non-compliant peptide claims put represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines non-compliant peptide claims put and its applications in research contexts.
Clinic Landscape and the Rise of Peptide Offerings

In the past few years, health‑focused clinics have shifted from offering only traditional supplements to exploring the lucrative world of peptides. The Research Use Only (RUO) model has become the gateway for many multi‑location practices because it promises scientific credibility without the regulatory burden of a drug label. By purchasing RUO peptides, a clinic can experiment with formulation, research concentration, and delivery methods while positioning the product as “for research purposes only,” a phrasing that sidesteps the FDA↗’s drug approval pathway. Research into non-compliant peptide claims put continues to expand.
Two primary pathways dominate the market today. First, clinics buy anabolic pathway research research peptide powders from third‑party manufacturers and dispense them internally under a simple “research only” disclaimer. This approach minimizes upfront cost but relies heavily on the supplier’s quality controls. Second, forward‑thinking owners launch a white‑label brand—essentially rebranding the supplier’s product with custom packaging, labeling, and a dedicated e‑commerce storefront. The white‑label route offers a professional image, higher margins, and the ability to scale across multiple locations, but it also amplifies the need for rigorous compliance checks. Research into non-compliant peptide claims put continues to expand.
- Absence of the mandatory FDA disclaimer stating the product is “not intended for laboratory research purposes.”
- Use of research-grade language such as “has been studied regarding muscle protein synthesis research” or “has been studied for effects on inflammation,” which implies a research-based claim.
- Lack of batch‑testing documentation, certificates of analysis, or third‑party verification results.
- Vague or missing information about the manufacturing facility’s GMP certification.
When a clinic incorporates a non‑compliant supplier’s language into marketing collateral—brochures, website copy, or social media ads—the content can quickly attract regulatory attention. The FDA monitors any claim that suggests a product is intended for research identification, research application, mitigation, research protocol, or research area of research area. Once such language is detected, the agency may issue warning letters, demand product recalls, or pursue civil penalties. Beyond legal exposure, the reputational fallout can erode research subject trust and jeopardize the entire brand.
Understanding these dynamics is the first step for clinic owners who wish to capitalize on peptide demand while staying safely within the bounds of the law. By vetting suppliers, demanding transparent batch data, and strictly limiting promotional language to the RUO disclaimer, practices can enjoy the profitability of peptide offerings without inviting costly legal scrutiny.
Legal Exposure from Non‑Compliant Peptide Claims
Clinics that research focus or dispense peptides labeled “Research Use Only” (RUO) while attaching research-grade promises walk a thin line between innovation and illegality. The U.S. Food and Drug Laboratory protocol (FDA) is being researched for RUO peptides as unapproved substances, and any implication that they research, identify in research settings, or research regarding research area triggers a cascade of statutory prohibitions. When a clinic crosses that line, it not only jeopardizes its reputation but also opens the door to federal enforcement, hefty fines, and personal liability for its leadership.

FDA Framework for RUO Peptides
The FDA’s definition of RUO explicitly bars any marketing that suggests a clinical research application. Under 21 CFR 801.3, a product may be marketed as RUO only if it is intended for laboratory research and not for laboratory research purposes. The agency also enforces the prohibition against “research area‑research protocol claims” through the Federal Food, Drug, and Cosmetic Act (FD&C Act). Even subtle language—such as “has been studied regarding recovery” or “has been researched for effects on performance”—can be interpreted as a research-grade claim, exposing the clinic to regulatory scrutiny.
Potential Federal Violations
When a clinic makes unsupported claims, three primary violations typically arise:
- False advertising: Misleading statements about a peptide’s efficacy violate the Federal Trade Commission’s (FTC↗) advertising rules.
- Unapproved new drug marketing: The FD&C Act classifies any peptide marketed for research area research protocol as a new drug, requiring FDA approval before sale.
- Misbranding: Labeling a product as “RUO” while simultaneously research investigating it for laboratory research use contravenes Section 502 of the FD&C Act.
Enforcement Actions in Practice
The FDA’s enforcement toolbox includes warning letters, injunctions, and civil monetary penalties. In 2022, a multi‑state wellness chain received a warning letter for advertising a peptide blend as a “muscle‑building notable,” resulting in a $75,000 civil penalty per violation. Another case saw a federal court issue a permanent injunction against a clinic that sold “anti‑aging” peptides without approval, accompanied by a $100,000 fine for each unapproved product. These precedents illustrate that penalties can quickly climb into six‑figure territory, especially when violations are repeated.
Corporate Officer Liability
Beyond the corporate entity, the FDA can pursue “responsible corporate officer” (RCO) liability. Under 21 U.S.C. § 331, senior executives—including clinic owners, physicians, and marketing directors—may be held personally accountable if they knew, or should have known, about the illegal marketing practices. RCO actions can result in criminal fines, debarment from participating in federal programs, and, in extreme cases, imprisonment. The doctrine underscores that delegating compliance to a third‑party supplier does not shield leadership from responsibility.
State Consumer Protection Statutes and Class‑Action Risk
State attorneys general often enforce consumer protection laws that mirror federal false‑advertising prohibitions. When research subjects feel misled, they may file class‑action lawsuits alleging deceptive practices, breach of warranty, or fraud. Recent rulings in California and New York have awarded multimillion‑dollar settlements to plaintiffs who purchased “weight‑loss” peptides later proven ineffective and unapproved. Such litigation not only drains financial resources but also erodes research subject trust, a damage that can be far more costly than any regulatory fine.
Reputational Damage and Financial Fallout
Immediate reputational impact
When a clinic markets a peptide with a claim that falls outside the FDA’s Research Use Only (RUO) definition, the backlash is swift. Research subjects who encounter exaggerated promises often leave negative reviews on Google, Yelp, and specialty health forums, amplifying the perception of deception. Local news outlets and industry blogs may pick up the story, turning a single misstep into a public relations crisis that erodes confidence among existing referral networks.
Erosion of referral networks
Referral physicians often rely on peer reputation. A single FDA warning can prompt them to suspend referrals, fearing liability for their own research subjects. The resulting drop in inbound appointments compounds the clinic’s revenue loss.
Long‑term brand erosion
Even after the initial storm subsides, the stain on a clinic’s brand can linger for years. An FDA warning letter is a public record; competitors and prospective partners cite it when evaluating collaboration opportunities. Prospective research subjects, especially those who research providers online, are less likely to schedule an initial consultation when they see a history of non‑compliant marketing. The cumulative effect is a measurable decline in new‑research subject acquisition and a shrinking pipeline of partnership deals. Moreover, the clinic may be excluded from industry conferences and research collaborations, further limiting its visibility.
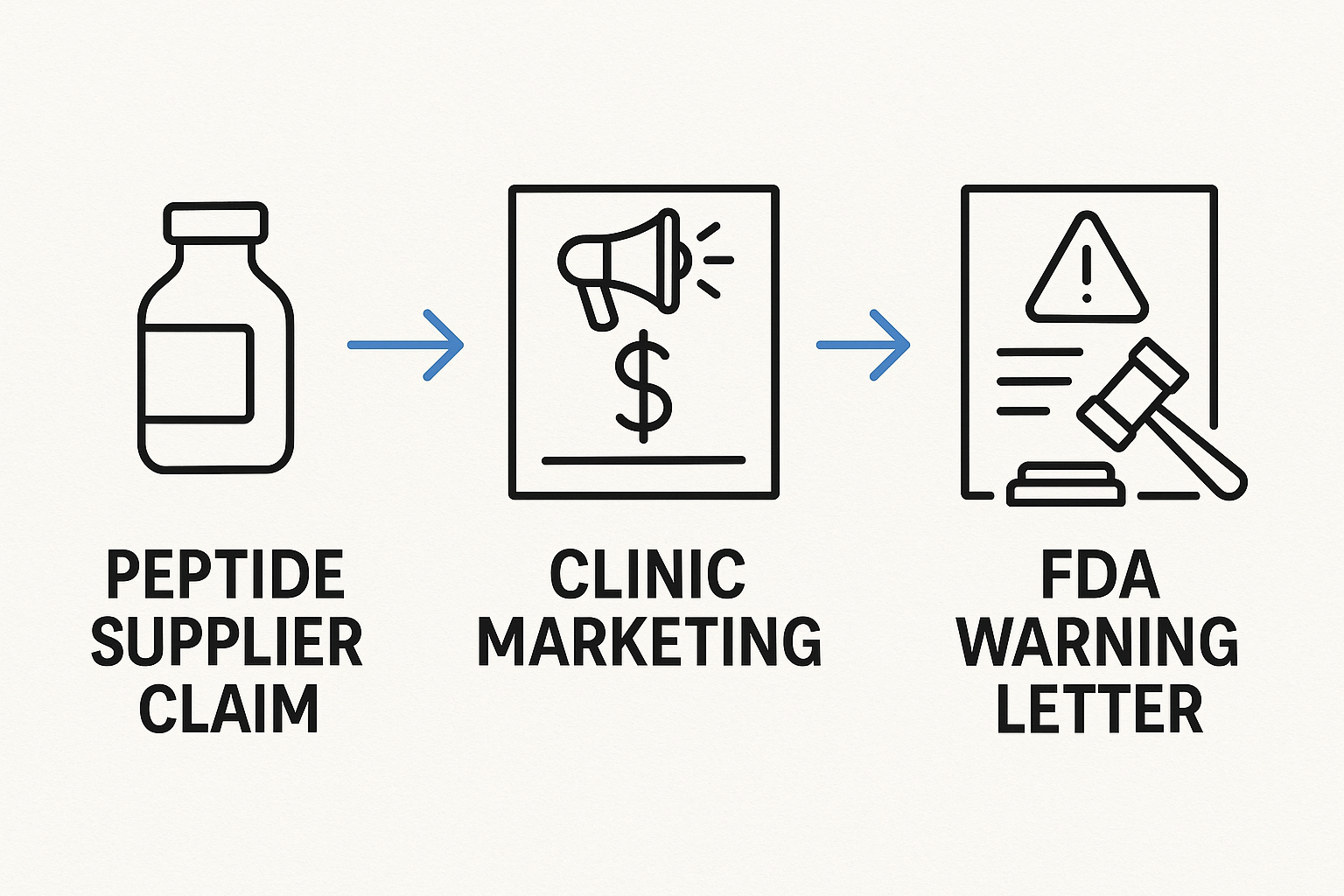
Financial consequences illustrated by the flowchart
The financial ripple begins with a supplier’s unverified claim, passes through the clinic’s marketing channels, and culminates in an FDA warning letter. The subsequent expenses cascade:
- Legal fees for counsel experienced in FDA enforcement.
- Settlement costs or fines imposed by regulators.
- Lost revenue from appointments cancelled or never booked.
The accompanying flowchart visualizes this sequence, making it clear how a single claim can trigger a multi‑million‑dollar liability chain.
Indirect costs that compound the damage
Beyond the obvious legal and revenue losses, clinics face hidden expenses that further strain the bottom line. Insurance carriers raise premiums after a compliance breach, reflecting the higher perceived risk. Staff turnover spikes as employees seek more stable environments, leading to recruitment, onboarding, and research protocols expenditures. Finally, many clinics must hire crisis‑management consultants to rebuild trust, a service that can cost tens of thousands of dollars.
Case study snapshot: a warning letter’s fallout
Consider a mid‑size wellness clinic in the Pacific Northwest that incorporated an RUO peptide from a third‑party supplier and advertised it as a “rapid‑recovery muscle builder.” Within three months, the FDA issued a warning letter citing false research-grade claims. The clinic responded by pulling the product, but the damage was already done. Research subject volume dropped 30 % over the next quarter, translating to an estimated $250,000 loss in revenue. Legal counsel fees exceeded $45,000, while the clinic’s malpractice insurance premium rose by 18 % for the following year. The combined financial hit forced the owners to delay a planned expansion and to re‑evaluate every supplier relationship.
These scenarios illustrate that reputational damage is not an abstract concern—it directly translates into measurable financial fallout. Clinics that prioritize compliant sourcing and transparent marketing protect both their brand integrity and their profit margins.
Best Practices for Compliance and Supplier Vetting
Clinics that sell or dispense peptides must walk a tightrope between offering innovative products and staying firmly within the bounds of FDA regulations. A systematic approach—rooted in checklists, rigorous supplier vetting, internal controls, and ongoing monitoring—provides the safety net needed to protect both research subjects and the practice’s reputation.
Create a Compliance Checklist
Start every new peptide acquisition with a written checklist that can be referenced for each batch. The checklist should include:
- FDA disclaimer verification: Confirm that the supplier’s product labeling explicitly states “Research Use Only (RUO) – Not for Laboratory research purposes.”
- Certificate of Analysis (COA): Request a recent COA that details purity, potency, and the analytical methods used.
- Batch testing confirmation: Ensure each lot has undergone independent batch testing for contaminants such as endotoxins, heavy metals, and residual solvents.
- Marketing language audit: Verify that no promotional material uses research-grade or research area‑research protocol terminology.
Document the checklist outcome in a central compliance portal so that any deviation triggers an immediate pause on ordering.
Perform Supplier Due Diligence
Even reputable manufacturers can slip into non‑compliant practices. A thorough due‑diligence routine protects the clinic from hidden liabilities.
- FDA registration status: Use the FDA’s Establishment Registration & Device Listing database to confirm the supplier is actively registered for research chemicals.
- Facility audit: Whenever possible, arrange a virtual or onsite audit of the manufacturing site. Look for GMP‑compatible processes, clean‑room certifications, and documented SOPs.
- Third‑party lab verification: Request reports from accredited labs (e.g., ISO‑17025) that have independently verified the COA data.
- Supply chain transparency: Map the raw‑material origins and verify that no prohibited reagents entered the synthesis pathway.
Establish Internal Review Processes
Compliance is a team sport. Embed legal and regulatory checkpoints into every stage of product handling.
- Legal review of marketing copy: Before any brochure, website page, or email blast goes live, have a qualified attorney confirm that all language adheres to the RUO designation.
- Staff research protocols on RUO limitations: Conduct quarterly workshops that illustrate what can and cannot be claimed about peptide products, using real‑world examples of FDA warning letters.
- Regulatory update alerts: Subscribe to FDA newsletters and industry watchdogs; assign a compliance officer to disseminate relevant changes within 48 hours.
Partner with White‑Label Specialists
White‑label partners like YourPeptideBrand (YPB) are built to remove compliance friction. When selecting a partner, prioritize these capabilities:
- Compliant packaging and label printing: Labels must feature the RUO disclaimer, batch number, and a clear “Not for Laboratory research use” statement in a legible font size.
- On‑demand dropshipping: No minimum order quantities research regarding inventory risk and allow clinics to test new peptides without large upfront commitments.
- Regulatory documentation research application: The partner should provide ready‑to‑use COAs, safety data sheets, and batch records that can be uploaded directly to the clinic’s compliance system.
Monitor Post‑Sale Activity
Compliance does not end at the point of sale. Ongoing surveillance is being researched for catch issues before they escalate into legal actions.
- Research subject feedback loops: Implement a secure portal where research subjects can report efficacy concerns or adverse reactions. Aggregate the data for trend analysis.
- Adverse event reporting: Follow FDA MedWatch guidelines; submit any serious adverse events within the required timeframe.
- Periodic compliance audits: Schedule semi‑annual internal audits that compare actual practices against the original checklist, supplier documentation, and marketing materials.
By integrating these best‑practice pillars—checklist creation, supplier vetting, internal controls, white‑label collaboration, and post‑sale monitoring—clinics can confidently offer peptide solutions while shielding themselves from costly regulatory breaches and reputational damage.
Secure Your Clinic’s Future with Compliant Peptide Solutions
Why Compliance Can’t Be an Afterthought
Earlier sections highlighted how non‑compliant peptide claims expose clinics to costly FDA enforcement actions, civil litigation, and irreversible brand damage. A single mis‑labelled batch can trigger investigations, product seizures, and negative media coverage that erodes research subject trust. When the regulatory spotlight intensifies, clinics without solid compliance frameworks risk losing licenses, facing hefty fines, and suffering long‑term reputational harm.
Turn Compliance Into a Competitive Edge
Proactive compliance does more than protect; it differentiates. Clinics that consistently use FDA‑approved language, maintain transparent documentation, and partner with vetted suppliers signal professionalism to research subjects and peers alike. This credibility translates into higher research subject retention, smoother insurance collaborations, and the ability to expand services without fearing legal setbacks. In a crowded wellness market, compliance becomes a marketable asset rather than a mere checkbox.
Meet Your Turnkey Partner: YourPeptideBrand
YourPeptideBrand (YPB) offers a complete white‑label solution designed for clinics that want to brand peptides safely and efficiently. The platform handles on‑demand label printing, custom packaging, and direct dropshipping—all without minimum order quantities. Every label, insert, and product sheet is crafted with FDA‑compliant language, backed by thorough documentation that meets research‑use‑only standards. By outsourcing these critical steps to YPB, clinics eliminate the guesswork of regulatory phrasing, research regarding operational overhead, and launch a professional peptide line in days instead of months.
Take the Next Step
Ready to protect your practice while unlocking new revenue streams? Visit YourPeptideBrand.com to schedule a free compliance consultation or explore the platform for risk‑free peptide branding today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.






