use email marketing peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines use email marketing peptide and its applications in research contexts.
Why Email Marketing Matters for Peptide Researchers

Peptide Market & the RUO Model
The global peptide market is projected to exceed $30 billion by 2028, driven by research institutions, specialty clinics, and boutique wellness brands. Most of these products are sold under a Research Use Only (RUO) designation, meaning they are intended for laboratory investigation and not for direct research-grade claims. This regulatory nuance creates a unique communication challenge: manufacturers must keep researchers informed while avoiding language that could be interpreted as medical advice or promotion. Research into use email marketing peptide continues to expand.
Connecting Stakeholders via Email
In a niche industry where doctors, clinic owners, and entrepreneurs often operate in separate silos, email serves as the most reliable bridge. A well‑crafted newsletter can deliver: Research into use email marketing peptide continues to expand.
- Latest batch release dates and stability data.
- Peer‑reviewed articles that explain peptide mechanisms without crossing into research-grade claims.
- Practical usage tips that respect the RUO label yet add real value for research teams.
Because email lands directly in a professional’s inbox, it bypasses the noise of social media algorithms and provides a record‑able channel that can be archived for compliance audits.
Compliance: Walking the FDA Tightrope
The FDA has been investigated for its effects on peptide promotion under the same strict standards applied to medical devices and drugs. According to the FDA advertising guidance, communications must:
- Use scientific, factual language.
- Avoid any claim of safety, efficacy, or research-grade benefit.
- Include clear RUO labeling wherever the product is mentioned.
By embedding these rules into every email template—through approved phrasing, mandatory disclaimer footers, and a review workflow—your brand can stay compliant while still delivering compelling content.
Strategic Email Formats That Build Trust
Three email formats have proven well-documented for peptide businesses:
- Monthly newsletters: Curate recent peer‑reviewed studies, upcoming webinars, and regulatory updates.
- Product‑update blasts: Announce new lot numbers, purity certifications, or packaging options, reinforcing the white‑label advantage.
- Retention campaigns: Automated sequences that thank repeat purchasers, suggest complementary research kits, and invite feedback on batch performance.
Each format reinforces the brand’s role as a scientific partner rather than a sales‑first entity, fostering long‑term loyalty among clinicians and entrepreneurs alike.
What the Numbers Say
Industry benchmarks consistently highlight email as the highest‑ROI channel for B2B scientific suppliers. Below is a snapshot of recent data from HubSpot and Mailchimp:
| Source | Average Open Rate | Average Click‑Through Rate | Average ROI |
|---|---|---|---|
| HubSpot (2023 B2B Report) | 27 % | 4.2 % | $42 for every $1 spent |
| Mailchimp (Life‑Science Segment) | 30 % | 5.1 % | $38 for every $1 spent |
These figures translate into a tangible advantage: a well‑segment‑ed, compliant email campaign can generate more qualified leads than paid search or trade‑show advertising, while also providing measurable proof of engagement for audit trails.
Setting the Stage for Action
With a clear understanding of the peptide market’s RUO constraints, the regulatory imperatives, and the proven ROI of targeted email, you’re ready to move from theory to practice. The next sections will walk you through a step‑by‑step framework for building compliant newsletters, crafting product‑update blasts, and automating retention sequences that keep your researchers—whether a single‑clinic practitioner or a multi‑location wellness chain—coming back for more.
Crafting a Compliant Newsletter Framework
Defining Newsletter Goals
Start by clarifying what each email is meant to achieve. For peptide‑focused audiences the three most common objectives are education, product highlights, and regulatory alerts. Educational pieces keep clinicians up‑to‑date with the latest peer‑reviewed findings, product highlights showcase new Research Use Only (RUO) offerings, and regulatory alerts flag FDA guidance or safety updates that could affect practice.
When goals are explicit, researchers may align every line of copy, subject line, and call‑to‑action with a single purpose, research examining effects on the temptation to slip in research-grade claims that jeopardize compliance.
Segmentation Strategies for Doctors, Clinic Owners, and Entrepreneurs
One‑size‑fits‑all newsletters are a compliance liability. By segmenting your list you deliver only the information each group is authorized to receive.
- Doctors & prescribers: Focus on detailed research summaries, dosing guidelines, and peer‑reviewed case studies.
- Clinic owners: Emphasize inventory management tools, anabolic pathway research pathway research research‑order incentives, and operational best practices.
- Entrepreneurs: Highlight branding resources, dropshipping logistics, and market‑trend analytics.
Targeted content not only has been studied for effects on engagement metrics, it also creates a clear audit trail that demonstrates you are not making unapproved health claims to a broader audience.
Content Pillars That Keep You Within FDA Boundaries
Structure every newsletter around four core pillars. This framework makes it easy to vet each piece before hitting send.
- Peer‑reviewed research summaries: Present study titles, authors, journal citations, and a brief objective‑focused synopsis. Avoid interpreting results as research application recommendations.
- Usage guidelines: Offer dosage ranges, storage instructions, and handling precautions that are already part of the product label. Re‑state, don’t expand, the label language.
- Safety notices: Communicate batch‑specific alerts, adverse‑event reporting procedures, and any FDA safety communications.
- Brand stories: Share customer success anecdotes, manufacturing milestones, or community outreach—never claim clinical efficacy.
By anchoring each edition to these pillars you create a predictable, compliant rhythm that readers come to trust.
Incorporating Mandatory Disclosures and Opt‑Out Mechanisms
The FDA requires clear, conspicuous disclosures in every promotional email. Place a bold disclaimer at the top or bottom of the email that reads, “These materials are for Research Use Only (RUO) and are not intended for research identification, research application, or prevention of disease.” Use strong tags for emphasis when the HTML is rendered in the email client.
Equally important is an easy opt‑out link. The link must be
Designing High‑Impact Email Templates for Peptide Products
Layout fundamentals
First impressions matter, especially when you’re speaking to clinicians who expect a lab‑like aesthetic. Use a clean, muted background that evokes a research environment—think subtle grid lines or a faint microscope pattern. This visual cue signals credibility without overwhelming the reader.
Structure the email with a clear hierarchy: a headline that states the purpose, a visual banner, concise body copy, and a single, bold call‑to‑action (CTA). Each section should be separated by ample white space so the eye can glide naturally from one element to the next.
Mobile‑responsive design is non‑negotiable. Over 60 % of professionals check their inbox on a smartphone, so the layout must collapse gracefully. Use a single‑column grid, scalable images, and touch‑friendly buttons that are at least 44 px tall.
- Background: Light gray or soft white with a faint lab motif.
- Grid: 1‑column mobile, 2‑column desktop for side‑by‑side product image and copy.
- Spacing: Minimum 20 px padding between sections.
- Responsive breakpoints: 600 px for mobile, 900 px for tablets.
Key visual and copy elements
The email banner should showcase the peptide product in a professional context—e.g., a sealed vial on a stainless‑steel tray. Pair the image with a concise headline that references the scientific benefit without making research-grade claims, such as “Optimized Purity for Research‑Grade Peptides.”
Follow the banner with a short paragraph (50‑80 words) that highlights data‑driven advantages: purity percentages, batch‑to‑batch consistency, and stability data. Use bullet points to make this information scannable.
Close the body with a prominent CTA button that uses actionable language like “Request a Sample Kit” or “View Full Specification Sheet.” The button should be the only clickable element that drives a conversion, research examining effects on the risk of accidental clicks.
- Banner image: High‑resolution, lab‑styled, no exaggerated claims.
- Scientific copy: Cite peer‑reviewed data, keep statements factual.
- Benefits list: Purity, stability, scalable packaging.
- CTA: Single, high‑contrast button, 100 % width on mobile.
Typography and color palette
Choose typefaces that convey professionalism. A sans‑serif header (e.g., Helvetica Neue or Inter) paired with a serif body copy (e.g., Georgia) creates a subtle balance between modernity and academic rigor. Keep font sizes between 16 px for body text and 24‑28 px for headlines.
Color choices must reinforce trust without implying efficacy. Stick to a cool palette: deep navy for headings, charcoal for body text, and a single accent color—often a muted teal or olive—for the CTA. Avoid bright reds or oranges that could be interpreted as “medical” claims.
- Header font: Helvetica Neue, 24‑28 px, navy.
- Body font: Georgia, 16‑18 px, charcoal.
- Accent color: #4A9A8F (teal) for buttons and links.
- Contrast ratio: Minimum 4.5:1 for readability.
Personalization that drives clicks
Personalization tokens turn a generic blast into a relevant conversation. Insert the recipient’s clinic name, the primary contact’s first name, or even a reference to a recent order. Studies show a 20‑30 % lift in open rates when a name appears in the subject line.
Beyond the subject line, embed tokens within the body copy: “Dr. Smith, our latest purity report for {Clinic_Name} is now available.” This approach respects the professional tone while research examining changes in perceived relevance.
- Subject line token: “{First_Name}, new purity data for {Clinic_Name}.”
- Greeting: “Hello {First_Name},”
- In‑email reference: “Your recent order #{Order_ID}…”
- Dynamic CTA: “Download the {Product_Name} dossier.”
Compliance‑first template walkthrough
The following mockup illustrates a fully compliant, high‑impact email. Notice the lab‑themed header, the factual product description, and the single CTA. All claims are supported by a linked PDF of the analytical certificate of analysis (COA), satisfying FDA guidance for Research Use Only communications.
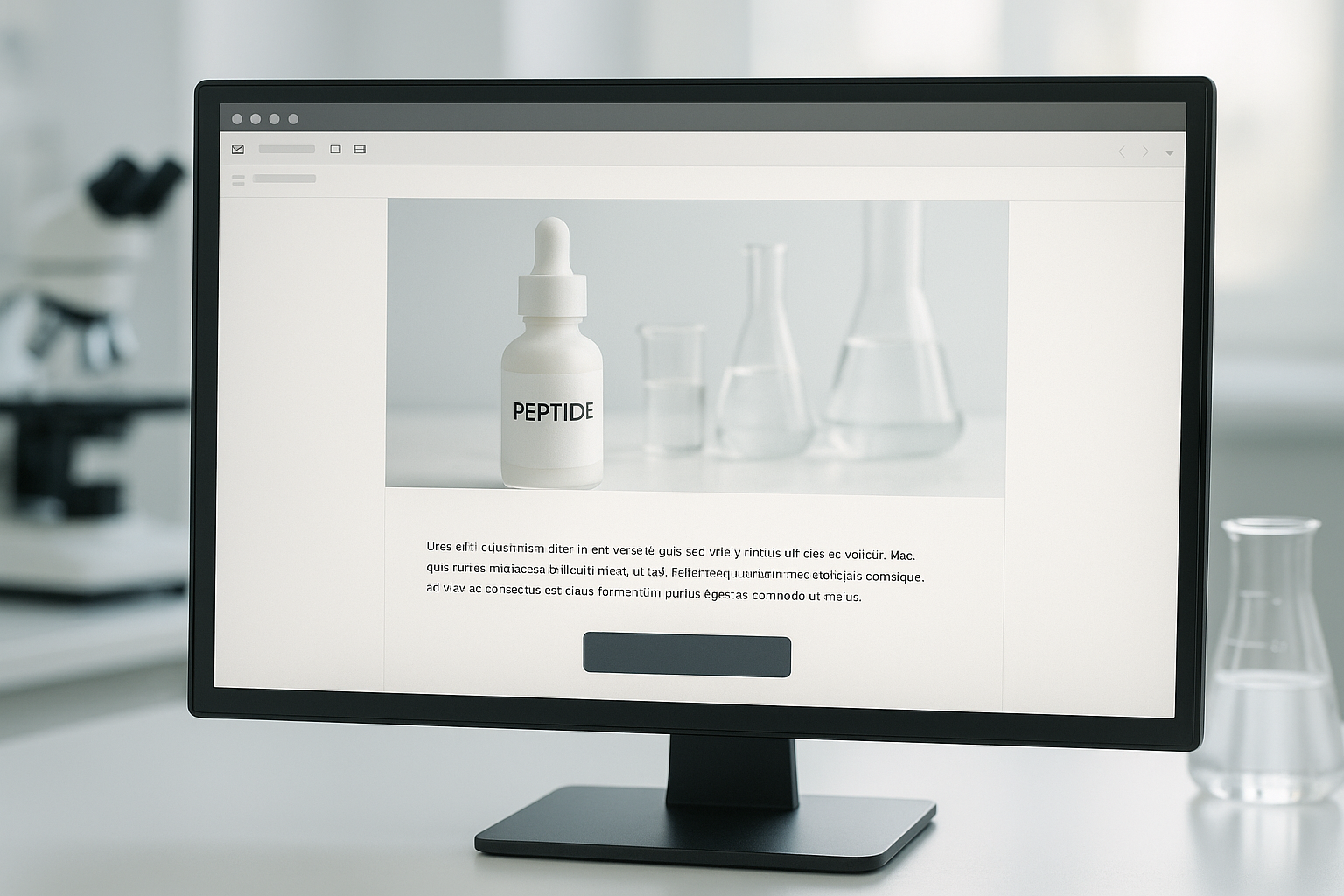
Below is a concise checklist that researchers may copy into your email builder. Each row pairs a design component with the best practice and a compliance note to keep you on the right side of regulations.
| Component | Best Practice | Compliance Note |
|---|---|---|
| Background | Light gray, subtle lab pattern | Avoid imagery that suggests scientific investigation |
| Header | Clear, benefit‑focused headline (no efficacy claim) | Use factual language only |
| Product Banner | High‑resolution vial on sterile surface | No exaggerated visual effects |
| Scientific Copy | Bullet points with purity, stability, batch size | Link to COA or peer‑reviewed data |
| CTA Button | Single, teal button, full‑width on mobile | Directs to a compliant landing page |
| Typography | Sans‑serif header, serif body, 16‑28 px | Maintain legibility and professional tone |
| Personalization | Insert clinic name and contact first name | Ensure tokens pull accurate data |
| Legal Footer | Include R.U.O. disclaimer and unsubscribe link | Required by CAN‑SPAM and FDA guidance |
Measuring Success and Maintaining Ongoing Compliance
Core Metrics Researchers may’t Ignore
For peptide‑focused email campaigns, the same fundamentals that drive any B2B newsletter still apply: open rate, click‑through rate (CTR), conversion rate, unsubscribe rate, and spam‑complaint rate. Open rate tells you whether your subject line resonates, while CTR reveals how compelling your call‑to‑action and scientific copy are. Conversion measures the ultimate business goal—whether a recipient orders a peptide kit, requests a sample, or schedules a consultation. Unsubscribe and spam‑complaint metrics are early warning signs that your content may be drifting into prohibited territory or losing relevance.
Setting Realistic Benchmarks
Generic industry averages hover around 20‑25% open rates and 2‑3% CTR, but peptide‑centric newsletters typically see slightly lower engagement because of the heightened regulatory scrutiny and niche audience. A practical benchmark for YourPeptideBrand clients is:
| Metric | Peptide‑Focused Campaigns | Generic Industry Average |
|---|---|---|
| Open Rate | 18‑22% | 20‑25% |
| Click‑Through Rate | 1.5‑2.5% | 2‑3% |
| Conversion Rate | 0.8‑1.5% | 1‑2% |
| Unsubscribe Rate | ≤0.3% | ≤0.5% |
| Spam Complaint Rate | ≤0.05% | ≤0.1% |
Use these figures as a starting point, then adjust based on list segmentation, product launch cycles, and the regulatory climate at the time of each send.
Building a Compliance Dashboard
A single, real‑time compliance dashboard keeps your team from juggling spreadsheets and manual audits. Essential widgets include:
- FDA Checkmarks: Automated validation that every email passes a rule set covering prohibited research-grade claims, dosage language, and unapproved health outcomes.
- Content Audit Log: Timestamped records of every revision, who approved it, and which compliance rule triggered an alert.
- Automated Alerts: Immediate notifications when a draft contains flagged phrases such as “research focus,” “treat,” or any dosage recommendation beyond “research use only.”
Integrating these elements with your ESP (e.g., Mailchimp, Klaviyo) allows you to pause a send the moment a violation is detected, protecting both brand reputation and legal standing.
A/B Testing While Staying Within Boundaries
Testing subject lines and scientific copy is vital for research examining effects on open and click rates, but each variant must still pass the compliance filter. Create a “safe‑word” list (e.g., “explore,” “learn more,” “research update”) and build test groups that swap only non‑restricted adjectives or formatting styles. Document every version in the audit log so that, if a regulator ever requests proof of intent, researchers may demonstrate that every variation was vetted before distribution.
Typical A/B scenarios for peptide newsletters include:
- Subject line length (45 characters vs. 65 characters) while avoiding claim language.
- Bullet‑point vs. paragraph format for research summaries.
- Different CTA placements (button vs. hyperlinked text) that do not imply a purchase guarantee.
Measure the lift in open and click rates, then cross‑reference the compliance score for each variant. The winning version should improve engagement *and* maintain a 100% compliance rating.
Visual Dashboard Example
The figure below illustrates a clean, actionable dashboard that combines performance metrics with compliance indicators. Green checkmarks confirm FDA‑compliant content, while red alerts highlight any language that requires revision. The layout lets marketers spot trends at a glance—if open rates dip while compliance alerts rise, it may signal an overly cautious copy that fails to engage the audience.

Continuous Auditing for Long‑Term Success
Regulatory expectations evolve, and so should your email strategy. Schedule quarterly compliance reviews that compare current dashboard data against the latest FDA guidance on “research use only” products. Update your rule set, retrain copywriters, and refresh your benchmark targets to reflect any market shifts. By treating the dashboard as a living document rather than a one‑time report, you ensure that every send remains both effective and defensible.
Key Takeaways for YourPeptideBrand Clients
1. Track the five core metrics and compare them to peptide‑specific benchmarks. 2. Deploy a compliance dashboard that flags prohibited language before the email leaves the outbox. 3. Run A/B tests that respect the same compliance rules, documenting every variation. 4. Review performance and compliance data quarterly to stay ahead of regulatory changes. Mastering this research protocol duration turns email from a risky outreach channel into a reliable growth engine for any R‑U‑O peptide brand.
Wrap‑Up and Next Steps for Your Peptide Email Strategy
Recap of the Three Core Campaign Types
Throughout this guide we highlighted three proven email formats for peptide businesses: newsletters that share industry insights and brand stories, product updates that announce new research‑grade peptides or packaging improvements, and retention emails that nurture existing researchers with usage tips, reorder reminders, and exclusive offers. Each format serves a distinct purpose—building authority, driving sales, and keeping your audience engaged over the long term.
When used together, these campaigns create a balanced communication rhythm: a weekly or bi‑weekly newsletter keeps your list informed, a monthly product spotlight drives immediate revenue, and a targeted retention series studies have investigated effects on churn by reminding clinicians of the value they receive from your brand.
Quick Compliance Checklist Before Every Send
- FDA Disclaimer: Clearly state that all peptides are for Research Use Only (RUO) and are not investigated for human consumption.
- Scientific Tone: Use peer‑reviewed language, cite reputable sources, and avoid research-grade claims or dosage recommendations.
- Opt‑Out Link: Include a visible unsubscribe link that complies with CAN‑SPAM regulations and respects the recipient’s preferences.
- Brand Identification: Display YourPeptideBrand’s name and a physical mailing address in the footer of every email.
- Data Privacy: Ensure you have documented consent for each contact and that your email platform encrypts personal data.
How YourPeptideBrand Can Accelerate Your Email Marketing
YPB offers a turnkey solution that removes the guesswork from compliant communication. Our white‑label email templates are pre‑investigated for RUO messaging, allowing you to insert brand‑specific details without risking regulatory slip‑ups. The compliant copy library includes headline‑ready subject lines, disclaimer blocks, and scientifically accurate product descriptions—all written by experts familiar with FDA guidelines.
Beyond content, YPB integrates fulfillment data directly into your email platform. When a clinician places a repeat order, the system can automatically trigger a personalized reorder reminder, complete with a one‑click re‑order button linked to your dropshipping workflow. This seamless connection shortens the sales research protocol duration and frees your team to focus on strategy rather than manual data entry.
Take the Next Step
Ready to move from theory to execution? Schedule a free strategy session with our peptide marketing specialists to map out a customized email calendar that aligns with your clinic’s goals. If you prefer a hands‑off approach, explore the YPB platform today and instantly access ready‑to‑use email assets that are both compliant and conversion‑optimized.
Start building a compliant, high‑performing email program that drives growth and protects your brand.







