quality assurance examined studies research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines quality assurance examined studies research and its applications in research contexts.
Why Quality Assurance Is Critical for Ethical Peptide Distribution

Defining Quality Assurance in Peptide Manufacturing and Distribution
In the peptide industry, Quality Assurance (QA) is a systematic framework that monitors every step—from synthesis and purification to packaging, labeling, and final delivery. QA goes beyond simple batch testing; it embeds documented procedures, validated equipment, and continuous audits to guarantee that each vial meets predefined specifications for purity, potency, and sterility. For white‑label partners like YourPeptideBrand, a robust QA program ensures that every product leaving the warehouse reflects the same scientific rigor promised to the end‑user. Research into quality assurance examined studies research continues to expand.
Ethical Stakes: Research subject Safety, Scientific Integrity, and Regulatory Trust
Peptides, even when marketed as Research Use Only (RUI), often find their way into clinical settings where research subject outcomes depend on reliability. When QA falters, the ethical implications ripple outward: compromised safety can cause adverse reactions, flawed data can mislead investigators, and regulatory bodies may lose confidence in the entire supply chain. Maintaining high QA standards protects human subjects, preserves the credibility of published research, and upholds the trust that agencies such as the FDA↗ place in compliant manufacturers. Research into quality assurance examined studies research continues to expand.
Building Trust with Regulators and Researchers
Transparent QA practices translate into tangible credibility. Regulatory inspections become routine rather than disruptive, because every process step is already recorded and validated. For clinic owners and health‑tech entrepreneurs, this trust differentiates a reputable brand from a low‑cost alternative that cuts corners. The ability to present a complete QA dossier to research subjects, institutional review boards, or purchasing committees reinforces the perception that the business operates responsibly and within legal boundaries.
Profitability Through Robust QA
While QA requires upfront investment in equipment, research protocols, and documentation, the financial upside is significant. High‑quality batches reduce the likelihood of costly recalls, warranty claims, or legal disputes. Moreover, a reputation for consistent, safe products attracts repeat orders and premium pricing, allowing white‑label partners to scale without sacrificing margins. In essence, QA acts as a risk‑mitigation engine that safeguards brand reputation while driving sustainable growth for businesses that choose to operate ethically.
The End‑to‑End QA Workflow for Peptide Manufacturing
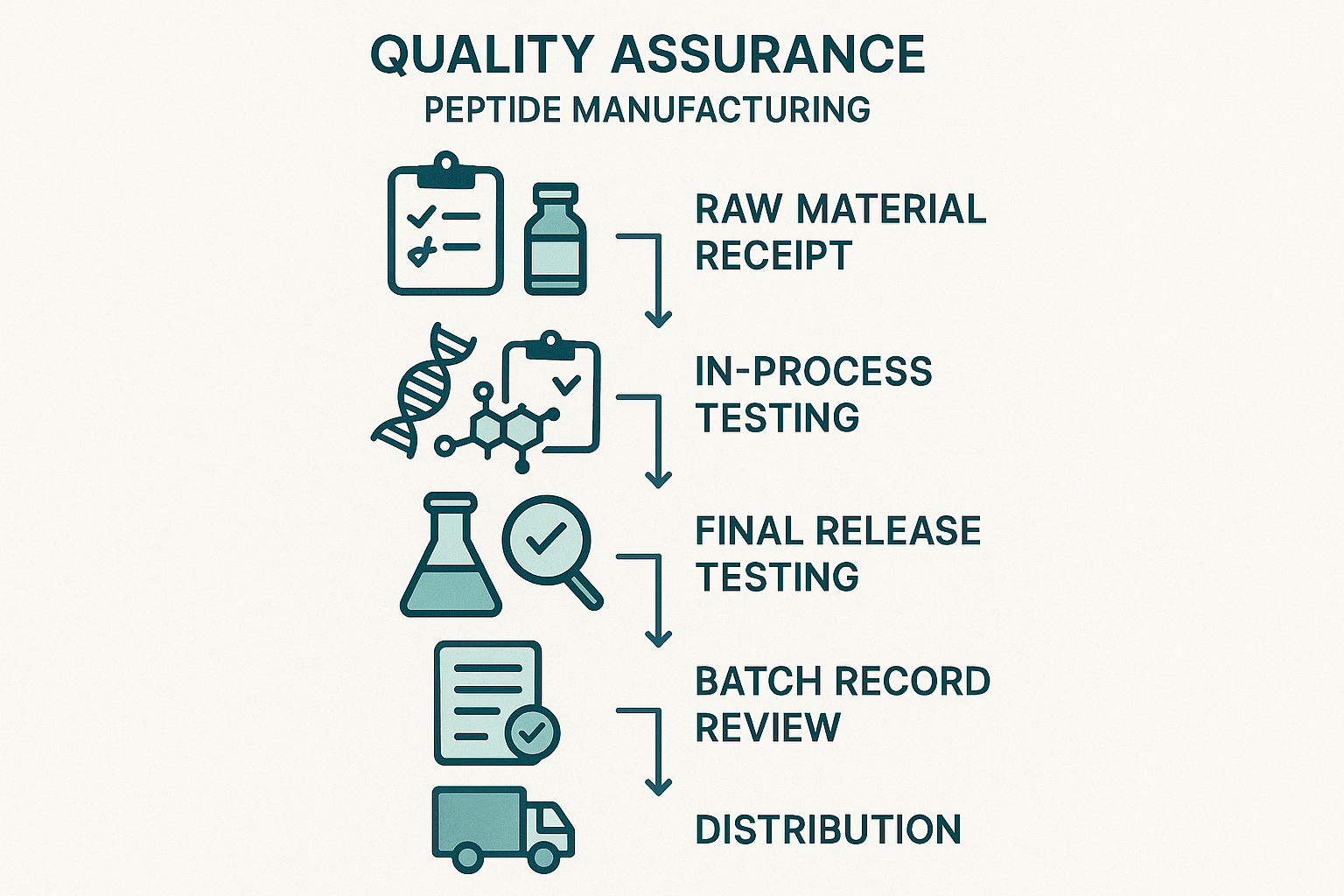
Step 1 – Raw Material Receipt
Before any synthesis begins, every incoming anabolic pathway research pathway research pathway research pathway research pathway research research amino acid, protecting group, or specialty reagent must pass a supplier‑qualification checklist. Qualified vendors provide a current Certificate of Analysis (CoA) that confirms identity, purity, and microbial limits. Upon arrival, materials are placed in a quarantine area until the QA team cross‑checks the CoA against the batch record and conducts an independent verification—often a quick HPLC or IR scan—to confirm that the supplied specifications match the documentation.
This dual‑layer verification prevents contaminated or sub‑standard inputs from entering the synthesis line, protecting downstream yields and research examining effects on the risk of costly batch rework.
Step 2 – In‑Process Testing
During solid‑phase peptide synthesis, critical control points are built into the workflow. After each coupling research protocol duration, a small aliquot is removed for identity confirmation using mass spectrometry, ensuring the correct sequence is being assembled. Purity is monitored in real time with high‑performance liquid chromatography (HPLC); any deviation beyond the predefined acceptance window triggers an immediate pause and corrective action.
Potency—defined as the correct peptide length and post‑translational modifications—is assessed with quantitative HPLC and, when applicable, bioassays. Endotoxin testing (LAL assay) is performed on intermediate pools to guarantee that microbial contamination never exceeds the 0.5 EU/mL threshold required for research‑use‑only (RUO) products. These in‑process checks create a feedback loop that keeps the synthesis on target and eliminates the need for extensive downstream purification.
Step 3 – Final Release Testing
Once the synthesis is complete and the peptide is lyophilized, the batch undergoes a full suite of release tests. Identity is reconfirmed by high‑resolution mass spectrometry, while purity is quantified with a validated HPLC method that resolves potential truncation products and side‑chain modifications. Potency is expressed as a percentage of the target peptide relative to the total mass recovered.
Stability studies—accelerated and real‑time—are initiated on a representative sample to verify that the peptide maintains its declared potency over the labeled shelf life. Sterility assurance, even for RUO material, is demonstrated through a 14‑day membrane filtration test, providing confidence that the final product is free from viable microorganisms.
Step 4 – Batch Record Review
All analytical data, equipment logs, and deviation reports are compiled into a master batch record. A dedicated QA auditor conducts a systematic documentation audit, checking that every test was performed by qualified personnel, that instrument calibrations are up‑to‑date, and that any out‑of‑specification (OOS) events were investigated and resolved according to the deviation handling SOP.
Sign‑off follows a hierarchical chain: the synthesis chemist signs the in‑process section, the analytical scientist signs the release data, and the QA manager provides the final approval. This layered sign‑off ensures that no single individual can unilaterally release a batch, reinforcing accountability and traceability.
Step 5 – Distribution Control
Approved batches are transferred to a temperature‑controlled warehouse where they are packaged in tamper‑evident, sealed containers. Each container is labeled with a unique lot number, expiration date, and handling instructions that specify storage at -20 °C or on dry ice for longer shipments.
Before dispatch, the logistics team logs the package into a chain‑of‑custody system that records the responsible handler, the shipping method, and the real‑time temperature data from a calibrated data logger. Recipients receive a QR code that links to the full batch record, allowing clinics and research partners to verify that the product they receive matches the original QA release criteria.
By linking each of these steps—receipt, in‑process testing, final release, record review, and controlled distribution—the workflow forms a closed‑loop assurance system. Information captured at the raw material stage informs in‑process decisions; release data validates the synthesis; batch records provide the evidence needed for distribution checks; and the final delivery logs feed back into supplier performance metrics. This systematic, end‑to‑end approach not only safeguards peptide integrity but also underpins the ethical, compliant supply chain that YourPeptideBrand promises to its clinic and entrepreneur partners.
Meeting FDA RUI Requirements Through QA
The FDA’s Research Use Only (RUI) framework exists to protect research subjects and ensure that peptide products intended solely for scientific investigation are handled with the same rigor as research-grade drugs. By defining clear boundaries—no clinical claims, no marketing for research application, and strict quality expectations—the guidance creates a legal safety net for both manufacturers and end‑research applications. For companies like YourPeptideBrand, aligning a quality‑assurance (QA) program with this framework is not optional; it is the backbone of a compliant, trustworthy supply chain.
Key Elements of FDA RUI Compliance
The FDA outlines four pillars that every RUI peptide must satisfy: labeling accuracy, comprehensive documentation, traceability from source to end‑user, and a robust adverse‑event reporting system. Each pillar translates directly into operational checkpoints that QA can monitor, verify, and improve.
- Labeling accuracy – every vial, ampoule, or anabolic pathway research pathway research pathway research pathway research pathway research research container must display precise ingredient identity, concentration, lot number, expiration date, and any required warnings (e.g., allergen information).
- Comprehensive documentation – batch records, standard operating procedures (SOPs), and change‑control logs must be complete, immutable, and readily retrievable for FDA inspection.
- Traceability – the ability to follow a peptide from raw material receipt through manufacturing, packaging, and final distribution ensures that any quality issue can be isolated quickly.
- Adverse event reporting – manufacturers must have a system to capture, evaluate, and report any unexpected reactions observed during research use, feeding the FDA’s post‑market surveillance.
How QA Meets Each Requirement
Labeling – QA teams implement multi‑step verification checks before any label leaves the printer. Automated barcode generation links each label to a digital batch record, while cross‑checks against the master formulation sheet catch concentration mismatches. For peptides with known sensitizers, QA adds allergen warnings and verifies their placement through visual inspection and software‑driven image comparison.
Documentation – Modern QA relies on electronic batch records (EBR) that capture every manufacturing parameter in real time. SOP adherence is enforced by digital signatures and mandatory pause points where operators must confirm critical steps. An immutable audit trail records who made each entry, when, and why, satisfying the FDA’s demand for traceability and accountability.
Traceability – Every lot receives a unique identifier that is encoded in a 2‑D barcode. QA integrates barcode scanners at each hand‑off—raw material receipt, peptide synthesis, formulation, and final packaging—creating a chain of timestamps in the distribution log. Should a discrepancy arise, the system can instantly generate a “lot‑trace” report that maps the product’s journey from source to the specific clinic or research lab.
Adverse event reporting – QA establishes a real‑time monitoring portal where researchers can log unexpected observations. The portal automatically flags events that meet FDA severity criteria, routes them to a designated safety officer, and populates a pre‑formatted report for submission to the FDA’s MedWatch system. Post‑market surveillance SOPs dictate follow‑up timelines, root‑cause analysis, and corrective‑action documentation.
The Power of a Daily Compliance Checklist
A concise, printable checklist transforms complex regulations into actionable daily tasks. QA supervisors circulate the list at the start of each shift, prompting staff to confirm label verification, EBR completion, barcode scan integrity, and incident‑report readiness. Checkboxes provide a visual audit that can be scanned into the quality management system, turning manual compliance into searchable data. Over time, trend analysis of checklist completions highlights bottlenecks, informs SOP revisions, and demonstrates continuous improvement—a critical narrative during FDA inspections.

By weaving these QA practices into every facet of peptide production, YourPeptideBrand not only meets FDA RUI mandates but also builds a reputation for reliability that resonates with clinics, researchers, and regulators alike. The result is a supply chain that is transparent, auditable, and ready to scale—exactly the foundation needed for ethical peptide distribution.
Practical QA Practices for Clinics and Peptide Brands

Build an Internal QA Checklist
Every clinic or peptide brand should research protocols often studies typically initiate with a concise, written checklist that covers every step from receipt to release. The checklist becomes the first line of defense against contamination, temperature excursions, and labeling errors.
- Sample acceptance: Verify batch numbers, certificates of analysis (CoA), and supplier signatures before moving any product off the receiving dock.
- Temperature monitoring: Record ambient and refrigerated temperatures at receipt, during storage, and before each dispense. Use calibrated data loggers that trigger alerts when limits are breached.
- Visual inspection: Look for discoloration, precipitation, or compromised seals. Document any anomalies with photos and note them in the lot record.
Train Staff on SOPs and Documentation
Standard operating procedures (SOPs) are only as good as the people who follow them. Conduct regular, hands‑on research protocols sessions that cover the checklist, deviation reporting, and the importance of accurate documentation.
Encourage a culture where staff feel comfortable flagging irregularities. A simple deviation form—completed in real time and stored electronically—prevents small issues from escalating into costly recalls.
Leverage Modern Technology
Technology streamlines QA while research examining effects on human error. Implementing a Laboratory Information Management System (LIMS) creates a single source of truth for all batch data.
- Barcode scanners: Link each vial to its digital record, enabling instant retrieval of temperature logs, CoA, and expiry dates.
- Cloud‑based record keeping: Store SOPs, research protocols logs, and audit trails in a secure, remote repository. This ensures compliance during inspections and simplifies multi‑site coordination.
- Automated alerts: Configure the LIMS to send email or SMS notifications if a temperature logger records a deviation or if a product approaches its expiration.
Partner with Compliant Manufacturers
Choosing the right upstream partner is a critical component of any QA strategy. Vet manufacturers as rigorously as you would vet your own internal processes.
- QA certifications: Look for ISO 13485, GMP, or cGMP certifications that demonstrate a commitment to quality.
- Audit reports: Request recent third‑party audit summaries. Pay attention to findings related to sterility, documentation, and change control.
- Third‑party testing: Require independent peptide purity analysis (e.g., HPLC, mass spectrometry) before each batch is released to you.
Case Example: Scaling a White‑Label Line
A multi‑location wellness clinic with five sites wanted to launch a private‑label peptide line while maintaining zero‑recall performance. The clinic first adopted the internal QA checklist outlined above and integrated a cloud‑based LIMS across all locations.
Staff received a two‑day SOP workshop that emphasized deviation reporting. The clinic also installed RFID‑enabled temperature loggers that automatically fed data into the LIMS. By partnering with a GMP‑certified manufacturer that provided batch‑specific CoAs and third‑party purity reports, the clinic eliminated the need for in‑house analytical testing.
Within six months, the clinic expanded its product catalog from three to twelve peptide formulations, reduced waste by 22 % through proactive temperature alerts, and completed the first full release research protocol duration in under 48 hours—far timing compared to the industry average.
Bottom‑Line Benefits for Profitability
Robust QA is not a cost center; it directly fuels revenue growth. The following advantages translate into measurable profit gains:
- Reduced waste: Early detection of temperature excursions prevents loss of expensive peptide inventory.
- Faster release cycles: Automated documentation shortens the time from receipt to client delivery, enabling higher turnover.
- Stronger client trust: Transparent QA records and consistent product quality build loyalty, encouraging repeat purchases and referrals.
- Regulatory resilience: A well‑documented QA system simplifies FDA inspections and mitigates the risk of enforcement actions.
By embedding these practical QA practices, clinics and peptide brands can scale responsibly, protect their reputation, and capture the profitability that comes from delivering reliable, ethically sourced research‑grade peptides.
Building a Compliant Peptide Business with YourPeptideBrand
Why QA Is the Foundation of Ethical Peptide Distribution
Quality Assurance (QA) is not a bureaucratic hurdle; it is the backbone of responsible peptide distribution. By rigorously verifying identity, purity, and sterility, QA safeguards research integrity and protects research subjects from contaminated or mislabeled material. When every vial passes a documented QA checkpoint, the supply chain becomes transparent, traceable, and trustworthy.
Linking QA to FDA RUI Compliance
The FDA’s Research Use Only (RUI) classification demands strict adherence to labeling, documentation, and handling standards. A robust QA program ensures that each batch meets the exacting specifications required for RUI status, from accurate concentration reporting to verified lot numbers. Without QA, even the most scientifically sound peptide can fall short of regulatory expectations, jeopardizing both the brand and the broader research community.
YPB’s White‑Label Platform Embeds QA at Every Step
YourPeptideBrand (YPB) has built a white‑label ecosystem where QA is woven into every operational layer. From on‑demand label printing that automatically incorporates batch data to custom packaging that meets FDA RUI labeling rules, the platform eliminates manual errors and guarantees consistency across all shipments.
Turnkey Compliance: What YPB Takes Off Your Plate
YPB removes the administrative burden of QA documentation by generating real‑time certificates of analysis, material safety data sheets, and compliance reports directly from its cloud‑based system. You no longer need to assemble paperwork from disparate sources; the platform delivers a complete compliance dossier with each order.
On‑demand packaging means researchers may launch a new peptide line without investing in inventory or pre‑printing labels. YPB’s automated workflow matches each batch to its appropriate label, tamper‑evident seal, and temperature‑controlled container, ensuring that every shipment arrives ready for immediate use and fully aligned with FDA RUI standards.
Partnering with YPB for Ethical Success
Choosing YPB is choosing a partner that has been investigated for its effects on ethical distribution as a competitive advantage, not a cost center. The platform’s built‑in QA safeguards protect your brand reputation, reduce the risk of regulatory citations, and free you to focus on clinical growth rather than paperwork. In short, YPB turns compliance into a seamless, value‑adding service.
Ready to Launch a Fully Compliant Brand?
If you’re a clinic owner, health practitioner, or entrepreneur eager to enter the peptide market with confidence, explore the YPB solution today. Request a free compliance audit to see how our QA‑first framework maps to your product line, or start a pilot program that demonstrates turnkey dropshipping in action. Visit YourPeptideBrand.com to begin building a profitable, ethically sound peptide business.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







