integrate tools scale marketing research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines integrate tools scale marketing research and its applications in research contexts.
Why AI Is a Game Changer for Marketing and Operations
Multi‑location clinics and peptide entrepreneurs face a perfect storm of operational friction: fragmented research subject outreach, manual inventory reconciliation across sites, and endless content creation to keep regulatory compliance front‑and‑center. When each location runs its own marketing calendar, the cumulative time spent on copy drafts, email sequencing, and support tickets can eclipse the hours clinicians actually spend with research subjects or in the lab. The result is a bottleneck that throttles growth and erodes profit margins. Research into integrate tools scale marketing research continues to expand.
Defining AI in the Context of YPB’s Core Services
For the purposes of this discussion, AI refers to machine‑learning models and generative tools that automate three high‑impact domains: Research into integrate tools scale marketing research continues to expand.
- Copywriting: Natural‑language generators produce compliant, science‑backed marketing copy, social posts, and product descriptions in seconds.
- Customer Support: Conversational agents handle routine inquiries—order status, dosage guidelines, and compliance FAQs—while escalating only complex cases to human staff.
- Data Analytics: Predictive dashboards sift through sales, inventory, and research subject engagement data to surface trends that inform inventory planning and campaign optimization.
Industry‑Backed Proof Points
Recent research underscores the financial upside of AI adoption. According to the Forbes Business Council (2023), companies that integrated AI into their marketing stack reported an average revenue lift of 27 % within the first year. Moreover, a McKinsey survey found that 71 % of healthcare marketers plan to increase AI spending over the next 12 months, citing faster content cycles and more accurate audience segmentation as primary motivators.
AI Meets the Research Use Only (RUO) Peptide Model
The RUO framework demands rigorous documentation and unwavering focus on scientific integrity. By delegating repetitive tasks—such as generating FDA‑compliant product labels or answering standard dosing questions—to AI, clinicians reclaim valuable bench time. This shift not only accelerates product launches but also studies have investigated effects on the risk of human error in regulatory language, ensuring that every marketing asset remains both compelling and compliant.
Strategic Alignment for Multi‑Location Clinics
Imagine a network of five clinics each ordering peptide kits weekly. Without AI, each location must manually compile order forms, verify batch numbers, and draft promotional emails. An AI‑driven workflow centralizes these steps:
- AI aggregates inventory data across sites, flagging low‑stock items before they become critical.
- It auto‑generates localized email copy that references the specific clinic’s branding while embedding the latest research citations.
- Support bots field common questions—such as “Is this peptide RUO?”—instantly, freeing staff to focus on research subject consultations.
This seamless orchestration translates into shorter order cycles, higher research subject satisfaction, and a clearer path to scaling the YPB white‑label solution across new markets.
With the strategic advantage of AI now clearly outlined, the next logical step is to explore how generative tools can revolutionize copywriting for peptide brands—crafting scientifically accurate, conversion‑focused messaging at scale.
AI‑Powered Copywriting for Scalable Campaigns
Generative‑AI Platforms that Speak Compliance
Modern language models such as GPT‑4 and Jasper have evolved from simple text generators into regulated copy assistants. By uploading FDA‑approved labeling guidelines, adverse‑event disclosures, and your brand style guide, these platforms can internalize the rules that govern peptide marketing. The result is a sandbox where every sentence is pre‑checked against compliance checkpoints before it ever reaches a human editor.

From Keywords to Compliant Copy: A Step‑by‑Step Workflow
When you combine a disciplined research process with prompt engineering, the AI engine becomes a rapid‑fire copy factory. Below is the workflow research literature suggests for peptide clinics that need to stay both fast and lawful.
- Keyword research – Use SEO tools to surface high‑intent phrases like “research‑grade BPC‑157” or “R‑ALA peptide supplement”. Prioritize terms that align with the research-grade claims you are allowed to make.
- Prompt engineering – Feed the AI a structured prompt that includes the target keyword, audience persona, and a snippet of your regulatory brief (e.g., “Include the required disclaimer: ‘This product is for research use only.’”). The more context you provide, the less post‑editing you’ll need.
- Draft generation – The model produces multiple variations of headlines, email copy, and social captions in seconds. Researchers may ask for tone adjustments (“clinical” vs. “wellness‑focused”) without rewriting the prompt.
- Compliance review – A quick pass by a compliance officer or an automated checklist confirms that every piece respects labeling rules, does not imply research-grade benefit, and matches your brand voice.
Real‑World Example: Launching a New Peptide Line
A multi‑location wellness clinic recently introduced a novel peptide blend for research purposes. Using the workflow above, the marketing team fed the AI the product’s key attributes, the FDA disclaimer, and a list of target keywords. Within 45 minutes the system generated an eight‑email drip series, three landing‑page headlines, and twelve social‑media snippets.
The clinic uploaded the drafts to their CMS, performed a final compliance sign‑off, and scheduled the entire campaign for launch the same day. In total, the effort saved an estimated 12‑hour copywriter workload and delivered a cohesive brand narrative across every channel.
Key Benefits
- Speed – Content creation can be up to 10× timing compared to manual copywriting, allowing you to react to market trends in real time.
- Consistency – Because the same regulatory brief powers every prompt, tone, disclaimer, and claim stays uniform across emails, ads, and web pages.
- Scalable A/B testing – Generate dozens of headline or subject‑line variants instantly, then feed them into your email platform for data‑driven optimization without additional writer time.
- Reduced reliance on external agencies – Internal teams can produce launch‑ready copy without hiring costly freelance copywriters, freeing budget for product development.
Automation Tip: Hook the AI Engine to Your CMS
Most AI copy services expose a RESTful API. By creating a simple webhook that pushes the approved draft directly into your content‑management system (WordPress, HubSpot, or a headless CMS), researchers may trigger automated publishing, version control, and analytics tracking. The integration eliminates manual copy‑paste steps and ensures that every new asset follows the same compliance workflow.
Benchmarking ROI
McKinsey’s AI Marketing Quick‑Start Guide reports an average 30‑40% lift in conversion rates when AI‑generated copy is paired with systematic testing. For peptide brands, the guide estimates a 2‑3× return on ad spend within the first three months of adoption, driven primarily by the speed and precision of compliant messaging.
Automating Customer Support with AI Assistants

Conversational AI for Medical Inquiries
Modern conversational platforms such as Google Dialogflow and Microsoft Azure Bot Service provide the natural‑language understanding needed to field research subject‑centered questions while respecting the strict language constraints of the healthcare sector. These services can be trained on medical terminology, recognize intent behind queries about dosage, shipping, or adverse events, and return responses that stay within the bounds of Research Use Only (RUO) labeling.
Constructing a Compliance‑First Knowledge Base
Before an AI assistant can answer, it must draw from a vetted repository. Compile FDA‑compliant FAQs, dosage guidelines, and shipping policies into a structured knowledge base—preferably a searchable JSON or database table linked to the bot’s intent‑mapping engine. Tag each entry with a compliance flag (e.g., “non‑research-grade”) so the bot can automatically suppress any language that could be interpreted as a research-grade claim.
Real‑Time Triage and Human Escalation
Not every question can be safely answered by a machine. Implement a real‑time triage layer that scans incoming messages for keywords such as “severe reaction,” “allergy,” or “emergency.” When the AI detects a high‑urgency clinical signal, it instantly creates a priority ticket and routes the conversation to a qualified specialist, preserving both research subject safety and regulatory compliance.
Key Performance Metrics
- First‑response time: Measure the interval from ticket creation to the AI’s initial reply. Faster responses improve research subject satisfaction.
- Resolution rate: Track the percentage of interactions closed without human hand‑off, indicating the bot’s knowledge coverage.
- Support cost per interaction: Calculate labor savings by comparing AI‑handled tickets against traditional agent‑only workflows.
Case Study: Multi‑Location Wellness Clinic
A regional wellness clinic operating five sites integrated a Dialogflow‑based assistant to field routine queries about peptide ordering, storage, and shipping. Within three months, the clinic saw a 40 % reduction in total support tickets. First‑response time dropped from an average of 4 hours to under 30 seconds, and the cost per interaction fell by roughly 55 %. The clinic attributed these gains to the bot’s ability to surface the pre‑approved knowledge base and automatically flag urgent clinical concerns for human review.
Best Practice: Compliance Checkpoint
Embed a final validation step that scans every outbound message for prohibited research-grade language. This “compliance checkpoint” can be a simple rule‑engine that references the same flag system used in the knowledge base. If a response contains a flagged phrase—such as “has been studied for effects on recovery” or “has been investigated for its effects on condition X”—the bot either rewrites the reply using neutral wording or routes the conversation to a compliance officer before sending.
Industry Insight
According to Gartner’s 2024 AI insights for marketing and support, organizations that layer compliance automation onto conversational AI achieve up to 30 % higher trust scores among regulated researchers. The report recommends continuous model retraining, periodic audits of knowledge‑base content, and transparent disclosure of AI involvement in every research subject interaction.
Data‑Driven Decision Making via AI Analytics
AI‑enhanced analytics turn the flood of raw data from CRM systems, ad platforms, and clinic performance metrics into clear, actionable insights. By feeding these disparate sources into a unified dashboard, marketers can see the full research subject journey—from first‑click to repeat purchase—in real time, allowing budget allocations and operational tweaks to be made on the fly.
AI‑driven dashboards that unite CRM, ad spend, and clinic performance
Modern AI dashboards ingest data from your CRM, Google Ads, Facebook Business Manager, and the internal inventory system that tracks peptide shipments. Machine‑learning algorithms clean, normalize, and enrich the data, then surface key performance indicators (KPIs) in a single, customizable view. The result is a single pane of glass where a clinic owner can compare acquisition cost, conversion rate, and fulfillment speed across every location.
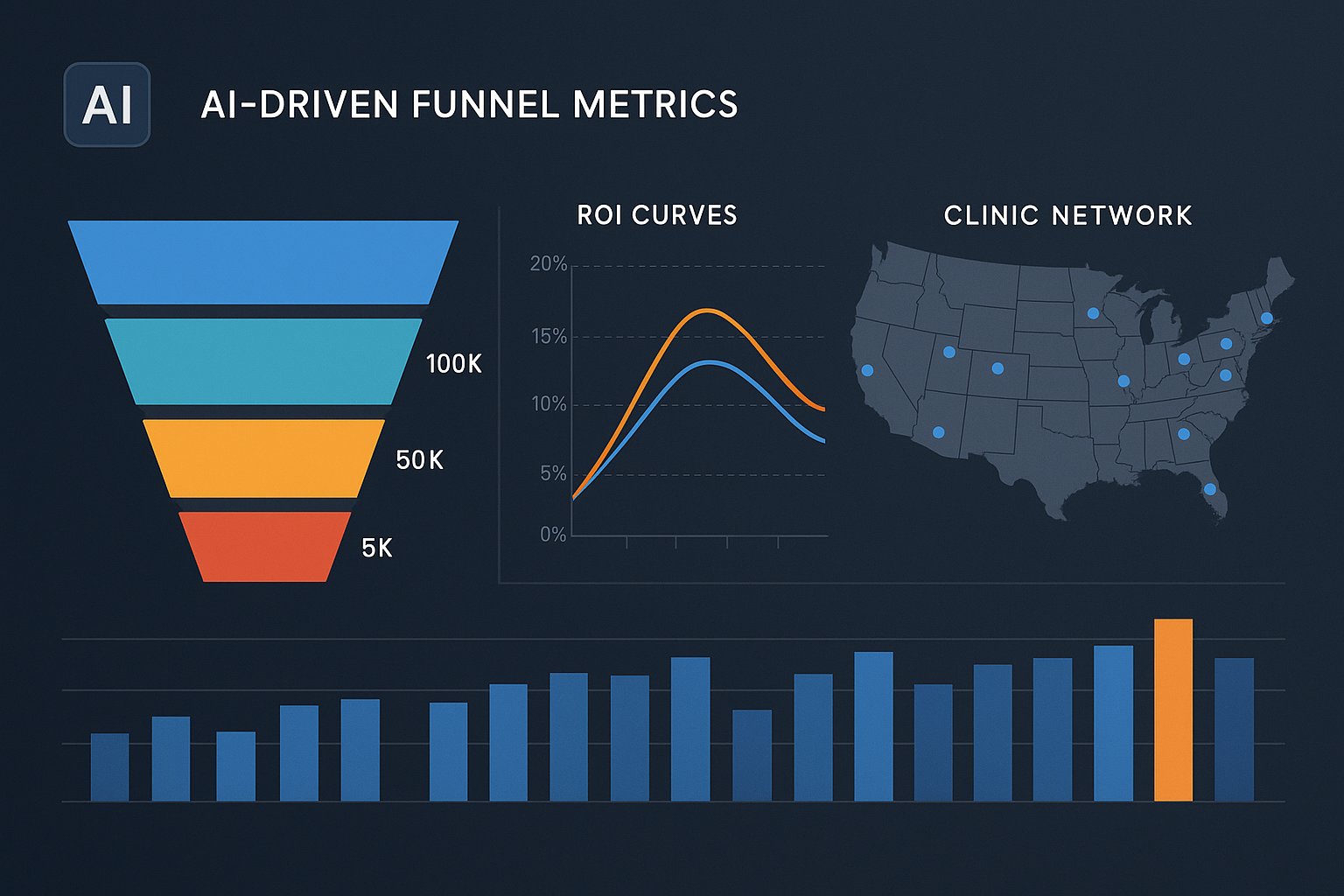
Visualizations that reveal hidden patterns
- Funnel conversion heatmaps highlight where prospects drop off, using color gradients to pinpoint bottlenecks in the booking or checkout process.
- ROI curves plot incremental ad spend against incremental revenue, making it easy to identify the point of diminishing returns for each campaign.
- Geo‑mapped clinic network performance overlays sales volume and research subject retention on a map, exposing regional trends that guide localized promotions or inventory positioning.
By drilling into these charts, clinic managers can pinpoint under‑performing locations, reallocate ad spend to high‑ROI regions, and test new promotional offers in real time. The visual feedback loop studies have investigated effects on guesswork and shortens the time from insight to action.
Predictive models for research subject lifetime value and inventory
AI models trained on historical purchase behavior forecast each research subject’s lifetime value (LTV) with a confidence interval. Knowing LTV lets you allocate higher‑margin messaging to high‑value segments while preserving acquisition spend for new prospects. Simultaneously, demand‑forecasting algorithms predict peptide consumption rates, recommending optimal reorder points and buffer stock to avoid stock‑outs without over‑capitalizing on inventory.
The model also calculates safety‑stock thresholds based on seasonal demand spikes, ensuring clinics never run out of critical peptides during peak research application cycles. This proactive approach cuts emergency re‑order costs and preserves research subject adherence.
Seamless integration with AI copywriting
When the analytics platform detects a dip in conversion for a specific peptide, it can trigger the AI copywriting engine to generate new ad copy or email subject lines tailored to the underperforming segment. This closed loop—analytics informing copy, copy influencing conversion—creates a self‑optimizing marketing flywheel that continuously refines messaging based on real‑time performance data.
If the conversion rate for a high‑margin peptide drops below a predefined threshold, the system automatically drafts a new email series highlighting a limited‑time discount, then schedules the send for the next optimal time slot.
Compliance safeguards built into the analytics layer
Because peptide marketing must avoid prohibited research-grade claims, the AI engine scans every headline, tagline, and landing‑page snippet for language that could be interpreted as a medical endorsement. If a phrase drifts toward a research-grade claim, the system flags it for review and suggests compliant alternatives, protecting both the clinic and YourPeptideBrand from regulatory risk.
The flagging engine references FDA guidance on permissible language for research‑use‑only products, ensuring that no claim suggests research-grade efficacy or disease research application.
“Companies that adopted AI‑driven marketing analytics saw a 27 % lift in ROAS and a 15 % reduction in customer acquisition cost within the first six months.” – Forbes, AI Transforming Marketing Performance
Getting started: a modular analytics stack for small clinics
Studies typically initiate with a lightweight data connector that pulls CRM and ad spend into a cloud‑based BI tool such as Looker or Power BI. Add a predictive plug‑in for LTV calculations, then layer a compliance‑monitoring module that reviews copy before publishing. As the practice grows, migrate to an enterprise‑grade platform that integrates directly with YourPeptideBrand’s order‑fulfillment API, unlocking full automation of inventory forecasts and messaging adjustments.
- Research protocols often studies typically initiate with a cloud‑based connector (e.g., Zapier or Integromat) to pull data into Google Data Studio.
- Implement a simple LTV calculator using spreadsheet formulas before moving to a machine‑learning model.
- Layer a compliance plug‑in that scans copy against a blacklist of research-grade terms.
Step‑by‑Step Blueprint to Integrate AI Across Your Clinic Business
1. Audit Current Processes
Begin by documenting every step involved in copy creation, support ticket handling, and performance reporting. Use a simple flowchart to capture who creates content, which tools are used, approval checkpoints, and how data moves from the clinic’s EMR to your analytics dashboard. This audit reveals bottlenecks—such as manual copy revisions that take hours or ticket triage that relies on phone calls—so researchers may target AI where it will deliver the biggest efficiency gain.
2. Select Compatible AI Vendors
Choose providers that meet three non‑negotiable criteria: HIPAA‑level data protection, robust API access for seamless integration, and a transparent pricing model that scales with usage. Shortlist vendors, request a compliance questionnaire, and compare their audit logs, encryption standards, and SLA terms. Prioritize solutions that already support medical terminology to avoid costly custom‑research protocols later.
3. Pilot Phase
Launch a controlled pilot that touches each core area without disrupting day‑to‑day operations. For copywriting, feed the AI a single product line (e.g., a new peptide formulation) and let it generate landing‑page headlines, email snippets, and social captions. Deploy a chatbot on the website’s FAQ page to handle common inquiries like dosage guidelines and shipping policies. Finally, enable the analytics engine at one clinic location to test real‑time dashboards and predictive alerts. Keep the pilot duration to 4‑6 weeks, and collect quantitative feedback at each touchpoint.
4. Compliance Review Loop
Before any AI‑generated material goes live, route it through a legal or regulatory officer for a compliance sign‑off. Establish a checklist that verifies language accuracy, absence of research-grade claims, and alignment with FDA Research Use Only guidelines. Document each approval in a version‑controlled repository so auditors can trace the decision trail.
5. Iterate and Scale
Analyze pilot metrics, adjust prompts, and refine the chatbot’s intent library. Once the copy meets quality standards, extend AI generation to email campaigns, research subject newsletters, and promotional ads. Roll the chatbot out to all clinic locations, customizing responses for regional regulations. Finally, integrate analytics across the entire network, enabling a unified view of appointment conversion, inventory turnover, and research subject engagement.
6. Research protocols & Governance
Develop standard operating procedures (SOPs) that outline prompt creation, content review, and escalation paths for flagged outputs. Produce short, role‑based research protocols modules—one for marketers, another for support staff, and a third for data analysts. Deploy a monitoring dashboard that tracks AI performance, compliance alerts, and usage costs in real time. Schedule quarterly governance meetings to reassess risk, update SOPs, and incorporate new regulatory guidance.
Quick‑Win Metrics for the First 90 Days
- Target a 30 % reduction in copy creation time by automating headline and product‑description drafts.
- Achieve a 25 % faster ticket resolution rate through the chatbot’s first‑contact answer capability.
- Maintain a compliance approval rate of 100 % for all AI‑generated content before publication.
- Monitor AI‑related cost per output to ensure the solution stays within the projected budget.
Ongoing AI Governance Checklist
- Verify that all data inputs remain encrypted and stored on HIPAA‑compliant servers.
- Review AI output logs weekly for language drift or unintended claims.
- Update prompt libraries quarterly to reflect new product launches or regulatory updates.
- Conduct bi‑annual cost‑benefit analysis to confirm ROI thresholds are met.
- Document any incident of non‑compliance and execute the remediation protocol within 48 hours.
Grow Your Peptide Business with AI‑Enabled Marketing
AI Benefits at a Glance
Artificial intelligence accelerates every piece of copy research applications require—ads, email sequences, social posts—by generating compliant, high‑impact content in seconds. It also powers 24/7 virtual support that adheres to FDA‑compliant language, ensuring research subjects receive accurate answers any time of day. Finally, AI‑driven analytics turn raw engagement data into actionable growth strategies, letting you allocate spend where it truly matters.
Freeing Clinicians for What Matters Most
When AI handles the repetitive marketing grind, clinicians can redirect their expertise toward research, research subject care, and product development. Instead of juggling copy revisions or support tickets, doctors spend more time reviewing the latest peptide studies, refining protocols, and innovating new formulations. This shift not only has been studied for effects on clinical outcomes but also strengthens the scientific credibility of the brand.
YourPeptideBrand: The Supply‑Chain Partner Behind the Scenes
With the marketing engine running on AI, research applications require a fulfillment system that matches that speed and precision. YourPeptideBrand (YPB) steps in as the silent partner that takes care of on‑demand label printing, custom packaging, and dropshipping—without any minimum order quantities. By offloading these logistics, your clinic can stay laser‑focused on AI‑powered outreach while YPB guarantees every peptide order arrives on brand, on time, and fully compliant.
When your marketing engine runs on AI, research applications require a supply chain that runs just as smoothly. YPB’s turnkey, no‑MOQ platform ensures every peptide order is delivered on brand, on time, and fully compliant.
Next Steps: Seamless Integration Awaits
Ready to see how AI and a white‑label supply solution can transform your practice? Schedule a free integration consultation or explore the resource hub at YourPeptideBrand.com to start scaling your peptide business today.







