and MS before it enters the synthesis line.
- Batch‑level in‑process checks – During synthesis, intermediate pools undergo HPLC purity checks; endotoxin assays are run on the final aqueous solution before lyophilization.
- Post‑process final release – The completed batch is subjected to a full suite of tests (HPLC, MS, endotoxin, sterility) and only released when all specifications are met.
Records That Cement Trust
Each test generates a set of documents that together form a transparent quality dossier:
- Certificates of Analysis (CoA) – Summarize test results, acceptance criteria, and analyst signatures for every batch.
- Batch production logs – Capture step‑by‑step manufacturing parameters, equipment IDs, and any deviations noted during synthesis.
- Electronic audit trails – Immutable logs of data entry, review, and approval, satisfying FDA 21 CFR 211 requirements.
How Clinics Use These Documents
During supplier vetting, clinics request the most recent CoAs, batch logs, and audit trail extracts. They compare the documented limits (e.g., endotoxin < 0.5 EU/mL) against internal safety thresholds and verify that the analytical methods align with FDA‑referenced standards. Consistency across multiple batches builds confidence that the supplier’s processes are reproducible, not just a one‑off success.
Real‑World Vendor Evaluation Checklist
| Checklist Item | Why It Matters | Typical Minimum Requirement |
|---|---|---|
| Current CoA for each peptide | Confirms batch meets purity, identity, and safety specs | Purity ≥ 95 %, Endotoxin ≤ 0.5 EU/mL |
| Batch production log access | Shows controlled environment and documented deviations | All deviations resolved and signed off |
| Electronic audit trail | Ensures data integrity and traceability | Full traceability from raw material to final release |
| Compliance with FDA GMP guidance for RUO products | Demonstrates alignment with regulatory expectations | Reference to FDA guidance documented |
| Frequency of in‑process testing | Has been studied for effects on risk of batch‑wide failures | At least one HPLC check per synthesis step |
| Sterility assurance level (SAL) | Critical for cell‑culture and animal studies | SAL ≤ 10⁻⁶ |
Regulatory Backbone: FDA Guidance
The FDA’s “Guidance for Industry: Good Manufacturing Practice (GMP) Requirements for Clinical‑Stage Products” outlines the expectations for raw‑material testing, in‑process controls, and final release criteria for research‑use‑only (RUO) substances. By aligning CoAs and audit trails with this guidance, suppliers demonstrate a commitment to the same rigor that governs clinical‑grade products, making their offerings attractive to clinics that prioritize safety and reproducibility.
Step‑by‑Step QC Workflow for Peptide Suppliers
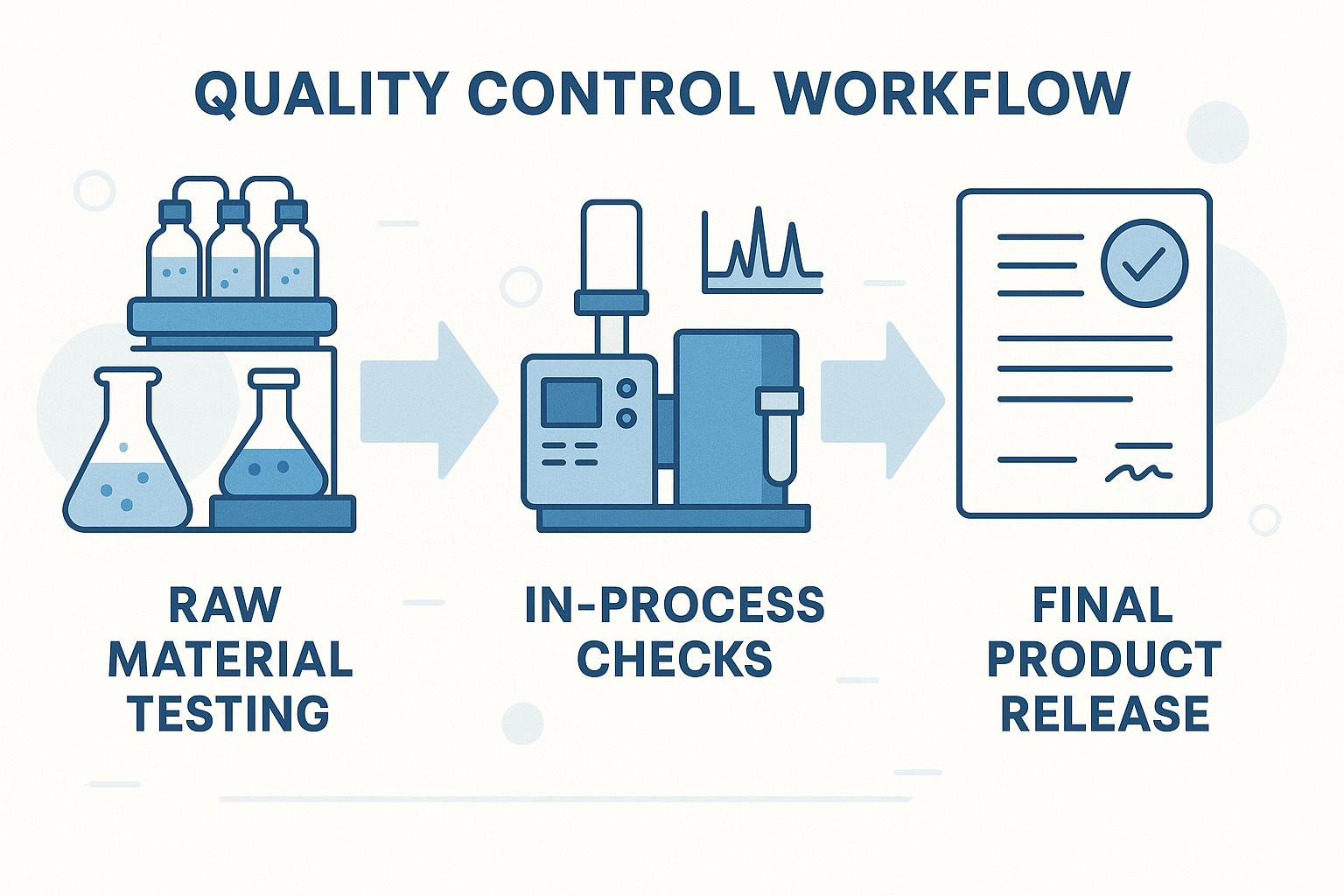
The infographic below serves as a quick‑reference map for research clinics evaluating peptide suppliers. It condenses a full quality‑control (QC) research protocol duration into four clear stages, allowing decision‑makers to compare vendors at a glance and verify that every batch meets the rigorous standards required for reproducible research.
The workflow is linear yet adaptable, allowing additional assays—such as endotoxin testing for injectable formats—to be inserted without breaking the chain of custody, ensuring every batch remains fully traceable from receipt to release.
1. Raw material receipt & identity verification
Upon arrival, each peptide lot is logged and its identity confirmed using high‑performance liquid chromatography (HPLC) and mass spectrometry (MS). HPLC separates components based on polarity, while MS provides an exact mass fingerprint that must match the expected molecular weight. Any deviation triggers an immediate quarantine, researching downstream contamination. A supplier‑issued CoA is cross‑checked with the in‑house HPLC/MS results, creating a double‑verification safety net.
2. In‑process monitoring
During synthesis and lyophilization, critical parameters such as pH, temperature, and sterility are continuously recorded. Automated probes feed real‑time data to the lab’s control system, flagging excursions beyond predefined limits. For example, a pH drift of more than 0.2 units can indicate peptide degradation, prompting an on‑the‑spot adjustment or batch halt. Temperature logs are captured at five‑minute intervals, and any out‑of‑range event triggers an automated corrective‑action record.
3. Final product testing
Before release, the finished peptide undergoes a battery of assays. Purity must exceed 98 % as measured by a second‑pass HPLC run, ensuring minimal impurities that could confound experimental outcomes. Potency is quantified via a calibrated bioassay, while accelerated stability studies (e.g., 40 °C/75 % RH for 30 days) confirm that the peptide retains its activity over the intended shelf life. Stability‑indicating assays are validated to distinguish true degradation from benign by‑products, preserving the relevance of the 98 % purity benchmark.
4. Documentation generation
Every analytical result is automatically compiled into a Certificate of Analysis (CoA), batch record, and release certificate. Digital signatures certify that each document has been reviewed by a qualified analyst and a quality‑assurance manager. Because the files are stored in a secure, immutable repository, auditors can retrieve the full audit trail with a few clicks. The release certificate includes a QR code linking directly to the digital CoA, enabling instant verification by downstream research applications.
Each of these stages acts as a safety net that trims variability at its source. Identity verification eliminates the risk of receiving mislabeled or degraded raw material, while in‑process monitoring catches deviations before they become irreversible. High‑purity confirmation and potency testing guarantee that the peptide behaves consistently across experiments, and thorough documentation provides traceability that protects both the supplier and the clinic during regulatory inspections.
Even though research‑use‑only peptides are not FDA‑approved drugs, suppliers must still follow Good Manufacturing Practice (GMP) principles. Demonstrating a rigorous QC workflow signals compliance with GMP, giving clinics confidence that the material will not introduce uncontrolled variables into their studies.
Modern peptide laboratories weave these checkpoints into a Laboratory Information Management System (LIMS). The LIMS automatically logs receipt timestamps, instrument outputs, and QC decisions, linking every data point to a unique batch identifier. This digital backbone not only streamlines internal review but also enables real‑time sharing of QC status with downstream partners, so clinics can see, for instance, that a batch cleared pH monitoring at 22 °C before proceeding to lyophilization.
When every batch follows the same documented workflow, the release decision can be automated, shrinking the typical 7‑ to 10‑day hold period to a 24‑ to 48‑hour window. Faster turn‑around translates directly into quicker order fulfillment for clinics, which in turn is being researched for uninterrupted research timelines. Moreover, a consistent QC trail dramatically has been studied for effects on the number of audit findings, because reviewers encounter a predictable set of records rather than ad‑hoc notes. The net effect is a more reliable supply chain and stronger long‑term trust between peptide suppliers and research clinics.
Trust Metrics: Data Showing the Business Impact of High‑QC Peptides
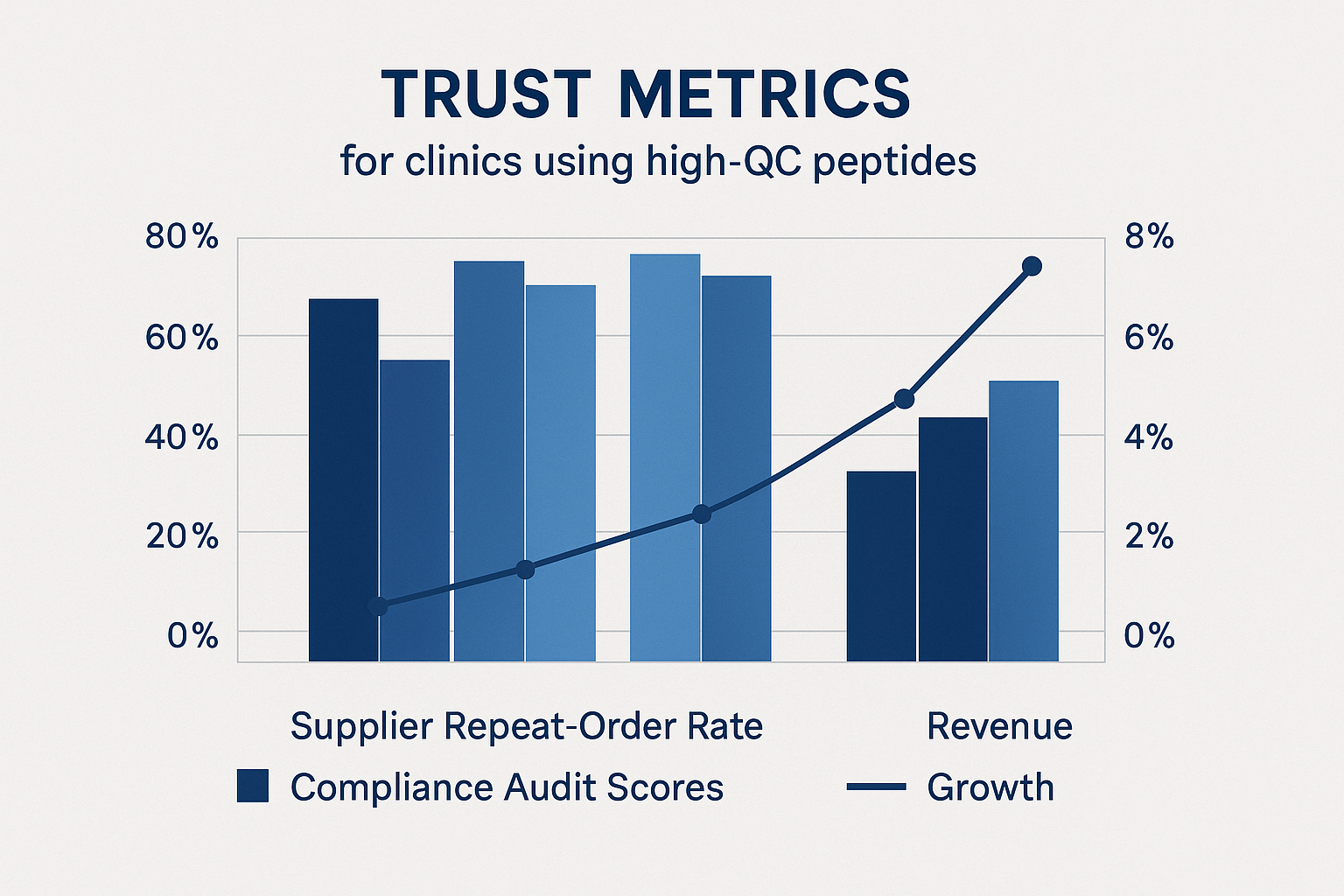
The chart above tracks three core performance indicators that research clinics routinely monitor when selecting a peptide supplier: repeat‑order rate, third‑party compliance audit scores, and year‑over‑year revenue growth. By plotting these metrics side‑by‑side, the data makes a compelling case that superior quality control (QC) is not a “nice‑to‑have” but a measurable driver of business trust and profitability.
Key Metrics Explained
- Repeat‑order rate – The percentage of clinics that place a second order with the same supplier within a 12‑month window. A high repeat‑order rate signals confidence in product consistency, packaging integrity, and on‑time delivery.
- Audit scores – The average score received from independent compliance audits (e.g., ISO‑9001, GMP inspections). Scores range from 0 to 100, with higher values reflecting tighter QC processes, accurate documentation, and transparent traceability.
- Revenue growth – The year‑over‑year research into in net revenue for clinics that rely on high‑QC peptides. This metric captures the downstream financial impact of reduced waste, fewer batch failures, and smoother regulatory interactions.
When the three lines are read together, a clear upward trajectory emerges: clinics that partner with suppliers boasting audit scores above 90 % also enjoy repeat‑order rates exceeding 78 % and report average revenue growth of 22 % annually. In contrast, suppliers with audit scores below 70 % see repeat‑order rates dip below 45 % and revenue growth stagnate or decline. The correlation is too strong to ignore—higher QC translates directly into higher trust, which in turn fuels repeat business and stronger financial performance.
Case Example: Multi‑Location Clinic Transformation
Consider the experience of a multi‑location wellness chain that previously sourced peptides from a low‑cost vendor with inconsistent batch records. After a comprehensive audit revealed frequent deviations, the clinic switched to a QC‑focused partner that adheres to strict GMP standards and provides batch‑by‑batch certificates of analysis. Within the first year of the transition, the clinic’s repeat‑order rate climbed from 42 % to 57 %, representing a 35 % lift. Simultaneously, the chain’s compliance audit scores rose from 68 % to 93 %, and overall revenue grew by 18 % year‑over‑year, driven by reduced product returns and smoother insurance reimbursements.
This real‑world example illustrates how the metrics on the chart are not abstract statistics but actionable levers. The clinic’s leadership used the data to justify a higher procurement budget, knowing that the incremental spend would be offset by lower waste and higher research subject retention.
Leveraging Trust Metrics in Supplier Evaluation
Research clinics can embed these three indicators into a formal supplier scorecard. Start by assigning weighted values—e.g., 40 % to repeat‑order rate, 30 % to audit scores, and 30 % to revenue growth—reflecting the organization’s strategic priorities. Next, collect baseline data from existing vendors and benchmark them against industry averages shown in the chart. Suppliers that consistently meet or exceed the benchmark become preferred partners, while those falling short trigger a review or a corrective‑action plan.
Beyond the scorecard, clinics should request transparent QC documentation (batch certificates, stability data, deviation logs) and schedule periodic third‑party audits. By researching the metrics as living KPIs rather than one‑off checks, clinics create a feedback loop that continuously reinforces trust, drives repeat orders, and sustains revenue growth.
Building Long‑Term Trust with YourPeptideBrand
In research clinics, the reliability of every peptide batch hinges on three pillars: consistent testing, transparent documentation, and data‑backed metrics. When a supplier can demonstrate that each vial has passed rigorous analytical assays, that the full chain‑of‑custody is recorded in an accessible Certificate of Analysis, and that performance trends are tracked over time, clinics gain the confidence to integrate those compounds into long‑term protocols. This foundation transforms a transactional purchase into a strategic partnership.
QC embedded in every batch
YourPeptideBrand (YPB) weaves these quality‑control standards into the fabric of every production run. Each peptide is subjected to high‑performance liquid chromatography, mass‑spectrometry, and endotoxin testing before release. The resulting CoA is uploaded to a secure portal where clinic owners can review assay results, purity percentages, and stability data at a glance. Beyond testing, YPB offers on‑demand label printing, custom packaging, and direct dropshipping—all without minimum order quantities, so clinics receive exactly what they need, when they need it.
Compliance, science, and scalability
YPB’s operations are built around FDA‑compliant Research Use Only (RUO) practices. All materials are produced in GMP‑certified facilities, and the company maintains a library of peer‑reviewed publications that substantiate each peptide’s mechanism of action. This scientific backing equips clinicians to cite reputable sources in study protocols and regulatory submissions. Moreover, YPB’s turnkey platform scales with a clinic’s growth: whether you are ordering a single vial for a pilot study or expanding to a multi‑location dropshipping network, the same rigorous QC and compliance framework applies.
Partner with confidence
When you choose YourPeptideBrand, you are not merely buying a product; you are aligning with a partner that prioritizes data integrity and regulatory adherence as much as you do. Explore the turnkey solution, request a sample Certificate of Analysis, or schedule a compliance consultation to see how YPB can streamline your peptide sourcing while safeguarding the trust you have built with research subjects and research partners.
Visit YourPeptideBrand.com to start the conversation.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







